April 2022 | VOL. 21, NO. 4| www.McGowan.pitt.edu
Drs. Kacey Marra and Eric Lagasse Recognized During Pitt’s Celebration of Innovation

Drs. Kacey Marra (top) and Eric Lagasse
The University of Pittsburgh Office of Innovation and Entrepreneurship presented its 2022 Celebration of Innovation on April 21, 2022. This event recognized the achievements of Pitt faculty and student innovators who are seeking to improve people’s lives through the commercialization of their ideas and discoveries. In addition to recognizing all faculty and students who submitted an invention disclosure, were issued a U.S. patent, or had their innovation licensed, there were seven special awards presented. The efforts of two McGowan Institute for Regenerative Medicine faculty members were recognized among these special awards.
Emerging Innovator
Kacey Marra, PhD, Professor of Plastic Surgery and Bioengineering and Vice Chair of Research for the Department of Plastic Surgery
The Emerging Innovator Award is presented to an early-to-mid career Pitt faculty member who has worked towards achieving impact for their research through commercial translation, and a dedication to mentoring the next generation of innovators and entrepreneurs.
Dr. Marra is a pioneering innovator around the problem of repairing large gap nerve injuries. She was inspired in this work after receiving funding from the Department of Defense for her research and meeting soldiers who had suffered significant nerve damage from combat wounds. Dr. Marra and her lab have demonstrated in animal studies the ability to restore up to 80 percent of nerve function in large-gap injuries through the application of a biodegradable tube containing a time-released protein growth factor.
During her time at the Pitt, she has submitted 23 invention disclosures to the Innovation Institute, has been issued six patents on her discoveries, and has had her innovations licensed six times, including the formation of three startup companies. This puts her in the top handful of women innovators in the university’s history.
Dr. Marra launched her own company, Nerve Repair Technologies, in 2018. She is presently raising a seed round and preparing to hire a CEO to help the company take its next steps toward an application to the Food and Drug Administration to conduct clinical trials.
Startup of the Year
Co-Founder: Eric Lagasse, PharmD, PhD, Associate Professor in the Department of Pathology, University of Pittsburgh, with a secondary appointment in Pitt’s Clinical and Translational Institute
For over a decade, Dr. Lagasse has been researching the regeneration of organ tissue as part of the McGowan Institute of Regenerative Medicine. This kind of work at the McGowan Institute represents the progression of discovery from the pioneering organ transplantation work of the late Thomas Starzl, MD, PhD.
Dr. Lagasse discovered that hepatocyte cells of the liver, when introduced into the lymph nodes, will grow functional liver tissue. In effect, the lymph nodes become bioreactors inside the body. This opens up the possibility of extending the lives of people suffering end stage liver disease, bridging them to a potential transplant, or even eliminating the need for a transplant.
Dr. Lagasse engaged with Michael Hufford, PhD, an entrepreneur in residence at the Pitt Innovation Institute, to begin exploring the commercial potential of his discovery. Dr. Hufford, a veteran life sciences executive and two-time Pitt alumnus, who had several successful startup exits under his belt, was impressed with the solid pre-clinical work that Dr. Lagasse and his co-founder McGowan Institute affiliated faculty member Paulo Fontes, MD, had done to lay a strong preliminary regulatory framework.
LyGenesis raised investment rounds totaling $7 million as it performed additional pre-clinical work recommended by the FDA. At the end of 2020, the company raised another $11 million investment round and announced it had been cleared to conduct a Phase 2a study of the safety, tolerability, and efficacy of its first-in-class novel cell therapy for patients with end-stage liver disease.
LyGenesis is currently also in earlier stages of development of similar cell therapies for the pancreas, kidneys, and thymus, and most recently it has also begun a program for the treatment of inborn errors of metabolism in infants that has demonstrated positive in vitro results.
The company last spring moved into offices in the city’s Squirrel Hill neighborhood and is poised for incredible growth, right here in Pittsburgh.
“Universities are places where new knowledge is developed and disseminated. At Pitt, our faculty and students are passionate about translating new knowledge into new products and services and companies that make an impact on the world and our regional economy,” said Evan Facher, PhD, MBA, Vice Chancellor for Innovation and Entrepreneurship.
Dr. Facher added, “The Celebration of Innovation is our opportunity to congratulate those Pitt innovators who have achieved significant milestones on the path to commercialization and to recognize those who are helping them on their journeys.”
Congratulations, all!
RESOURCES AT THE MCGOWAN INSTITUTE
May Histology Special – Verhoeff-Van Gieson (VVG) Staining
Elastin is a protein in connective tissue that allows many tissues in the body to resume their shape after stretching or contracting. Elastin helps skin to return to its original position when it is poked or pinched.
Elastin serves an important function in arteries as a medium for pressure wave propagation to help blood flow and is particularly abundant in large elastic blood vessels such as the aorta. Elastin is also very important in the lungs, elastic ligaments, the skin, and the bladder, elastic cartilage. It is present in all vertebrates above the jawless fish
Verhoeff’s stain forms a variety of cationic, anionic and non-ionic bonds with elastin, the main constituent of elastic fiber tissue. Elastin has a strong affinity for the iron-hematoxylin complex formed by the reagents in the stain and will hence retain dye longer than other tissue elements, which allows elastin to remain stained while other tissue elements are decolorized. Elastic fibers and cell nuclei are stained black, collagen fibers are stained red, and other tissue elements including cytoplasm are stained yellow.
You’ll receive 25% off VVG Staining in May when you mention this ad.
Contact Julia at the McGowan Core Histology Lab by email: Hartj5@upmc.edu or call 412-624-5265.
Sample Submission Procedures: In response to COVID-19, we ask that you contact us to schedule a drop off time. When you arrive at the building you can call our laboratory at (412) 624-5365. Someone will meet you in the lobby to collect your samples. When your samples are completed, you will receive an email to schedule a pickup time.
SCIENTIFIC ADVANCES
Bone Super Glue Travels to Space

Since 2014, the International Space Station (ISS) National Laboratory has partnered with Boeing to award grants through an annual MassChallenge “Technology in Space Prize,” which supports startups with ideas for innovative research and development onboard the ISS National Laboratory. Boeing reported in SpaceNews about RevBio, a Lowell, Mass.-based medical device company that makes a patented bone adhesive called Tetranite, which fixes fractures and accelerates bone repair—especially in patients suffering from osteoporosis. After learning that even a short stay in microgravity can cause loss of bone mass, the company entered the “Technology in Space” competition and was awarded the prize in 2015.
RevBio’s first ISS mission was an in vitro experiment focused on the response of osteoblasts (a bone subtype) to Tetranite. RevBio is now preparing for its second mission in October 2022, an in vivo experiment using Tetranite to treat 40 mice with artificially created bone defects while concurrently experimenting on 40 control mice on Earth to study the differences in microgravity. The experiment will examine the biomaterial’s osteoconductivity when used in a microgravity environment where bone density and the ability to regenerate new bone tissue is significantly compromised.
The general theory, said principal investigator and University of Pittsburgh School of Dental Medicine associate professor of periodontics and preventive dentistry Giuseppe Intini, DDS, PhD, who studies the biology of bone, is that microgravity impairs bone tissue regeneration by inhibiting differentiation of skeletal stem cells, and that Tetranite can reverse this inhibition and promote bone regeneration.
“We’re going to investigate how Tetranite—a bone super glue that’s also injectable as a bone filler—behaves in space,” said Dr. Intini, an affiliated faculty member of the McGowan Institute for Regenerative Medicine. “The hypothesis is that the Tetranite will be able to stimulate the skeletal stem cells and promote bone formation and regeneration. Many people suffer from osteoporosis, and this could be extremely useful to treat those fractures on Earth.”
The company is initially developing this technology for use in the dental market and recently initiated two clinical studies for the use of Tetranite to immediately stabilize dental implants placed in sites that lack sufficient primary stability. The company is also working to develop adhesive applications for the broader orthopedics market.
Podcast: In-Space Production of Biomanufacturing with Ms. Christine Kretz and Dr. William Wagner

Ms. Christine Kretz (top) and Dr. William Wagner
The International Space Station (ISS) U.S. National Laboratory and the McGowan Institute for Regenerative Medicine (MIRM) are working on a multi-year research alliance to push the limits of biomedical research and development aboard the orbiting laboratory. The alliance — a core element of the ISS National Laboratory Industrial Biomedicine Program — serves as a benchmark for how the ISS National Laboratory develops similar programs in the future involving research and development activities aboard the space station.
The ISS National Laboratory and MIRM are collaborating with partners from industry, other academic research centers, and government agencies to drive the progress of regenerative medicine research onboard the ISS. The program focuses on microgravity life sciences research and development, with a line of sight toward products and services for clinical application on Earth. For instance, exploiting the unique behavior of stem cells in microgravity could improve cell-based therapies for a variety of diseases and impairments, such as traumatic brain injury and type I diabetes. Similarly, microgravity could allow 3D printers to create complex tissue structures that are difficult to achieve in the presence of full gravity.
Christine Kretz, Vice President of Programs and Partnerships for the ISS National Laboratory, Center for the Advancement of Science in Space, and William Wagner, PhD, Director of the McGowan Institute as well as Distinguished Professor of Surgery, Chemical Engineering, and Bioengineering at the University of Pittsburgh, recently spoke with Regenerative Medicine Today host John Murphy, McGowan Institute Executive Director, about
- Expanded capabilities on the International Space Station (ISS) regarding biotechnology
- How space station access and current costs associated with low earth orbit (LEO) research is supporting LEO biomanufacturing today
- Current biotechnology areas well suited to LEO biomanufacturing include stem cell behaviors, microphysical systems (organ-on-a-chip), biomaterials self-assembly, 3D printing
- Astronaut-driven vs future automated research processes
- Quality management systems on ISS and their impact on FDA approvals for product developments
Listen to their conversation here.
Illustration: International Space Station National Laboratory (Kretz)/McGowan Institute for Regenerative Medicine (Wagner)
Hepato-Healing: A Treatment from Within
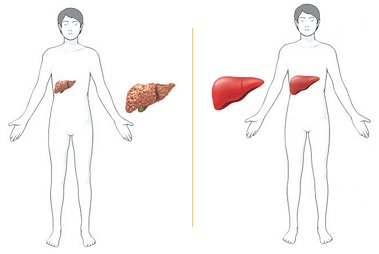
The same technology used in COVID-19 vaccines holds promise as a new approach to liver disease treatment. Michele Dula Baum, Senior Writer/Project Manager, Strategic Communications, Office of the Senior Vice Chancellor, Health Sciences, reports in PittMed where the science is heading in this new therapy methodology.
Growing up in Guadalajara, Mexico, Alejandro Soto-Gutiérrez, MD, PhD, associate professor in the Department of Pathology at the University of Pittsburgh, an affiliated faculty member of the McGowan Institute for Regenerative Medicine, Children’s Hospital of Pittsburgh of UPMC, and the Thomas E. Starzl Transplantation Institute, felt helpless when a beloved uncle died of liver disease. Since then, the physician-scientist has dedicated his career to changing the odds for people facing the condition’s devastating end stage, for which the only cure is a transplant.
Now, a messenger RNA-based therapy—the same technology that Pfizer and Moderna used for their vaccines for COVID-19—holds promise for liver disease treatment. This tool, says Dr. Soto-Gutiérrez, is “changing the way we’re going to treat patients in the coming years.
“I feel excited every morning.”
His studies of how liver cells function—and why they sometimes fail—have taken Dr. Soto-Gutiérrez around the world. Before joining Pitt in 2009, he earned his medical degree in Mexico and completed a PhD and surgical fellowship in Japan, as well as a surgery research fellowship at Massachusetts General Hospital.
Fortunately, Pitt was home to a scientist Dr. Soto-Gutiérrez had been chasing a chance to work with for a decade: McGowan Institute faculty member and professor of surgery Ira Fox, MD, a Pitt-trained transplant surgeon who had returned to Pittsburgh from the University of Nebraska in 2008. Dr. Fox leads the Center for Innovative Pediatric Regenerative Therapies at UPMC Children’s Hospital of Pittsburgh.
At the joint Pitt-UPMC Pittsburgh Liver Research Center and McGowan Institute for Regenerative Medicine, Drs. Fox and Soto-Gutiérrez teamed up to explore the possibility of “reprogramming” failing liver cells to do a 180—in essence, to heal themselves.
In the United States, more than 12,000 people are waiting for a liver, yet only about 8,000 livers from deceased donors become available each year. About two-thirds of all living-donor liver transplant procedures in the country take place at UPMC (the system celebrated its 500th such procedure last year); still, demand for donor organs outpaces supply. The need to find alternatives is acute.
For years, Drs. Fox and Soto-Gutiérrez systematically studied livers scarred and impaired by alcohol-induced cirrhosis or nonalcoholic fatty liver disease, looking for genetic and cellular mechanisms of action that contribute to end-stage liver disease.
They identified a key player called hepatocyte nuclear factor 4-alpha (HNF4a), a transcription factor (protein) that serves as a main control panel to regulate much of the gene expression in liver cells. Drs. Fox and Soto-Gutiérrez found that cirrhotic rats with nonfunctioning HNF4a experienced liver failure.
When the team altered gene activity to re-express the protein—an approach they call transcriptional therapy—liver function came back online. They then analyzed diseased liver tissue from transplant patients and confirmed the finding in humans, as well. HNF4a, it seemed, could be key to reversing liver disease.
In a recent Hepatology Communications paper, the team described the results of taking cultured cells from failing human livers and treating them with mRNA, an approach that delivered instructions to the cells to turn HNF4a back on. The treatment worked, and fast. Function returned in about 24 hours.
“What we’re talking about, essentially, is a new approach . . . that might bring patients back from terminal liver failure,” says Dr. Soto-Gutiérrez, explaining that, if successful, the technology could end the need for surgical transplant and lifelong immunosuppression.
“The development of these kinds of technologies in the area of personalized medicine will really make clinical trials and the development of new drugs more effective,” he adds. “The change that a patient will see will be dramatic.”
Drs. Fox, Soto-Gutiérrez, and their associate, Alina Ostrowska, PhD, a liver-cell processing expert, have established the start-up Pittsburgh ReLiver to work with investors and pharmaceutical companies to translate these findings into clinical trials. If all goes well, Dr. Soto-Gutiérrez says, clinical trials could begin within two years.
Illustration: Alejandro Soto-Gutiérrez.
Cardiomyopathy Leads to Heart Transplantation for Sharpsville Man

Cardiomyopathy is a disease that affects the heart muscle. In cardiomyopathy, the damaged heart muscle becomes enlarged, thick, or rigid, and cannot pump blood effectively. It’s the leading cause of heart failure and the most common reason for needing a heart transplant. Unlike other heart problems, cardiomyopathy frequently affects younger people.
McGowan Institute for Regenerative Medicine affiliated faculty member David Kaczorowski, MD, is the Surgical Director of the Advanced Heart Failure Center of the University of Pittsburgh Medical Center Heart and Vascular Institute and an Associate Professor of Cardiothoracic Surgery, Department of Cardiothoracic Surgery, University of Pittsburgh. Dr. Kaczorowski’s clinical work is devoted mainly to treating patients with advanced heart failure and cardiogenic shock through the use of temporary mechanical support modalities such as extracorporeal membrane oxygenation (ECMO) and durable strategies, including left ventricular assist devices (LVAD) and cardiac transplantation.
As Sharpsville, PA, native Thomas Findley, now 38 years old, recently completed his physical therapy, he found his life taking one more step toward returning to normal — not just because of his heart transplant several months ago, but after years of dealing with heart-related illnesses. His story of recovery was reported by David Dye, a staff writer for The Herald.
In mid-October 2021, Mr. Findley was taken to UPMC Presbyterian in Pittsburgh and was there until mid-November, when he was placed on the list for a heart transplant. About a week before his transplant, he was given an artificial pump due to his failing heart. He was later placed on the transplant list on Nov. 10 and received a heart on Nov. 12.
Dr. Kaczorowski, who was involved in caring for Mr. Findley during his stay at the hospital, said his patient suffered from cardiomyopathy. At the point Mr. Findley received his heart, Dr. Kaczorowski said it was difficult to predict how much longer he could have survived without the heart transplant.
“I would say he was becoming fairly ill,” Dr. Kaczorowski said. “I think we were worried that his liver was becoming dysfunctional, and his kidney function was getting worse.” Dr. Kaczorowski said the transplant itself went “smoothly,” while Mr. Findley was able to leave the hospital on the 12th day after surgery, instead of the usual several weeks it has taken other patients.
Dr. Kaczorowski said there are different steps people can take to maintain a healthy heart, such as regular visits to a physician, exercise, and avoiding activities such as smoking.
Collaboration Shapes Extracellular Vesicle Retention Strategy

Drs. Phil Campbell (top) and Xi (Charlie) Ren
Leveraging unique expertise in the spirit of collaboration is one of Carnegie Mellon University’s (CMU’s) formulas for success. Over the past three years, two CMU and McGowan Institute for Regenerative Medicine affiliated faculty members, Phil Campbell, PhD, and Xi (Charlie) Ren, PhD, partnered on research related to spatial control of extracellular vesicles (EVs). Their efforts have yielded a novel strategy that enables long-term EV spatial retention, a key variable to enable future tissue engineering and regenerative medicine applications.
“EVs can be thought of as the universal communicators—not only in the body, but in all living things,” explained Dr. Campbell, research professor of biomedical engineering at CMU. “They occur naturally, are biocompatible, and can be used to deliver messages between cells at a nanoscale level.”
Studies have shown that EV-based therapeutics are less likely to trigger adverse immune responses and do not pose the same logistical and regulatory concerns as therapies based on living cells. However, while rich in potential, EVs delivered in their natural form are usually prone to rapid clearance and typically lack controlled targeted delivery. In some applications, repeated extended dosing is required, which presents challenges with both overall effectiveness and efficacy.
“Our work stemmed from a very simple question,” said Dr. Ren, assistant professor of biomedical engineering at CMU. “There are many aspects of controlling the biologic function of EVs, but if we can provide retention (of EVs), can we do something great? One of the best ways to advance research is to talk to other faculty who have different expertise. We have taken the chemistry aspects and tools from my lab and coupled them with the EV platform developed by Phil’s lab to present innovative new technology.”
In recent research published in Biomaterials, the group described a method for immobilizing mesenchymal stem cell (MSC)-derived EVs in collagen hydrogels to augment angiogenesis, or the formation of new blood vessels, which is a critical step for most reparative and regenerative applications. Practically speaking, diabetic patients who suffer from vascular disease, where arteries are hardened throughout their bodies, could benefit from pro-angiogenic biomaterials like these.
To accomplish this, the researchers incorporated a selective chemical tag onto EV’s exterior surface, which does not affect its morphological or functional properties. Through this chemical tag, EVs can be effectively rooted within hydrogel implants and elicit more robust host cell infiltration. In the study, angiogenic and immunoregulatory responses compared to 10 times the higher dose required by using conventional, non-immobilized EVs.
“In a nutshell, what we now have the ability to do is to spatially control where we put EVs and keep them there under controlled conditions,” summarized Dr. Campbell. “We specifically looked at promoting angiogenesis for this paper, but broader than that, this technique could lead to increased therapeutic applications for wound healing and other regenerative and reparative therapies.”
Efforts to apply this EV platform to bone tissue engineering as an alternative to titanium is also under way. The group is designing scaffolding and exploring functionalization to overcome the current challenges titanium presents as a limited regenerative when implanted within the body.
“We hope that we can design a way to infuse non-biological materials, such as a metal implant, with this hydrogel, with EV loaded, that could encourage the body to take the foreign implant amongst the body parts,” said Dr. Ren.
New Hope for Predicting and Treating Heart Failure in Babies Born with Deadly Heart Defect

Almost one third of babies born with hypoplastic left heart syndrome, or HLHS, die from heart failure before their first birthday. By uncovering cellular processes that drive heart failure in these young patients, a new study may hold the answers to identifying and treating those at highest risk of early death.
Published today in Cell Stem Cell by scientists at the University of Pittsburgh School of Medicine, the study found that two commonly used medications, Viagra and an over-the-counter drug called tauroursodeoxycholic acid (TUDCA), restored processes that drive heart failure in cells derived from patients, opening potential avenues for new HLHS treatments. McGowan Institute for Regenerative Medicine affiliated faculty member Sruti Shiva, PhD, Professor of Pharmacology and Chemical Biology, a Principal Investigator in the Vascular Medicine Institute, and Director of the University of Pittsburgh Center for Metabolism and Mitochondrial Medicine (C3M), is a co-author of the study.
“HLHS is one of the most lethal types of congenital heart disease,” said Cecilia Lo, PhD, chair of the Department of Developmental Biology at Pitt, and senior author of the study. “What causes heart failure in HLHS patients who die before one year of age is unknown, and the only treatment option is a heart transplant, which often is not possible. If we can find the cause, then there is hope for therapy.”
To get to the heart of severe HLHS causes, Dr. Lo and Xinxiu (Cindy) Xu, PhD, first author of the study and a postdoctoral researcher in Dr. Lo’s lab, collected skin cells from three healthy people and 10 HLHS patients, who had either milder disease, surviving past age five without a transplant, or severe HLHS, meaning they died or required a heart transplant in their first year of life.
First, Dr. Xu reprogrammed patient skin cells into so-called induced pluripotent stem cells, which can become any type of cell. Next, she added a mixture of growth factors and nutrients that prompt the stem cells to develop into heart cells.
By observing the heart cells under a microscope, the researchers noticed clear differences among cells from different patient groups. Just as a living heart squeezes and releases to pump blood, so too do heart cells in a dish, even without blood to circulate. Cells from patients with milder HLHS looked and behaved similarly to those from healthy people, pulsing quickly and steadily. In contrast, cells from the severe group throbbed in a more languid manner, eerily reminiscent of what doctors see in the hearts of many living HLHS patients.
Looking more closely, the researchers found that heart cells from both patient groups had defective mitochondria, the energy-generating centers, but this dysfunction was worse in the severe group. Heart cells from severe HLHS patients also failed to rally natural defenses against stress caused by the mitochondrial defect.
Having identified what was wrong at a cellular level, the researchers now had targets for therapies. They found that sildenafil, commonly known as Viagra, and TUDCA rescued the mitochondrial defect in heart cells from patients with severe disease.
Dr. Xu explained that if the heart is a car, the mitochondrion is like an engine. She thinks of sildenafil as engine oil, “lubricating” mitochondrial function, preventing it from overheating. TUDCA, on the other hand, is like coolant, also protecting the engine from overheating. Either of these drugs can reduce stress caused by defective mitochondria, helping heart cells from severe patients to achieve what patients with milder HLHS accomplish naturally.
“We chose these drugs because we know they are safe and already approved in the clinic for other conditions,” added Dr. Lo. “This means it will take less time to get treatments to patients compared with developing entirely new drugs.”
For HLHS patients, this could mean new therapies — that don’t rely on heart transplant — are on the horizon to treat heart failure.
Dr. Lo said that the study opens possibilities for using stem cell-derived heart cells to model heart failure in a dish and screen drugs faster than traditional animal model approaches.
The findings could also lead to development of tests that prioritize patients for early heart transplant.
“Because the defect is on a cellular level, a simple blood test might be able to detect defective mitochondria, allowing early identification of patients most vulnerable to heart failure,” said Dr. Lo. “Ultimately, these findings will help achieve our goal of improving the clinical care and quality of life for patients with the congenital heart disease such as HLHS.”
R01 Grant Received for Research of Cholestatic Liver Diseases

Liver transplantation is the only effective treatment for patients with end-stage liver diseases; however, the shortage of donor livers makes this therapy very limited. Comprehensive and detailed understanding of innate liver regeneration in diseased livers will provide significant insights into developing therapeutic strategies to promote liver regeneration in the patients. Our studies will delineate the molecular mechanisms underlying hepatocyte-to-cholangiocyte reprogramming-mediated biliary regeneration and suggest HDAC1/2 inhibitors as promising therapeutic drugs to promote the reprogramming-mediated regeneration in patients with cholestatic liver diseases.
In support of these studies, McGowan Institute for Regenerative Medicine affiliated faculty member Donghun Shin, PhD, Associate Professor, Department of Developmental Biology, University of Pittsburgh School of Medicine, is the principal investigator on a National Institute of Diabetes and Digestive and Kidney Diseases R01 grant which will run for 4 years beginning April 1, 2022.
The abstract reads:
Biliary epithelial cells (BECs; also called as cholangiocytes) that line the hepatic biliary tree control bile composition and flow. Injury to the BECs leads to cholestasis, which can progress to fibrosis, cirrhosis, and liver failure. Cholestatic liver diseases are associated with high morbidity and mortality; however, few effective therapies are available. In fact, liver transplantation is the only life-extending treatment for end-stage cholestatic liver diseases, but the shortage of donor livers makes this therapy extremely limited. In the injured liver with biliary damage, hepatocytes (HCs) can contribute to BECs to recover from the loss of BECs. Recent studies in mice have shown that HC-derived BECs contribute to the intrahepatic bile ducts, thereby restoring appropriate bile flow. Patients with biliary obstruction or cholangiopathies also exhibit biliary marker expression in HCs, suggesting their reprogramming into BECs. Thus, augmenting innate HC-to-BEC reprogramming in cholestatic liver diseases is an attractive therapeutic alternative to ameliorate cholestasis and subsequent cirrhosis. To develop such a therapy, it is crucial to better understand the molecular mechanisms underlying HC-to-BEC reprogramming. Furthermore, identifying small molecules that can augment the reprogramming should provide promising therapeutic drugs for patients with cholestatic liver diseases. Our long-term goal is to completely delineate the molecular mechanisms underlying HC-to-BEC reprogramming. As a first step in pursuit of that goal, the objective of this proposal is to determine the cellular and molecular characteristics of HC-to-BEC reprogramming-driven biliary regeneration in our two innovative zebrafish models and to elucidate how histone deacetylase 1 (hdac1) regulates HC-to-BEC reprogramming. Based on our preliminary data obtained from pharmacological and genetic studies, we hypothesize that Hdac1 inhibition promotes HC-to-BEC reprogramming by derepressing the Notch receptor gene notch2 and the signal transducer and activator of transcription 3 gene (stat3). We will test this hypothesis and accomplish the objective of this application by (1) elucidating the entire process of HC-to-BEC reprogramming-driven biliary regeneration in the two zebrafish models, in which complete absence of BECs is achieved and subsequently HCs convert to BECs, (2) determining the effects of Hdac1 inhibition on HC-to-BEC reprogramming in both zebrafish and mice, and (3) elucidating the molecular mechanisms by which Hdac1 inhibition promotes the reprogramming. The successful accomplishment of the proposed research will not only provide novel molecular mechanisms underlying HC-to-BEC reprogramming but also suggest HDAC1/2 inhibitors as promising therapeutic drugs to promote the reprogramming in patients with cholestatic liver diseases.
Illumination of Immune Checkpoint LAG3 ‘Black Box’ Could Yield New Cancer and Autoimmune Therapies

Fighting cancer and chronic disease is tiring work for the immune system. When T cells are engaged in this kind of lengthy battle, they can become exhausted, or unable to function properly. One immunotherapy that revitalizes these cells by blocking an immune checkpoint protein called LAG3 was recently approved by federal regulators. But exactly how LAG3 works has remained a mystery — until now.
Published in Nature Immunology, a new study pinpoints how LAG3 modulates T cell activity, providing important insights for development of other LAG3-blocking therapies for cancer and autoimmune disorders. McGowan Institute for Regenerative Medicine affiliated faculty member Simon Watkins, PhD, the founder and director of the Center for Biologic Imaging at the University of Pittsburgh, a member of the UPMC Hillman Cancer Institute, and a distinguished professor and vice chairman within the Department of Cell Biology, is a co-author of the study.
“Although LAG3 has been studied extensively as a potential immunomodulatory target, there hadn’t been an awful lot known about how it works. It truly was a black box,” said co-senior author Dario A.A. Vignali, PhD, distinguished professor and vice chair of immunology at the University of Pittsburgh School of Medicine. “I believe this is a landmark study because we finally know how LAG3 works. This will help the development of new LAG3-targeting therapeutics.”
T cells protect the body by eliminating threats such as cancer and infection. Molecules called antigens from cancer cells and pathogens are recognized by T cell receptors, activating these immune cells for battle. But exhausted T cells don’t engage normally with antigen-presenting cells because immune checkpoint proteins slow immune responses like brakes on a car.
Research on two immune checkpoints called PD-1 and CTLA-4 led to development of breakthrough drugs called immune checkpoint inhibitors more than a decade ago. These therapies work by releasing the immune system’s brakes and revving up T cell function.
In March 2022, the U.S. Food and Drug Administration approved the first LAG3 inhibitor for advanced melanoma, and 18 other LAG3-inhibitiors are in clinical trials — despite murkiness around how this checkpoint inhibits T cell function.
To “look under the hood” of LAG3, Dr. Vignali started this research in his previous position at St. Jude Children’s Research Hospital along with first author Clifford Guy, Ph.D., who was a postdoc in his lab at the time, and co-senior author Creg Workman, Ph.D., who is now a research assistant professor of immunology in Vignali’s lab at Pitt.
When T cells engage with an antigen-presenting cell, they form a contact point called an immunological synapse that gets flooded with T cell receptors.
“We found that LAG3 binds to T cell receptors, but it doesn’t inhibit them directly,” explained Dr. Workman. “Instead, LAG3 uses T cell receptors to hitch a ride to the immunological synapse.”
Using special dye that measures acidity, the team found that accumulation of LAG3 in the synapse creates acidic conditions that disrupt association between coreceptors — CD4 on helper T cells or CD8 on killer T cells — and a signaling enzyme called Lck, thwarting a key requirement for T cell activation and signaling.
Current LAG3-blocking drugs target an interaction between LAG3 and so-called MHC Class II molecules, which were thought to be essential for LAG3 function.
But the new study found that LAG3 suppression of T cell activity occurred with or without these molecules.
“This research calls into the question the design of LAG-blocking therapeutics in development,” said Dr. Vignali, who is also associate director for scientific strategy and co-leader of the Cancer Immunology & Immunotherapy Program at UPMC Hillman Cancer Center. “They obviously work, but do they work optimally? They’ve been designed to block Class II interactions, but this study shows that LAG3 can function in absence of Class II.”
“Now that we know LAG3 binds to the T cell receptor, we might be able to generate optimal blockers that target this interaction,” he added.
Beyond cancer, Dr. Vignali said that the research could inform new treatment approaches for autoimmune and inflammatory disorders. Because these diseases are caused by overactive immune response, potential drugs could enhance — rather than block — LAG3 activity.
“To help the body fight cancer, you want to release the immune system brakes,” explained Dr. Vignali. “But for autoimmunity, you want to pump the brakes harder.”
April is Donate Life Month

McGowan Institute for Regenerative Medicine affiliated faculty member George Mazariegos, MD, pediatric liver and intestine transplant surgeon and the Director of the Hillman Center for Pediatric Transplantation at the Children’s Hospital of Pittsburgh of UPMC, spoke to Ellis Cannon of WPXI/Pittsburgh in support of April’s Donate Life Month. The video interview is a part of the station’s UPMC/WPXI Community Matters segment.
UPMC Children’s Hospital of Pittsburgh established the nation’s first pediatric transplant center in 1981 under the guidance of transplant pioneer Thomas E. Starzl, MD, PhD. The hospital has performed more pediatric transplants than any other pediatric transplant center. The pediatric transplant program achieves patient survival rates that are among the highest in the world. Children’s also is recognized as a leader in pediatric transplantation-related research.
During the interview, Dr. Mazariegos answers the following questions:
- Why would a child need a transplant?
- What are the benefits of an organ transplant?
- What is the process for getting an organ transplant?
- Why is living donation beneficial?
Watch the interview here.
Welcome: Drs. Ramakrishna Mukkamala and Hamza Yazdani

The McGowan Institute for Regenerative Medicine welcomes new affiliated faculty members Ramakrishna Mukkamala, PhD and Hamza Yazdani, MD.
Dr. Mukkamala is a Professor in the Departments of Bioengineering and Anesthesiology and Perioperative Medicine at the University of Pittsburgh. He received graduate and post-doctoral training in Bioelectrical Engineering at the Massachusetts Institute of Technology.
He has been a dedicated cardiovascular researcher throughout his career. He received an NSF CAREER Award and an AHA Scientist Development Grant for his early research on blood pressure waveform analysis and helped Retia Medical translate this research to hospital use. He received an IEEE EMBS Most Impactful Paper Award and the Michigan State University (MSU) All-University Innovation of the Year Award in 2019 for his recent research on cuff-less blood pressure measurement.
He is also a devoted educator with two teaching awards from MSU and now teaches Biological Signals and Systems at Pitt.
In addition, Dr. Mukkamala currently serves as an Associate Editor for IEEE Transactions on Biomedical Engineering and the Chair of the IEEE EMBS Technical Committee on Cardiopulmonary Systems and Physiology-Based Engineering.
 Dr. Yazdani is a Research Assistant Professor in the Department of Surgery at the University of Pittsburgh. He completed a Postdoctoral Research Fellowship in Pitt’s Department of Surgery, received a MSc, Surgery, at the 2nd Affiliated Hospital of Harbin Medical University in China, and obtained a MD also at Harbin Medical University.
Dr. Yazdani is a Research Assistant Professor in the Department of Surgery at the University of Pittsburgh. He completed a Postdoctoral Research Fellowship in Pitt’s Department of Surgery, received a MSc, Surgery, at the 2nd Affiliated Hospital of Harbin Medical University in China, and obtained a MD also at Harbin Medical University.
Dr. Yazdani’s research, clinical, and/or academic interests include liver transplant, liver ischemia-reperfusion, sterile inflammation; surgical stress in regulating tumor growth, metastasis, and the tumor microenvironment; the interaction between innate and adaptive immunity in the setting of surgical stress; exercise training in surgical stress and metastasis; and exercise in training innate and adaptive immunity.
His current grant work includes Pittsburgh Liver Research Center Pilot and Feasibility Grant (2021-2022) and the Community Liver Alliance 2022 Research Grant (2022-2023).
In 2020, he received an Excellence in Research Award at the Eighteenth Annual Richard L. Simmons Lecture in Surgical Science and Department of Surgery Research Day. He also received in 2021 the Best Research Presentation Award at the Nineteenth Annual Richard L. Simmons Lecture in Surgical Science and Department of Surgery Research Day.
Dr. Yazdani is a member of the American Association of Cancer Research (AACR) and The American Association of Immunologists (AAI).
Welcome!!
AWARDS AND RECOGNITION
Dr. William Wagner Named 2022-2023 President-Elect for the Society For Biomaterials
 The Society For Biomaterials hosted its 2022 Annual Meeting at the Baltimore Marriott Waterfront, April 27-30, 2022. The theme for the 2022 Annual Meeting was “The Perilous Fight to Translate Innovative Research to Commercial Viability.” This year’s program focused on ideas that involve breakthroughs in biomaterials science, translation of basic research to clinical application, next-generation medical devices, and content that explores standards development, regulatory issues, and commercial realities.
The Society For Biomaterials hosted its 2022 Annual Meeting at the Baltimore Marriott Waterfront, April 27-30, 2022. The theme for the 2022 Annual Meeting was “The Perilous Fight to Translate Innovative Research to Commercial Viability.” This year’s program focused on ideas that involve breakthroughs in biomaterials science, translation of basic research to clinical application, next-generation medical devices, and content that explores standards development, regulatory issues, and commercial realities.
During this year’s meeting, it was announced that McGowan Institute for Regenerative Medicine Director William Wagner, PhD, Distinguished Professor of Surgery, Chemical Engineering, and Bioengineering at the University of Pittsburgh, was named the 2022-2023 President-Elect for the Society For Biomaterials, an honor bestowed to him by his fellow members.
Dr. Wagner has been an active member of the Society For Biomaterials since 1992, having organized programming, served on committees, and acted as the Society For Biomaterials representative to the International Union of Societies for Biomaterials Science and Engineering (IUSBSE). In his directorship of the McGowan Institute, he has successfully championed and facilitated the formation of spin-out companies to complete the Institute’s mission of delivering solutions to patients suffering from tissue and organ failure. To date, 35 companies have been launched from the Institute. In recent years he has been awarded the Society For Biomaterials Founders Award and the Clemson Award for Applied Research, the Chancellor’s Distinguished Research Award by the University of Pittsburgh, the Zhu Kezhen Distinguished Lectureship from Zhejiang University, and the Senior Investigator Award by TERMIS-Americas. In 2017 he was inducted into the National Academy of Inventors and in 2018 was named Inventor of the Year by the Pittsburgh Intellectual Property Law Association. In 2019 he was appointed to the rank of Distinguished Professor of Surgery at the University of Pittsburgh, the highest recognition available to the faculty. In 2020 the 4th edition of the best-selling biomaterials textbook, “Biomaterials Science,” was published with Dr. Wagner taking over the role of lead editor in partnership with a new generation editorial team. Dr. Wagner’s research interests are in cardiovascular engineering with projects that address medical device biocompatibility and design, biomaterial development, tissue engineering, and targeted imaging.
Congratulations, Dr. Wagner!
EV2 Technologies Takes 2nd Place in 2022 Randall Family Big Idea Competition, Wins $15K Prize

EV2 Technologies’ Biocarpet is a flexible and fully biodegradable endovascular device that uses a special thermoforming technique and provides treatment of peripheral arterial disease in small arteries and across joints. The team’s approach minimizes vascular wall stress thus reducing restenosis. This technology was developed in the laboratories of the McGowan Institute for Regenerative Medicine and won the $15,000 prize at the 2022 Randall Family Big Idea Competition. Collaborators on this effort include:
- Ali Behrangzade, PhD, Research Assistant, Soft Tissue Biomechanics Laboratory, Department of Bioengineering
- Trace Maule, Student, Department of Computer Science
- Jonathan Vande Geest, PhD, Professor, Department of Bioengineering, University of Pittsburgh, and Principal Investigator of the Soft Tissue Biomechanics Laboratory
- William Wagner, PhD, Director of the McGowan Institute for Regenerative Medicine as well as Distinguished Professor of Surgery, Chemical Engineering, and Bioengineering at the University of Pittsburgh
- John Pacella, MD, Associate Professor in the School of Medicine at the University of Pittsburgh and an Interventional Cardiologist within the UPMC Heart and Vascular Institute
Since its inception in 2009, this competition has attracted hundreds of interdisciplinary teams seeking to bring their big ideas to market. The competition is offered to all Pitt undergraduate and graduate students across all schools and departments. Post-doctoral fellows are included. The Randall Family Big Idea Competition is made possible through the generous support of the Randall family. Robert P. Randall, a Pitt alumnus and trustee, was president and chief executive officer of the Three Rivers Aluminum Company (TRACO), a prominent regional door and window company, which was acquired by Alcoa and is now a division of Kawneer. Mr. Randall continues to give back to the community through his position on the Board of Trustees at the University of Pittsburgh, his work with the United States Chamber of Commerce, and as a board member of both the Allegheny Conference on Community Development and the Pittsburgh Regional Alliance.
Congratulations to all!
Illustration: University of Pittsburgh Big Idea Center.
ISHLT Names Newly Elected Professional Community Leaders

Drs. Mary Amanda Dew (top) and Pablo Sanchez
The International Society of Heart and Lung Transplantation (ISHLT) has announced the results of the 2022 member elections for Professional Community Leaders, who serve as representatives to the four ISHLT Interdisciplinary Network Steering Committees. Seventeen leaders across seven professional specialties were elected to serve three-year terms beginning May 1, 2022, immediately following the 2022 ISHLT Annual Meeting. Two of the 17 leaders elected are McGowan Institute for Regenerative Medicine affiliated faculty members:
Mary Amanda Dew, PhD
Professor of Psychiatry, Psychology, Epidemiology, Nursing, and Biostatistics at the University of Pittsburgh and a Professor at the Clinical and Translational Science Institute. She is the Director of the Clinical Epidemiology Program, Western Psychiatric Institute and Clinic, and the Director of the Quality of Life Research, Artificial Heart Program, Adult Cardiothoracic Transplantation, at UPMC.
Research and Immunology Representative, Advanced Heart Failure and Transplantation Interdisciplinary Network
Brack G. Hattler Professor in Cardiothoracic Transplantation
Chief, Division of Lung Transplant and Lung Failure, Department of Cardiothoracic Surgery, at the University of Pittsburgh School of Medicine
Cardiothoracic Surgery Representative, Advanced Lung Failure and Transplantation Interdisciplinary Network
“The Professional Communities are our most direct connection to ISHLT members, their views, and ways in which we can promote and improve the care of patients with advanced thoracic organ disease,” says Joseph Rogers, MD, Chair of the Governance Nominating Committee and ISHLT Past President. “They also serve a critical role in ISHLT project development, in the identification and development of the next group of leaders, and by adding to the richness of our multi-disciplinary society. We are fortunate to have these talented leaders galvanize and guide our Professional Communities and serve as vital links with our 3,500 members around the world.”
The International Society for Heart and Lung Transplantation is a not-for-profit, multidisciplinary professional organization dedicated to improving the care of patients with advanced heart or lung disease through transplantation, mechanical support, and innovative therapies. With members in more than 50 countries, ISHLT is the world’s largest organization dedicated to the research, education, and advocacy of end-stage heart and lung disease. ISHLT members represent more than 15 different professional disciplines. For more information, visit www.ishlt.org.
Department of Physical Therapy Professor Named a Gerontological Society of America Fellow

McGowan Institute for Regenerative Medicine affiliated faculty member Jessie VanSwearingen, PhD, PT, Professor of Physical Therapy in the School of Health and Rehabilitation Sciences, was named a Fellow of the Gerontological Society of America (GSA) for her outstanding contributions to the group.
GSA is the nation’s largest interdisciplinary organization devoted to the field of aging. Being named a Fellow is an acknowledgment of not only remarkable research, but also extraordinary dedication to the field of gerontology.
Dr. VanSwearingen is also a Catherine Worthingham Fellow of the American Physical Therapy Association, the association’s highest membership category.
Congratulations, Dr. VanSwearingen!
Dr. Ernest Manders Receives Scientific Achievement Award

The Scientific Achievement Award sponsored by Thermo Fisher Scientific Inc. and awarded by Life Sciences PA recognizes a scientist in the Pennsylvania life sciences community who has demonstrated outstanding achievement by advancing scientific knowledge, innovation, and/or patient care in their lifetime. McGowan Institute for Regenerative Medicine affiliated faculty member Ernest Manders, MD, Professor of Plastic Surgery (Emeritus), Department of Plastic Surgery, University of Pittsburgh, received this year’s award.
With a career that has spanned over 40 years, Dr. Manders pioneered numerous technologies within the field of medical devices. His differential breast expander has been the basis of soft tissue expansion for breasts for over 30 years. The differential breast expander design revolutionized breast reconstruction with expanders, creating an expansion following mastectomy that better mimicked the shape of the breast. This innovation is still the basis of soft tissue expander designs used today.
His innovations around the sterilization and preservation of tissues are the basis of Promethean LifeSciences, Inc., a Pittsburgh-based biotechnology company founded by Dr. Manders. Products based on his inventions have been used on millions of patients around the world.
Congratulations, Dr. Manders!
Regenerative Medicine Podcast Update
The Regenerative Medicine Podcasts remain a popular web destination. Informative and entertaining, these are the most recent interviews:
#232 –– Dr. Urszula Zandowicz discusses her work in orthopaedic surgery and treatments for knee injury.
Visit www.regenerativemedicinetoday.com to keep abreast of the new interviews.
PUBLICATION OF THE MONTH
Author: Xinxiu Xu, Kang Jin, Abha S. Bai, Wenjuan Zhu, Hisato Yagi, Timothy N. Feinstein, Phong K. Nguyen, Joseph D. Criscione, Xiaoqin Liu, Gisela Beutner, Kalyani B. Karunakaran, Krithika S. Rao, Haoting He, Phillip Adams, Catherine K. Kuo, Dennis Kostka, Gloria S. Pryhuber, Sruti Shiva, Madhavi K. Ganapathiraju, George A. Porter Jr., Jiuann-Huey Ivy Lin, Bruce Aronow, Cecilia W. Lo
Title: Uncompensated mitochondrial oxidative stress underlies heart failure in an iPSC-derived model of congenital heart disease
Summary: Hypoplastic left heart syndrome (HLHS) is a severe congenital heart disease with 30% mortality from heart failure (HF) in the first year of life, but the cause of early HF remains unknown. Induced pluripotent stem-cell-derived cardiomyocytes (iPSC-CM) from patients with HLHS showed that early HF is associated with increased apoptosis, mitochondrial respiration defects, and redox stress from abnormal mitochondrial permeability transition pore (mPTP) opening and failed antioxidant response. In contrast, iPSC-CM from patients without early HF showed normal respiration with elevated antioxidant response. Single-cell transcriptomics confirmed that early HF is associated with mitochondrial dysfunction accompanied with endoplasmic reticulum (ER) stress. These findings indicate that uncompensated oxidative stress underlies early HF in HLHS. Importantly, mitochondrial respiration defects, oxidative stress, and apoptosis were rescued by treatment with sildenafil to inhibit mPTP opening or TUDCA to suppress ER stress. Together these findings point to the potential use of patient iPSC-CM for modeling clinical heart failure and the development of therapeutics.
Source: Cell Stem Cell. 2022 Apr 7;S1934-5909(22)00101-1. doi: 10.1016/j.stem.2022.03.003. Online ahead of print.
GRANT OF THE MONTH
PI: Donghun Shin
Title: Delineating the molecular mechanisms of hepatocyte-to-cholangiocyte reprogramming
Description: Biliary epithelial cells (BECs; also called as cholangiocytes) that line the hepatic biliary tree control bile composition and flow. Injury to the BECs leads to cholestasis, which can progress to fibrosis, cirrhosis, and liver failure. Cholestatic liver diseases are associated with high morbidity and mortality; however, few effective therapies are available. In fact, liver transplantation is the only life-extending treatment for end-stage cholestatic liver diseases, but the shortage of donor livers makes this therapy extremely limited. In the injured liver with biliary damage, hepatocytes (HCs) can contribute to BECs to recover from the loss of BECs. Recent studies in mice have shown that HC-derived BECs contribute to the intrahepatic bile ducts, thereby restoring appropriate bile flow. Patients with biliary obstruction or cholangiopathies also exhibit biliary marker expression in HCs, suggesting their reprogramming into BECs. Thus, augmenting innate HC-to-BEC reprogramming in cholestatic liver diseases is an attractive therapeutic alternative to ameliorate cholestasis and subsequent cirrhosis. To develop such a therapy, it is crucial to better understand the molecular mechanisms underlying HC-to-BEC reprogramming. Furthermore, identifying small molecules that can augment the reprogramming should provide promising therapeutic drugs for patients with cholestatic liver diseases. Our long-term goal is to completely delineate the molecular mechanisms underlying HC-to-BEC reprogramming. As a first step in pursuit of that goal, the objective of this proposal is to determine the cellular and molecular characteristics of HC-to-BEC reprogramming-driven biliary regeneration in our two innovative zebrafish models and to elucidate how histone deacetylase 1 (hdac1) regulates HC-to-BEC reprogramming. Based on our preliminary data obtained from pharmacological and genetic studies, we hypothesize that Hdac1 inhibition promotes HC-to-BEC reprogramming by derepressing the Notch receptor gene notch2 and the signal transducer and activator of transcription 3 gene (stat3). We will test this hypothesis and accomplish the objective of this application by (1) elucidating the entire process of HC-to-BEC reprogramming-driven biliary regeneration in the two zebrafish models, in which complete absence of BECs is achieved and subsequently HCs convert to BECs, (2) determining the effects of Hdac1 inhibition on HC-to-BEC reprogramming in both zebrafish and mice, and (3) elucidating the molecular mechanisms by which Hdac1 inhibition promotes the reprogramming. The successful accomplishment of the proposed research will not only provide novel molecular mechanisms underlying HC-to-BEC reprogramming but also suggest HDAC1/2 inhibitors as promising therapeutic drugs to promote the reprogramming in patients with cholestatic liver diseases.
Source: National Institute of Diabetes and Digestive and Kidney Diseases
Term: April 1, 2022 – March 31, 2026
Amount: $514,858 (one year)
