What’s Happening At The McGowan Institute?
September 2012 | VOL. 11, NO. 9 | www.mcgowan.pitt.edu
New Biomimetic Controlled-Release Capsules May Help in Gum Disease
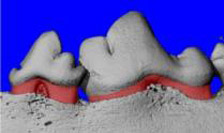 Progress is being made in opening a new front in the battle against gum disease, the leading cause of tooth loss in adults and sometimes termed the most serious oral health problem of the 21st century.
Progress is being made in opening a new front in the battle against gum disease, the leading cause of tooth loss in adults and sometimes termed the most serious oral health problem of the 21st century.
“Our technology uses controlled-release capsules filled with a protein that would be injected in the pockets between the gums and the teeth,” said McGowan Institute for Regenerative Medicine faculty member Steven Little, PhD, chair of the Department of Chemical and Petroleum Engineering, associate professor, and Bicentennial Alumni Faculty Fellow of the University of Pittsburgh Swanson School of Engineering, who reported on the research at the 244th National Meeting & Exposition of the American Chemical Society.
“That’s ground-zero for periodontal disease ? ‘gum disease’ ? the place where bacteria breed and inflammation occurs. The capsules dissolve over time, releasing a protein that acts as a homing beacon. It guides immune cells to the diseased area, reducing inflammation, creating an environment that fights the disease process and even could create conditions favorable for gum tissue to regrow.”
Dr. Little and University of Pittsburgh colleagues including McGowan Institute for Regenerative Medicine faculty member Charles Sfeir, DDS, PhD, associate professor at Pitt’s Department of Oral Medicine and Pathology, have evidence from pre-clinical laboratory experiments that the approach does foster healing and regrowth of gum tissue damaged by periodontal disease.
A bacterial infection causes periodontal disease. It first appears as mild tenderness and bleeding of the gums. It leads to inflammation and, if left untreated, can damage the gums so that they recede and lose their attachment to the teeth. It may progress even further and damage bone and other tissues that hold teeth firmly in place. Surprisingly, gum disease has a number of deleterious effects outside the mouth, with some studies linking inflammation in the gums to an increased risk of heart disease, stroke, and preterm delivery in pregnant women.
Treatment includes scaling, root planing, and other procedures to remove the plaque and bacteria that have accumulated in pockets between the teeth and gums. Dentists may combine this with antibiotics to fight the bacteria involved in gum disease.
Many scientists are seeking alternative treatments that kill the bacteria. Dr. Little’s group is taking an entirely different approach. They are targeting the inflammation process. “Although bacteria start the disease, inflammation is what keeps it going and causes progressive damage,” Dr. Little explained.
To reduce inflammation at the gums, Dr. Little and colleagues designed injectable controlled-release capsules containing a protein encased inside a plastic-like polymer material. The polymer is already used in medicine in dissolvable sutures. After the capsules are injected, the polymer slowly breaks down, releasing the protein encapsulated inside. The protein, termed a chemokine, is already produced by the body’s existing cells in order to summon specialized white blood cells to a specific site. Scientists previously tried to keep those cells, termed lymphocytes, away from the gums so as to block inflammation from occurring in the first place.
“It seems counterintuitive to lure in a lymphocyte, which is traditionally thought of as an inflammatory cell, if there’s inflammation,” Dr. Little pointed out. “But remember that a certain level of natural inflammation is required to fight off an infection. Inflammation is inherently a good thing, but too much of it is a bad thing. That’s why we aim to restore the immune balance, or homeostasis.”
During the pre-clinical tests, Dr. Little’s team injected the capsules and discovered evidence that disease symptoms are dramatically reduced and that proteins and other substances involved in regrowth of gum tissue had appeared. Dr. Little said that this finding offers encouragement that the treatment could not only rebalance the immune system, but also prompt regrowth of lost gum and bone tissue in the mouth.
OPPORTUNITIES
Request for Proposals: $100,000 Awards for Translational Research
 The Wallace H. Coulter Translational Research Partners II (TPII) Program at the University of Pittsburgh funds translational research projects in healthcare, with the goal of accelerating the introduction of new technologies into patient care to address unmet clinical needs. Examples of desirable translational research projects and outcomes include systems and devices for the improved diagnosis and treatment of disease including both diagnostic and therapeutic solutions.
The Wallace H. Coulter Translational Research Partners II (TPII) Program at the University of Pittsburgh funds translational research projects in healthcare, with the goal of accelerating the introduction of new technologies into patient care to address unmet clinical needs. Examples of desirable translational research projects and outcomes include systems and devices for the improved diagnosis and treatment of disease including both diagnostic and therapeutic solutions.
Application criteria include:
- One Co-PI with a faculty appointment in the Department of Bioengineering
- One Co-PI who is a clinical practitioner in Pitt’s Schools of the Health Sciences
- Student-sourced projects are eligible to apply
- Research must translate to applications in health care
- Outcome benefits patients when technology/therapy is commercialized
Evaluation of each proposal will be on the basis of scientific and clinical merit, potential health care impact and significance, experience of the investigative co-PIs and collaborators, timeline and pathway to commercialization, and potential for obtaining further financial investment to translate the particular innovative technology to a healthcare solution.
The project funding period is July 1, 2013, through June 30, 2014. The deadline for your letter of intent is November 7, 2012. This 1-page document must identify the business potential of the proposed healthcare innovation.
Please contact Lindsay Rodzwicz, Coulter Program Administrator, with your questions. She can be reached at (412) 624-3495 or email: rodzwicz@pitt.edu.
Regenerative Medicine and Rehabilitation Meeting in Pittsburgh
 Building on last year’s overwhelming success, the McGowan Institute for Regenerative Medicine is very excited to announce the Second Annual Symposium on Regenerative Rehabilitation, to be held in Pittsburgh, Pennsylvania, on November 12-13, 2012.
Building on last year’s overwhelming success, the McGowan Institute for Regenerative Medicine is very excited to announce the Second Annual Symposium on Regenerative Rehabilitation, to be held in Pittsburgh, Pennsylvania, on November 12-13, 2012.
This symposium, the only one of its kind, represents a unique opportunity to bring together scientists and clinicians in the fields of Regenerative Medicine and Rehabilitation. This symposium has the goal of initiating new insights and discussions as to how to best synergize the two fields, with a mutual benefit to each, and most importantly, to the patients these therapies affect. Rehabilitation specialists possess a unique cadre of tools that may be of great benefit to regenerative medicine scientists as they develop exciting new therapies. Are regenerative medicine scientists in tune to the host of clinically available approaches that may enhance the efficacy of their biological therapies?
Scott Rodeo, MD, associate team physician for the current Super Bowl Champions the New York Giants, physician for the USA Olympic Swimming Team, and professor, orthopaedic surgery, Weill Medical College of Cornell University, during his Keynote Address will discuss the integration of regenerative medicine approaches for soft tissue healing after sports injuries.
Other sessions will include:
- Pre-Conference Workshop (optional)—Regenerative Medicine 101
- Mechanotransduction as a Tool in Tissue Healing
- Current Controversies in Regenerative Medicine
- The Impact of Rehabilitation on Cellular Therapies
- Regenerative Rehabilitation in Military Medicine
- Training the Transplant: Implications for the Treatment of Neurological Pathologies
- Regenerative Rehabilitation in Education
- Clinical Innovations in Regenerative Rehabilitation (plenary sessions on brain computer interfaces, biological scaffolds, and modeling of inflammation)
In response to attendee feedback from last year, the number of plenary sessions has increased, which have been designed to highlight the scientist to practitioner continuum, and will include presentations from the regenerative medicine scientist, the rehabilitation specialist, and patient testimonials. A scientific poster session will allow for a lively discussion and may help promote new ideas and collaborations. Poster abstract submissions are now being accepted!!
Thanks to the generous support of the National Institute of Child Health & Development, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, and the National Institute of Neurological Disorders and Stroke of the NIH, travel awards are available to attend this year’s symposium. These awards are available to students, fellows, residents, post-doctoral fellows, and junior investigators. Please visit the Travel Grant tab on the symposium website for more details on how to apply.
We hope you will join us for what promises to be an exciting event. For more information regarding the agenda, registration, abstract submission, or travel award application.
Pittsburgh Played Host to TMJ Bioengineering Conference
 The McGowan Institute for Regenerative Medicine was one of several co-sponsors of the TMJ Bioengineering Conference, Pittsburgh, Pennsylvania, held September 20-22, 2012. The meeting provided a unique opportunity for bioengineers, clinicians, and scientists to discuss the challenges presented by temporomandibular joint disorders (TMJD, in the medical literature TMD, or TMJ syndrome) and to devise strategies to address those challenges.
The McGowan Institute for Regenerative Medicine was one of several co-sponsors of the TMJ Bioengineering Conference, Pittsburgh, Pennsylvania, held September 20-22, 2012. The meeting provided a unique opportunity for bioengineers, clinicians, and scientists to discuss the challenges presented by temporomandibular joint disorders (TMJD, in the medical literature TMD, or TMJ syndrome) and to devise strategies to address those challenges.
The overall meeting objective was to organize a scientific conference exclusively dedicated to the TMJ, and to advance the field of TMJ research and strengthen the continuity among clinicians, scientists, and bioengineers in this pursuit. This overall objective was addressed by the following specific objectives:
- Provide recommendations to the National Institutes of Health’s National Institute of Dental and Craniofacial Research TMJD Program on novel scientific approaches and critical research topics that will advance the field.
- Establish a benchmark for current treatments and scientific knowledge.
- Bring outside perspective from experts in areas pertinent to TMJ research.
TMJD is an umbrella term covering acute or chronic pain, especially in the muscles of mastication and/or inflammation of the TMJ, which connects the mandible to the skull. The primary cause is muscular hyper- or para-function, as in the case of bruxism, with secondary effects on the oral musculoskeletal system, like various types of displacement of the disc in the TMJ. The disorder and resultant dysfunction can result in significant pain, which is the most common TMD symptom, combined with impairment of function. Because the disorder transcends the boundaries between several health-care disciplines — in particular, dentistry and neurology — there are a variety of treatment approaches.
The TMJ is susceptible to many of the conditions that affect other joints in the body, including ankylosis, arthritis, trauma, dislocations, developmental anomalies, neoplasia, and reactive lesions. Signs and symptoms of TMJD vary in their presentation and can be very complex, but are often simple. On average the symptoms will involve more than one of the numerous TMJ components: muscles, nerves, tendons, ligaments, bones, connective tissue, and the teeth. Ear pain associated with the swelling of proximal tissue is a symptom of TMJD.
SCIENTIFIC ADVANCES
Excellence, Strong Research Help UPMC Reach 3,000 Heart, Lung Transplants
 William Ferry almost never made it to UPMC for the transplant that would eventually save his life. Nervous about a surgery that had been performed so little in 1980, Ferry contemplated turning the car around and not showing up when word came that a donor heart had been found for him.
William Ferry almost never made it to UPMC for the transplant that would eventually save his life. Nervous about a surgery that had been performed so little in 1980, Ferry contemplated turning the car around and not showing up when word came that a donor heart had been found for him.
More than 30 years later, Mr. Ferry is the longest surviving heart transplant recipient at UPMC. And now he can add a new distinction to his name: One of the 3,000.
Recently, UPMC became only the second transplant center in the country to have performed 3,000 heart and lung transplants. The 3,000 include patients from 43 states and 9 countries and sets UPMC apart as a leader not only in clinical excellence, but also in research. UPMC is one of only a few centers in the country that can boast heart recipients who are at or beyond 30 years post-transplant and also one of the few that performs combined heart/lung transplants.
“What distinguishes us from other heart and lung transplant programs is our years of experience and that’s why this milestone is so important to us. We are tremendously proud of the work we do here,” said Christian Bermudez, MD, chief of cardiothoracic transplantation at UPMC and a McGowan Institute for Regenerative Medicine faculty member.
A pioneer in transplantation since Dr. Thomas E. Starzl, MD, PhD, came to Pittsburgh in the 1980s, UPMC performed its first heart transplant in June 1980 and first combined heart and lung transplant in May 1982. Its first lung transplant occurred in April 1985, and the area’s first double-lung transplant happened at UPMC 3 years later.
Over the years, UPMC helped in the development of mechanical assist devices used as a bridge to transplant for heart and lung patients, pioneered minimally invasive lung transplant techniques, performed lobectomies when large donor lungs could not fit into smaller patients, and often treated high-risk recipients turned away from other centers because of diseases such as scleroderma and cystic fibrosis. Also, its association with the University of Pittsburgh School of Medicine led to important research developments in the area of immunosuppressive medications, or anti-rejection drugs, which has transformed the way recipients live their lives after transplants.
“Being an academic medical center, our patients have access to cutting-edge medical trials and devices that they might not get elsewhere,” said Dennis McNamara, MD, director of UPMC’s Heart Failure Transplantation Program and a McGowan Institute for Regenerative Medicine affiliated faculty member.
Eleven years after his transplant, heart recipient Mr. Ferry returned to UPMC for a second heart transplant. He was in the hospital for just 2 months after his surgery; the first time, it had been 6 months. And even now, he only takes four pills a day, compared to 30 a day he took after his first transplant.
“That was breakfast, all those pills,” joked Mr. Ferry, now 69, of Volant, Pa.
Another unique aspect of UPMC’s cardiothoracic transplant program is its patient-centered approach to care, said Joseph Pilewski, MD, medical director of UPMC’s Lung Transplantation Program. Patients who come in for a transplant not only meet with a surgeon, but also a team made up of pulmonary physicians, nurses, social workers, and pharmacists all dedicated to their care, Dr. Pilewski said. The same team provides comprehensive post-transplant care to minimize complications and optimize outcomes.
“We spend a lot of time with our patients and they really become like family to us,” Dr. Bermudez said. “They entrust their care to us, and we take that responsibility very seriously.”
Gretchen Warner became one of the 3,000 in November 2011. The Kentucky resident suffers from scleroderma and other hospitals had refused her a transplant because of complications from the disease. She couldn’t walk far without getting out of breath and came to UPMC for a double lung transplant.
Ms. Warner’s condition worsened and doctors performed a lobectomy on a pair of large donor lungs that didn’t originally fit her frame in hopes of keeping her alive. The rare surgery worked, and today Ms. Warner is back home with her husband and extended family, living life to the fullest.
“I do Zumba two times a week and try and walk a mile every day,” Ms. Warner said.
 Targeted Oxidation-Blocker Prevents Secondary Damage after Traumatic Brain Injury
Targeted Oxidation-Blocker Prevents Secondary Damage after Traumatic Brain Injury
According to McGowan Institute for Regenerative Medicine affiliated faculty members (pictured left and right)
- David Okonkwo, MD, PhD, assistant professor with the Department of Neurological Surgery, University of Pittsburgh Medical Center, director of Neurotrauma and of the Spinal Deformity Program, clinical director of the Brain Trauma Research Center, and associate director of the Center for Injury Research and Control, and
- Valerian Kagan, PhD, professor and vice-chairman in the Department of Environmental and Occupational Health as well as a professor in the Department of Pharmacology and the Department of Radiation Oncology at the University of Pittsburgh, and also the director of the Center for Free Radical and Antioxidant Health, and
a research team from the University of Pittsburgh School of Medicine, Graduate School of Public Health, and Department of Chemistry in a report published online in Nature Neuroscience, treatment with an agent that blocks the oxidation of an important component of the mitochondrial membrane prevented the secondary damage of severe traumatic brain injury and preserved function that would otherwise have been impaired.
Annually, an estimated 1.7 million Americans sustain a traumatic brain injury (TBI) due to traffic accidents, falls, assaults, and sports participation, said the study’s senior author Hülya Bay?r, MD, associate professor, Department of Critical Care Medicine, University of Pittsburgh School of Medicine. She added that 52,000 of those injured die, and 85,000 are left with significant disability.
“We don’t yet have a specific therapy for TBI, but can provide only supportive care to try to ease symptoms,” Dr. Bay?r said. “Our study drug shows promise as a neuroprotective agent that might help address this important public health problem.”
For the study, the research team conducted a global assessment of all the phospholipids in rat brain cells. This revealed that damage from TBI was nonrandom and mostly involved cardiolipin, a phospholipid that is found in the membranes that form mitochondria, the cell’s powerhouse. They noted that in the healthy animal, only 10 of the 190 cardiolipin species were modified by oxygen, but after a brain injury, the number of oxidized species rose many-fold.
The researchers then developed an agent, called XJB-5-131, which can cross the blood-brain barrier and prevent the oxidation of cardiolipin. Using an established research model of severe TBI, the agent or a placebo was injected into the bloodstream of rats 5 minutes and again 24 hours after head injury.
In the weeks that followed, treated animals performed akin to normal on tests of balance, agility, and motor coordination, learning, and object recognition, while placebo-treated animals showed significant impairment. The results indicate that blocking cardiolipin oxidation by XJB-5-131 protected the brain from cell death.
“The primary head injury might not be that serious,” Dr. Bay?r noted. “But that initial injury can set into motion secondary cellular and molecular events that cause more damage to the brain and that ultimately determine the outcome for the patient.”
She added that a targeted oxidation-blocker might also be beneficial in the treatment of other neurological disorders, such as Parkinson’s disease, amyotrophic lateral sclerosis, or ALS, and stroke.
Sperm Precursor Cells Made in the Lab Could One Day Restore Male Fertility
 McGowan Institute for Regenerative Medicine affiliated faculty member Gerald Schatten, PhD, professor and vice chair of obstetrics, gynecology and reproductive sciences at the University of Pittsburgh, a deputy director at Magee-Womens Research Institute, and director of the Pittsburgh Development Center, was a member of the team of researchers at the University of Pittsburgh School of Medicine who found that human embryonic stem cells (hESCs) and human induced pluripotent stem cells (hiPSCs) can be coaxed into becoming precursor sperm cells, suggesting that it might be possible one day to restore fertility for sterile males with an easily obtained skin sample. Their findings are now available in the online version of Cell Reports.
McGowan Institute for Regenerative Medicine affiliated faculty member Gerald Schatten, PhD, professor and vice chair of obstetrics, gynecology and reproductive sciences at the University of Pittsburgh, a deputy director at Magee-Womens Research Institute, and director of the Pittsburgh Development Center, was a member of the team of researchers at the University of Pittsburgh School of Medicine who found that human embryonic stem cells (hESCs) and human induced pluripotent stem cells (hiPSCs) can be coaxed into becoming precursor sperm cells, suggesting that it might be possible one day to restore fertility for sterile males with an easily obtained skin sample. Their findings are now available in the online version of Cell Reports.
Infertility can be a side effect of some cancer treatments because the drugs work by destroying rapidly-dividing cells, which includes sperm precursor cells, explained the study’s lead author Charles Easley, PhD, formerly a post-doctoral fellow in the Department of Obstetrics, Gynecology and Reproductive Sciences at the University of Pittsburgh School of Medicine, and now a faculty member at Emory University.
“Sperm can be banked for future artificial insemination procedures, but that does not help some patients, such as pre-pubertal boys,” Dr. Easley said. “There are procedures to store testicular tissue prior to cancer therapy, but men who didn’t have the opportunity to save tissue are permanently sterile, and so far there are no cures for their sterility.”
There is growing research evidence that adult somatic cells, such as those of the skin, can be induced or biologically prodded to return to a more primitive state and then redirected to become different cell types. To see if it was possible to derive sperm cells Dr. Easley and his colleagues cultured lab-grade hiPSCs from commercially available skin samples, as well as hESCs from established cell lines, in conditions typically used to sustain spermatogonial stem cells.
They found that both kinds of stem cells were able to generate key cells, including the spermatogonial stem cells, spermatocytes containing a full complement of chromosomes prior to cell division known as meiosis, then post-meiotic spermatocytes with half the chromosome number, and round spermatids, which are precursors to sperm. Testing of certain chromosome sites showed correct parent-of-origin genomic imprints in these haploid cells as well, the researchers noted.
“No one has been able to make human sperm from pluripotent stem cells in the lab, but this research indicates it might be possible,” Dr. Easley said. “This model also gives us a unique opportunity to study the molecular signals that govern the process, allowing us to learn much more about how sperm are made. Perhaps one day this will lead to new ways of diagnosing and treating male infertility.”
AWARDS AND RECOGNITIONS
Regenerative Medicine Awards
 The McGowan Institute for Regenerative Medicine congratulates the following award winners honored at this year’s Tissue Engineering International & Regenerative Medicine Society (TERMIS) World Congress – Vienna, Austria:
The McGowan Institute for Regenerative Medicine congratulates the following award winners honored at this year’s Tissue Engineering International & Regenerative Medicine Society (TERMIS) World Congress – Vienna, Austria:
- TERMIS-AM Senior Scientist Award – Stephen F. Badylak, DVM, PhD, MD
- TERMIS-AM Educational Award – Kacey G. Marra, PhD
- Mary Ann Liebert, Inc. Outstanding Student Award – Brian M. Sicari
- TERMIS-AM Lifetime Achievement Award – Alan J. Russell, PhD
- TERMIS-AM Young Investigator Award – Karen L. Christman
- TERMIS-AM Innovation/Commercialization Award – Andrew J. Marshall
TERMIS-AM Senior Scientist Award – Stephen F. Badylak, DVM, PhD, MD
Stephen F. Badylak, DVM, PhD, MD, a deputy director of the McGowan Institute for Regenerative Medicine, professor in the Department of Surgery, and director of the Center for Pre-Clinical Tissue Engineering within the Institute, is a pioneer in the use of inductive biologic scaffolds for tissue reconstruction. His discoveries and 250 plus peer-reviewed publications have helped develop an important medical device industry that has changed the practice of medicine and more specifically, surgery. He is an international thought leader, has helped organize TERMIS as a global regenerative medicine society, and his scientific and organizational skills are heavily sought after by peers, regulatory agencies, and international policy makers. At the same time, Dr. Badylak’s scientific and patient-oriented discoveries have driven the clinical translation of biologic scaffolds for tissue repair and reconstruction – his passion for clinical translation is a notable driving force. His work spans the entire range of basic science, preclinical studies, and patient care.
Dr. Badylak is clearly the father of biologic scaffolds which forms the basis of an entire segment of the fields of tissue engineering and biomaterials. While his more than 250 scientific publications and 50 US patents are one metric of measuring achievement, another is the impact of his work on the practice of medicine and surgery. More than 2 million patients have benefitted from his earliest discovery of small intestinal submucosa (SIS) as biologic material and his subsequent work relating matrix-mediated tissue reconstruction to developmental biology, stem cell biology, and clinical practice continues to lead the field. His work has impacted not only general surgery but also plastic/reconstructive, thoracic, and trauma surgery, gastroenterology, obstetrics and gynecology, urology, dermatology, orthopedics, and wound care. Arguably, his work represents the most successful and widely utilized tissue engineering technique that has ever reached clinical practice. Of central importance has been his recognition of the role of regulatory, reimbursement, and industry activities which will help provide a roadmap for the advancement of medical research to the bedside. His proven ability to promote interdisciplinary collaboration, recognize cross disciplinary applications of scientific discovery, and challenge existing paradigms, while maintaining the highest degree of scientific integrity and ethics, sets a standard for the field.
The Senior Scientist Award is based on an individual’s significant contributions to the tissue engineering and regenerative medicine field. Individuals nominated for the Senior Scientist Award must have received their terminal degree 10 years or greater from the time of the award, be actively contributing to the tissue engineering and regenerative medicine field, and their contributions as a body of work should be well recognized within the community.
TERMIS-AM Educational Award – Kacey G. Marra, PhD
Kacey G. Marra, PhD, associate professor, Department of Surgery, director of the Plastic Surgery Laboratory, as well as co-director of the Adipose Stem Cell Center, shares a special bond with every student she teachers and advises. Dr. Marra’s likeable personality applied to her motivational work ethic is what defines her and qualifies her as an extraordinary mentor and educator. It is not surprising that this driven and charismatic woman successfully manages a productive research laboratory. Thanks to Dr. Marra’s positive attitude and problem-solving strategies, students are always able to make ends meet and overcome any potential obstacles.
Dr. Marra teaches an Introduction to Tissue Engineering course. She increased the course’s student capacity after an overwhelming number requested to experience the class before graduating. While the class did, of course, supply students with knowledge and concepts of tissue engineering and regenerative medicine, Dr. Marra’s above-and-beyond teaching methods provided them with valuable public speaking skills they utilize to this day. Dr. Marra’s vibrant and lively teaching practices engage her entire class (which now consists of over 40 undergraduate and graduate students from Pitt’s bioengineering, chemical engineering, and pathology departments). Dr. Marra positively affects the lives and education of her students.
Dr. Marra has inspired countless young minds in the local community to pursue science, math, and engineering degrees. In addition to her work with the Pittsburgh Tissue Engineering Initiative (just one of the numerous local outreach programs to which she is dedicated), Dr. Marra recently spoke to hundreds of high school students through a Westinghouse Science series. The day after her lecture to these students, Dr. Marra’s e-mail inbox exploded with messages from audience members indicating interest to shadow or intern in the Adipose Stem Cell Center.
The Educational Award is presented based on the educational accomplishments of an individual (advisor/supervisor) within the field of tissue engineering and regenerative medicine. Educational accomplishments include but are not limited to: published textbooks, new initiatives within the field, mentoring, curriculum development, educational resources, educational collaborations, etc.
Mary Ann Liebert, Inc. Outstanding Student Award – Brian M. Sicari
Brian M. Sicari is a doctoral student at the University of Pittsburgh in the field of Cellular and Molecular Pathology. He has shown an enthusiasm for both learning and the application of that learning to medical problems that others typically consider “too tough” to tackle. He has spent these past 4 years becoming proficient in the scientific method, developing new laboratory techniques, applying the principles of engineering and cell biology to practical and very real problems in skeletal muscle pathology, and becoming a most well-rounded scientist. He challenges himself, his peers, and superiors, and does not accept traditional explanations for accepted paradigms without solid supporting data.
Mr. Sicari’s thesis work, “Constructive Remodeling by Extracellular Matrix Scaffolds within the Aging Skeletal Muscle Microenvironment,” is not only relevant to an aging demographic in the United States and worldwide, it also addresses fundamental principles of soft tissue healing, developmental biology, and the effects of age upon such fundamental processes. His work builds upon the evidence-based research upon the strategic use of extracellular matrix to promote skeletal muscle regeneration, including the associated innervation, vascularization, and functionality of such new muscle tissue. His work focuses upon the loss of progenitor cells in an older population, the role of macrophages in this process, and mechanisms to delay or reverse this degenerative process. In the process of this work, he has also developed a new model of volumetric muscle loss.
Mr. Sicari has been the recipient of several well-deserved awards and fellowships during his student experience, including the 2011 Armed Forces Institute for Regenerative Medicine Career Enrichment Award, 2011 University of Pittsburgh Pathology Retreat First Place Poster Award, 2012 National Institutes of Health National Research Service (F31) Predoctoral Fellowship Award, 2012 McGowan Institute for Regenerative Medicine Annual Retreat Poster Award, and 2012 McGowan Institute for Regenerative Medicine Grant of the Month. Beyond work at the laboratory bench, Mr. Sicari is personable and engaging. He serves as the nucleus for the students in the laboratory and fosters professional camaraderie that results in trust and support.
The Outstanding Student Award will be presented to a student (undergraduate/graduate) who has shown outstanding research accomplishments within the tissue engineering and regenerative medicine field. The student will be selected based on a non-published manuscript submitted with the nomination and must be a member of TERMIS-Americas. The award manuscript will be published in the journal, Tissue Engineering, Part A with appropriate recognition. The manuscript must be original work; not a review.
TERMIS-AM Lifetime Achievement Award – Alan J. Russell, PhD
Alan J. Russell (PhD in Biological Chemistry, Imperial College of Science and Technology, University of London) is the founding director of the McGowan Institute for Regenerative Medicine. Most recently, Dr. Russell was appointed to the position of Highmark Distinguished Career Professor at Carnegie Mellon University. In addition, Dr. Russell is the executive director of the Pittsburgh Tissue Engineering Initiative, Inc. He has founded three biotechnology companies: ICX Agentase LLC, NanoSembly LLC, and O2Cyte LLC, and was also the founding president of TERMIS.
The Lifetime Achievement Award has been established to recognize an individual who has contributed immensely to the tissue engineering and regenerative medicine field. The award will be presented to an individual whose work has impacted and assisted with laying the foundation stones for the field.
TERMIS-AM Young Investigator Award – Karen L. Christman
The Young Investigator Award is presented to an individual who has demonstrated outstanding achievements within the tissue engineering and regenerative medicine field. The individual is selected based on a publication or a collection of manuscripts focused within a particular area of research. Candidates for the award must be within 10 years of receipt of their terminal degree and must not be tenured at the time of the nomination (if working within academia).
TERMIS-AM Innovation/Commercialization Award – Andrew J. Marshall
The Innovation/Commercialization Award recognizes the application of tissue engineering and regenerative medicine in the production of a product or technology that ultimately will benefit patients. This award recognizes either an individual or a team. The award is presented for an existing product or for a newly developed product that has been launched in the last 5 years, or for a technology launched in the last 5 years that can facilitate commercialization of a product.
McGowan Institute for Regenerative Medicine Faculty Member Received 2012 IEEE Medal for Innovations in Healthcare Technology
 McGowan Institute for Regenerative Medicine faculty member Savio L-Y. Woo, PhD, DSc, DEng, a world-leading bioengineer whose pioneering biomechanics research has profoundly impacted sports medicine and the management of ligament and tendon injuries leading to improved patient recovery, was honored by the Institute of Electrical and Electronics Engineers (IEEE) with the 2012 IEEE Medal for Innovations in Healthcare Technology. IEEE is the world’s largest technical professional association.
McGowan Institute for Regenerative Medicine faculty member Savio L-Y. Woo, PhD, DSc, DEng, a world-leading bioengineer whose pioneering biomechanics research has profoundly impacted sports medicine and the management of ligament and tendon injuries leading to improved patient recovery, was honored by the Institute of Electrical and Electronics Engineers (IEEE) with the 2012 IEEE Medal for Innovations in Healthcare Technology. IEEE is the world’s largest technical professional association.
The medal, sponsored by the IEEE Engineering in Medicine and Biology Society, recognizes Dr. Woo for pivotal contributions to biomechanics and its application to orthopedic surgery and sports medicine. The award was presented at the IEEE Honors Ceremony in Boston, Massachusetts.
Dr. Woo is a pioneer in biomechanics known around the world for his over 40 years of translational research concerning the healing and repair of tissues. Biomechanics involves applying the engineering principles of mechanics to study body movement and the effects of forces acting on the musculoskeletal system. From research at the cellular and tissue level to developing computer and robotic models of joints, Dr. Woo, together with his 600 students, post-doctoral research fellows, and junior colleagues, has provided key insight to understanding the function of bone and connective tissue. His contributions have spurred advancements in the surgical treatment and rehabilitation of ligament and tendon injuries, ultimately leading sports medicine and orthopedic surgery into the 21st Century.
Dr. Woo helped develop the “controlled motion is good” concept, showing the benefits of joint movement and early weight-bearing activities during rehabilitation compared to immobilization following surgery. His approach to robotic testing of knee and shoulder movement helped define the beneficial effects of motion on healing. He applied computer modeling and robotic technology to study joint mechanics and the effects of injury on joint function. Dr. Woo used robots to produce motions that occur during everyday activities and to determine the forces that the motions generate in the ligaments of the joints. Dr. Woo’s work has resulted in much faster recovery time for patients with soft-tissue injuries. His methods also assist with surgical planning and can predict the success of surgical techniques.
Dr. Woo has also focused on using novel functional tissue engineering to heal and regenerate ligaments and tendons at the cellular, tissue, and organ levels using bioscaffolds. Bioscaffolds are small intestinal submucosa from genetically altered pigs that can be used to heal the anterior cruciate ligament of the knee. Dr. Woo is also leading the efforts of applying degradable metallic materials to regenerate ligaments and tendons severely damaged during sports injuries.
Dr. Woo began his career at the University of California, San Diego, as a professor of surgery and bioengineering in 1970 and moved to the University of Pittsburgh in 1990. In Pittsburgh, he founded the Musculoskeletal Research Center (MSRC) within the Departments of Orthopaedic Surgery and Bioengineering. The MSRC is a multidisciplinary education center where countless orthopedic surgeons, biologists, and bioengineering students have studied and worked.
Dr. Woo is a member of the U.S. Institute of Medicine, the U.S. National Academy of Engineering, and the Republic of China’s Academia Sinica. His honors include the H.R. Lissner Medal from the American Society of Mechanical Engineers (1991), the Muybridge Medal from the International Society of Biomechanics (1995), and the Olympic Prize for Sports Science from the International Olympic Committee (1998). Dr. Woo received his bachelor’s degree in mechanical engineering from California State University, Chico, and his master’s degree in mechanical engineering and doctorate in bioengineering from the University of Washington, Seattle. Dr. Woo is currently a Distinguished University Professor and Director of the Musculoskeletal Research Center within the University of Pittsburgh’s Department of Bioengineering and Swanson School of Engineering, Pittsburgh, Pennsylvania.
McGowan Institute for Regenerative Medicine Affiliated Faculty Member Named Fellow of Prestigious Biomedical Engineering Society
McGowan Institute for Regenerative Medicine affiliated faculty member Philip LeDuc, PhD, professor of mechanical engineering at Carnegie Mellon University (CMU), has been named a fellow of the Biomedical Engineering Society (BMES) for his exceptional achievements and experience in the field, including cell and molecular biomechanics. Dr. LeDuc, one of nine fellows named this year, was honored during the BMES annual meeting in Atlanta.
“This is a wonderful honor for me to be recognized by my peers as I work to improve the lives of people worldwide and to excel in biomedical engineering research,” said Dr. LeDuc, who at CMU holds courtesy appointments in the Biomedical Engineering, Biological Sciences, and Computational Biology departments.
Dr. LeDuc is studying how a protein’s shape and form determine how it functions in the human body from a mechanics perspective. Misguided proteins, for example, have been linked to disease such as cancer and arthritis, and problems in wound healing.
Dr. LeDuc has won numerous awards, including a National Science Foundation Career Award, the Grand Challenges Exploration Award from the Bill & Melinda Gates Foundation, and the Beckman Foundation Young Investigators Award. He is a faculty member of the prestigious Sloan Foundation Minority Ph.D. Program.
With more than 5,000 members worldwide, the BMES is the leading society of professionals devoted to developing and using engineering and technology to advance human health and well-being.
“We applaud Phil LeDuc’s latest accolade and his innovative work, which spans the gamut from exploring nutrition for the healthy growth of infants and children in underdeveloped countries to novel molecular biomechanics research,” said Yu-Li Wang, head of CMU’s Biomedical Engineering Department.
Richard E. Waugh, BMES president and a biomedical engineering professor at the University of Rochester, praised Dr. LeDuc for his great energy and enthusiasm.
“Phil has been a terrific asset to BMES. He played a key role in the development of our strategic plan and was instrumental in bringing that process to a productive close. He is also spearheading an international group to explore ways in which BMES can extend its reputation and influence outside the U.S.,” Dr. Waugh said.
McGowan Institute for Regenerative Medicine Affiliated Faculty Member Selected to Attend the Frontiers of Engineering Education Symposium
 McGowan Institute for Regenerative Medicine affiliated faculty member and University of Pittsburgh Chemical and Petroleum Engineering Department assistant professor Ipsita Banerjee, PhD, has been selected to attend the Frontiers of Engineering Education (FOEE) Symposium to be held October 14-17, 2012, at the National Academies’ Beckman Center in Irvine, California. FOEE faculty members attending are actively teaching in U.S. engineering programs and have recently implemented significant innovations in their classes. Attendees participate in discussions and workshops focused on applying pedagogies to student learning of engineering fundamentals, as well as personal, interpersonal, and professional skills.
McGowan Institute for Regenerative Medicine affiliated faculty member and University of Pittsburgh Chemical and Petroleum Engineering Department assistant professor Ipsita Banerjee, PhD, has been selected to attend the Frontiers of Engineering Education (FOEE) Symposium to be held October 14-17, 2012, at the National Academies’ Beckman Center in Irvine, California. FOEE faculty members attending are actively teaching in U.S. engineering programs and have recently implemented significant innovations in their classes. Attendees participate in discussions and workshops focused on applying pedagogies to student learning of engineering fundamentals, as well as personal, interpersonal, and professional skills.
The Symposium brings together some of the nation’s most engaged and innovative engineering educators in order to recognize, reward, and promote effective, substantive, and inspirational engineering education through a sustained dialogue within the emerging generation of innovative faculty.
Attendees have multiple opportunities to share their innovations with others, learn from best practice, and network with colleagues. Attendees present new approaches they are taking to classroom, laboratory, project, experiential, computer-based, or other modes of preparing engineering students to work and lead in the 21st century. They discuss the current and evolving contexts that should shape engineering and engineering education. The program will assist them in forming an on-going, self-supporting cohort committed to innovation and sustained excellence in engineering education.
FOEE seeks to make 21st century engineering education exciting, creative, rigorous, adventurous, demanding, and empowering by:
- Presenting and propagating innovative and effective learning strategies that prepare future engineers to face 21st century challenges and opportunities.
- Raising the professional prominence of faculty who are selected fort the FOEE.
- Enhancing opportunities for professional collaboration and mutual support among FOEE faculty.
- Stimulating awareness of and interest in FOEE findings and events across all U.S. engineering schools.
- Documenting, over the long-term, enhanced student knowledge, skills, abilities, and attitudes as a result of local and widespread implementation of FOEE innovations.
- Enhancing, over the long-term, the recruitment, retention, and professional success of engineering students by virtue of local and widespread implementations of FOEE innovations.
McGowan Institute for Regenerative Medicine Affiliated Faculty Member Named Chair of Department of Neurobiology at Pitt School of Medicine
 Since October 1, 2012, the Department of Neurobiology at the University of Pittsburgh School of Medicine has a new leader: Peter Strick, PhD, Distinguished Professor of Neurobiology and McGowan Institute for Regenerative Medicine affiliated faculty member, succeeded as former department chair Susan Amara, PhD, became scientific director of the National Institute of Mental Health (NIMH), part of the National Institutes of Health.
Since October 1, 2012, the Department of Neurobiology at the University of Pittsburgh School of Medicine has a new leader: Peter Strick, PhD, Distinguished Professor of Neurobiology and McGowan Institute for Regenerative Medicine affiliated faculty member, succeeded as former department chair Susan Amara, PhD, became scientific director of the National Institute of Mental Health (NIMH), part of the National Institutes of Health.
“While it will be hard to say goodbye to Dr. Amara, but I am delighted to have a scientist of Dr. Strick’s caliber ready to take over the leadership of the Department of Neurobiology,” said Arthur S. Levine, M.D., senior vice chancellor for the health sciences and dean, School of Medicine. “He is a leading expert – if not the leading expert – on the neural basis of movement and cognition. He pioneered the use of viruses to track circuits in the central nervous system, and it is this body of work especially that led to his recent election to the U.S. National Academy of Sciences.”
Dr. Strick also holds the Endowed Chair of Systems Neuroscience and is director of the Systems Neuroscience Institute and co-director of the Center for the Neural Basis of Cognition at Pitt and Carnegie Mellon University; a leader of the Center for Military Medicine Research; and a senior VA research career scientist at the VA Pittsburgh Healthcare System. He was interim chair of the neurobiology department from 2001 to 2003.
“It’s an exciting time in neuroscience at the University of Pittsburgh,” Dr. Strick said. “I am thrilled to be in a position where I can facilitate the careers of young people and enable cutting edge research.”
Dr. Strick received his doctorate in anatomy and his undergraduate degree in biology from the University of Pennsylvania. Before joining Pitt in 2000, he worked at NIMH, the VA Medical Center in Syracuse, and at the State University of New York (SUNY)-Upstate Medical Center.
 Regenerative Medicine Podcast Update
Regenerative Medicine Podcast Update
The Regenerative Medicine Podcasts remain a popular web destination. Informative and entertaining, these are the most recent interviews:
#113 –– Dr. Bruno Péault is Professor and Chair at the University of Edinburgh, Center for Vascular Science, and Professor of Orthopaedic Surgery at UCLA. Dr. Péault discusses how he uses stem cells in his research.
Visit www.regenerativemedicinetoday.com to keep abreast of the new interviews.
Publication of the Month
| Authors: | Komori J, Boone L, Deward A, Hoppo T, Lagasse E. |
| Title: | The mouse lymph node as an ectopic transplantation site for multiple tissues. |
| Summary: | Cell-based therapy has been viewed as a promising alternative to organ transplantation, but cell transplantation aimed at organ repair is not always possible. Here we show that the mouse lymph node can support the engraftment and growth of healthy cells from multiple tissues. Direct injection of hepatocytes into a single mouse lymph node generated enough ectopic liver mass to rescue the survival of mice with lethal metabolic disease. Furthermore, thymuses transplanted into single lymph nodes of athymic nude mice generated functional immune systems that were capable of rejecting allogeneic and xenogeneic grafts. Additionally, pancreatic islets injected into the lymph nodes of diabetic mice restored normal glucose control. Collectively, these results suggest the practical approach of targeting lymph nodes to restore, maintain or improve tissue and organ functions. |
| Source: | Nature Biotechnology. 2012 Sep 23. [Epub ahead of print] |
Grant of the Month
| PI | Naftali Kaminski |
| Co-PI | Michael John Becich and Stephen R Wisniewski |
| Title | Sarcoidosis and A1AT Genomics & Informatics Center |
| Summary: | Alpha-1 antitrypsin deficiency (A1 AT), an autosomal recessive genetic disease that is associated with a variable risk of COPD, and Sarcoidosis, a systemic disease characterized by the formation of granulomatous lesions especially In the lungs, liver, skin, and lymph nodes that leads to a dramatically heterogeneous set of clinical manifestations, differ in etiology and clinicl presentation but share a variable and unpredictable course. To improve disease classification, facilitate biomarker discovery and accelerate advent of novel therapy an integrative approach that combines the results of clinical studies with molecular phenotyping results is required. The Sarcoidosis and A1 AT Genomics and Informatics Center (SAGIC) will facilitate this process by addressing the following objectives for the NHLBI Genomic Research In A1 AT and Sarcoidosis (GRADS) program: 1) Coordination of Clinical Centers activities that include patient recruitment and phenotyping and biospecimen collection. 2) Performance of transcriptome and microbiome analyses of the samples obtained by the Clinical Centers, 3) Data analysis and integration, 4) Dataset preparation for deposit in the NHLBI BloLINCC repository. The objectives will be addressed through two research projects. Project 1: A1 AT microbiome – will address the hypothesis that shifts in the lung microbiome determine the extent of lung involvement In A1 AT and that they are reflected in mRNA and microRNA changes in surrogate tissues, and Project 2: Novel molecular phenotypes in Sarcoidosis – will address the hypothesis that systemic Inflammation as reflected in gene expression changes in PBMC Is indicative of disease extent, microbiome shifts and granuloma molecular networks. |
| Source: | NHLBI |
| Term | 05/01/12 – 04/30/15 |
| Amount | $2,068,387 |
Newsletter Comments or Questions: McGowan@pitt.edu
