August 2020 | VOL. 19, NO. 8| www.McGowan.pitt.edu
Pigs Grow New Liver in Lymph Nodes
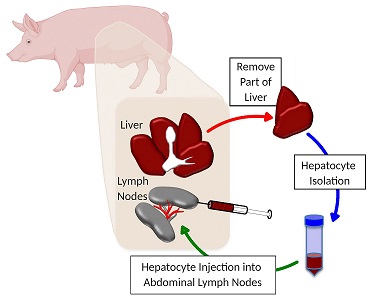
Hepatocytes — the chief functional cells of the liver — are natural regenerators, and the lymph nodes serve as a nurturing place where they can multiply. In a new study published in the journal Liver Transplantation, researchers in the Lagasse Laboratory showed that large animals with ailing livers can grow a new organ in their lymph nodes from their own hepatocytes. A human clinical trial is next.
“It’s all about location, location, location,” said senior author McGowan Institute for Regenerative Medicine faculty member Eric Lagasse, PharmD, PhD, associate professor of pathology at Pitt. “If hepatocytes get in the right spot and there is a need for liver functions, they will form an ectopic liver in the lymph node.”
The cells of the liver normally replenish themselves, but need a healthy, nurturing environment to regenerate. However, in end-stage liver disease, the liver is bound up by scar tissue and too toxic for the cells to make a comeback.
“The liver is in a frenzy to regenerate,” said Dr. Lagasse, who also is a member of the Pittsburgh Liver Research Center. “The hepatocytes try to repair their native liver, but they can’t and they die.”
Nearly a decade ago, Dr. Lagasse noticed that if he injected healthy liver cells into the lymph nodes of a mouse, they would flourish, forming an auxiliary liver to take over the tasks of the animals’ genetically induced malfunctioning liver.
But mice are small. Dr. Lagasse and colleagues needed to show that a large animal could grow a meaningful mass of secondary liver tissue to overcome liver disease.
To mimic human liver disease in pigs, the researchers diverted the main blood supply from the liver, and at the same time, they removed a piece of healthy liver tissue and extracted the hepatocytes. Those liver cells were then injected into the abdominal lymph nodes of the same animal they came from.
All six pigs showed a recovery of liver function, and close examination of their lymph nodes revealed not only thriving hepatocytes, but also a network of bile ducts and vasculature that spontaneously formed among the transplanted liver cells.
The auxiliary livers grew bigger when the damaged tissue in the animals’ native liver was more severe, indicating that the animals’ bodies are maintaining an equilibrium of liver mass, rather than having runaway growth akin to cancer.
These findings bolster the results of another recent study, in which Dr. Lagasse and colleagues at Mayo Clinic showed that healthy liver tissue grown in the lymph nodes of pigs with a genetic liver defect spontaneously migrated to the animals’ livers, where they replaced diseased cells and cured the animals’ liver disease.
So, regardless of the cause of liver disease, from hepatitis to alcoholism, Dr. Lagasse expects growing auxiliary livers in the lymph nodes will help.
Additional authors on the study include McGowan Institute affiliated faculty member Paulo Fontes, MD, James Marsh, MD, and Roberto Lopez, MD, all of West Virginia University and previously at UPMC; Junji Komori, MD, PhD, of Takamatsu Red Cross Hospital and previously at Pitt.
Illustration: Growing a New Liver in the Lymph Nodes: Part of the liver is removed and hepatocytes isolated and injected back into the lymph nodes of the same animal.
RESOURCES AT THE MCGOWAN INSTITUTE
September Histology Special
The Histology lab offers decalcification, processing and embedding, cutting and staining of those difficult bone samples. Our staff has years of experience with bone samples that include special staining and IHC on large and small bone samples that can be difficult to work with. We have even developed a technique to ensure your sample will not lift from the slide during aggressive antigen retrieval protocols.
Below are just some of the special stains we offer at the McGowan Histology Core:
The McGowan Histology Core offers TUNEL staining on paraffin embedded or frozen tissue.
You’ll receive 25% off bone samples in September when you mention this ad. Contact Julia at the McGowan Core Histology Lab by email: Hartj5@upmc.edu or call 412-624-5265.
New Sample Submission Procedures: In response to COVID-19, we ask that you contact us to schedule a drop off time. When you arrive at the building you can call our laboratory at (412)624-5365. Someone will meet you in the lobby to collect your samples. When your samples are completed, you will receive an email to schedule a pickup time.
Save the Date!
ICCAI 2020 Will Be Virtual

The 2020 International Conference on Complex Acute Illness (ICCAI’2020), titled “Meaningful AI in the Age of COVID-19”, will be held on September 10-11, 2020 entirely online, using Zoom teleconferencing technology. ICCAI is the annual meeting of the Society for Complex Acute Illness (SCAI), which, since its inception over 15 years ago, has successfully bridged the domains clinical practice of critical care medicine and quantitative approaches to understanding critical illness, with a strong translational focus. On the methodological side, it has emphasized eclecticism, covering, through its interdisciplinary membership, modeling approaches ranging from pure mechanistic differential equations models and agent-based models to purely data-driven machine learning techniques.
Please view the program and access the free registration site at http://iccai.org/
SCIENTIFIC ADVANCES
Spinal Stimulators Repurposed to Restore Touch in Lost Limb

Imagine tying your shoes or taking a sip of coffee or cracking an egg but without any feeling in your hand. That’s life for users of even the most advanced prosthetic arms.
Although it’s possible to simulate touch by stimulating the remaining nerves in the stump after an amputation, such a surgery is highly complex and individualized. But according to a new study from the University of Pittsburgh’s Rehab Neural Engineering Labs, spinal cord stimulators commonly used to relieve chronic pain could provide a straightforward and universal method for adding sensory feedback to a prosthetic arm. McGowan Institute for Regenerative Medicine affiliated faculty member Michael Boninger, MD, Senior Medical Director for Post-Acute Care for the Health Services Division of UPMC and President of UPMC Innovative Homecare Solutions and Professor in the Department of Physical Medicine & Rehabilitation at the University of Pittsburgh, School of Medicine, with joint appointments in the Departments of Bioengineering, Rehabilitation Science and Technology, and the Clinical and Translational Science Institute, is a co-author on this work.
For this study, published today in eLife, four amputees received spinal stimulators, which, when turned on, create the illusion of sensations in the missing arm.
“What’s unique about this work is that we’re using devices that are already implanted in 50,000 people a year for pain — physicians in every major medical center across the country know how to do these surgical procedures — and we get similar results to highly specialized devices and procedures,” said study senior author Lee Fisher, PhD, assistant professor of physical medicine and rehabilitation, University of Pittsburgh School of Medicine.
The strings of implanted spinal electrodes, which Fisher describes as about the size and shape of “fat spaghetti noodles,” run along the spinal cord, where they sit slightly to one side, atop the same nerve roots that would normally transmit sensations from the arm. Since it’s a spinal cord implant, even a person with a shoulder-level amputation can use this device.
Dr. Fisher’s team sent electrical pulses through different spots in the implanted electrodes, one at a time, while participants used a tablet to report what they were feeling and where.
All the participants experienced sensations somewhere on their missing arm or hand, and they indicated the extent of the area affected by drawing on a blank human form. Three participants reported feelings localized to a single finger or part of the palm.
“I was pretty surprised at how small the area of these sensations was that people were reporting,” Dr. Fisher said. “That’s important because we want to generate sensations only where the prosthetic limb is making contact with objects.”
When asked to describe not just where but how the stimulation felt, all four participants reported feeling natural sensations, such as touch and pressure, though these feelings often were mixed with decidedly artificial sensations, such as tingling, buzzing, or prickling.
Although some degree of electrode migration is inevitable in the first few days after the leads are implanted, Dr. Fisher’s team found that the electrodes, and the sensations they generated, mostly stayed put across the month-long duration of the experiment. That’s important for the ultimate goal of creating a prosthetic arm that provides sensory feedback to the user.
“Stability of these devices is really critical,” Dr. Fisher said. “If the electrodes are moving around, that’s going to change what a person feels when we stimulate.”
The next big challenges are to design spinal stimulators that can be fully implanted rather than connecting to a stimulator outside the body and to demonstrate that the sensory feedback can help to improve the control of a prosthetic hand during functional tasks like tying shoes or holding an egg without accidentally crushing it. Shrinking the size of the contacts — the parts of the electrode where current comes out — is another priority. That might allow users to experience even more localized sensations.
“Our goal here wasn’t to develop the final device that someone would use permanently,” Dr. Fisher said. “Mostly we wanted to demonstrate the possibility that something like this could work.”
Dr. Kacey Marra Forms AxoMax Technologies to Commercialize Nerve Repair Research

The path to impact for basic life science research can be long and filled with big gaps: in time and in resources.
A case in point is the research of McGowan Institute for Regenerative Medicine faculty member Kacey Marra, PhD, Professor in the Departments of Plastic Surgery in the School of Medicine and Bioengineering in the School of Engineering at the University of Pittsburgh and is the Vice Chair of Research for the Department of Plastic Surgery, on repairing large gaps in traumatically injured nerves.
Dr. Marra had come back to the University of Pittsburgh in 2002, where she had earned her undergraduate degree and a PhD in organic chemistry in 1996 and set up a lab following a research stint at neighboring Carnegie Mellon University.
About 15 years ago she filed an invention disclosure with the Innovation Institute for a device to repair severe nerve injuries with long gaps (more than two inches). A provisional patent application was filed, and she continued her research after attracting the attention of the Department of Defense, which saw the opportunity to advance treatments for soldiers injured in battle.
“I met a soldier who had been shot through his calf and had no sensation in his foot. To create something that could help people like him live a more independent and fulfilling life strengthened my resolve to do what it takes to commercialize our lab’s discoveries,” Dr. Marra said.
In her DOD-funded animal study, Dr. Marra demonstrated that a biodegradable tube containing a time-released protein growth factor could restore up to 80 percent of nerve function.
Looking beyond battlefield injuries, Dr. Marra said the nerve guide device could have applications for people injured in car accidents, machinery accidents, and potentially, facial nerve paralysis, newborn nerve injuries incurred during delivery, nerve damage due to tumor removal, and diabetic neuropathy.
Early on in her DOD-funded research, Dr. Marra participated in an eight-week summer program on commercializing research through the National Science Foundation.
With her research showing continuing promise, when Pitt updated its policies on faculty participation in startups in 2018, Dr. Marra began to plan for the launch of her own company, AxoMax Technologies.
“It was becoming a better environment at Pitt to be an entrepreneur,” she said, referring to the support she has received from entrepreneurs in residence at the Innovation Institute that she has consulted along the way.
Dr. Marra admits that the added responsibility of starting a company on top of her regular duties has been challenging.
“At first, I would try to block off one day a week to do startup-up related work, but things happen every day, so it’s become more nights and weekends. If you want to make it successful, that’s what you need to do,” she said.
Not that the work is not without reward.
In June, Dr. Marra entered the Equalize2020 competition for female academic entrepreneurs. She was selected as one of 12 finalists to present a pitch. She received the People’s Choice award, which she said was significant because it meant that she was able to make the technology understandable to the audience.
That is important as she embarks on raising a seed round of investment in her company. She is also looking to hire a CEO to help the company take its next steps towards applying for approval from the Food and Drug Administration and conducting clinical trials.
Dr. Jenny Yu and the Treatment of Thyroid Eye Disease at Home

Chartwell Pennsylvania, LP, a provider of home infusion, specialty pharmacy, and enteral nutrition, recently became the first provider to administer TEPEZZA (teprotumumab-trbw) in a home-infusion setting. Approved on January 21, 2020, TEPEZZA is the first and only FDA-approved medicine for the treatment of thyroid eye disease (TED), a rare and serious autoimmune disease. Home infusion therapy continues to ease the burden of care placed on hospitals and other health care facilities throughout the COVID-19 outbreak.
The first TEPEZZA home infusion therapy was administered on TMarch 24, 2020, at the patient’s residence. The medication is administered once every three weeks for a total of eight infusions.
A prescribing physician for multiple Chartwell teprotumumab-trbw patients, Jenny Yu, MD, Vice Chair of Clinical Operations for the Department of Ophthalmology, UPMC Eye Center, an Assistant Professor of Ophthalmology and Otolaryngology, University of Pittsburgh, and a McGowan Institute for Regenerative Medicine affiliated faculty member, called the situation “the perfect storm” for many patients with TED. Dr. Yu, who also serves on the UPMC Orbital, Oculoplastic, and Aesthetic Surgery Service, cited not only the pandemic, but also the importance of timing in the patient’s treatment.
“The drug in treatment is time-sensitive, in terms of catching the patient in the inflammatory phase,” Dr. Yu said. “Of course, we also had COVID-19 to tend with. Home infusion limits the kind of exposure to infection that you would find in a hospital or outpatient setting. Younger patients without many other comorbidities are excellent candidates for the initial infusion in the home.”
“I see a lot of patients from other UPMC locations coming into Pittsburgh for a specialty evaluation,” Dr. Yu said. “To be able to bring the treatment options closer to the patient’s home or in their home—it just makes sense.”
Blood-Thinner with No Bleeding Side-Effects Is On the Horizon

Patients who suffer from thrombosis, pulmonary embolism or stroke are usually put on drugs that help their blood flow more smoothly through their body. Occupying a large section of the drug market, anticoagulants, or “blood thinners” as they are popularly known, can keep blood clots from forming or getting bigger, and can therefore help with recovery from heart defects or prevent further complications.
But there is a catch: blood thinners work by blocking enzymes that help to stop bleeding after an injury. Because of this, virtually every blood thinner available today can lead to serious, and even life-threatening bleeding following an injury.
The problem remained unsolved until a few years ago, when a study was carried out on mice that had been genetically modified to be deficient in an enzyme that normally helps blood clot. The enzyme is called “coagulation factor XII” (FXII), and the mice without the enzyme had a very reduced risk of thrombosis without having bleeding side-effects. The discovery triggered a race for FXII inhibitors.
Finally, a synthetic inhibitor
Participating in the race, the Laboratory of Therapeutic Proteins and Peptides of Professor Christian Heinis, PhD, at Ecole Polytechnique Fédérale de Lausanne (EPFL) has developed the first synthetic inhibitor of FXII. The inhibitor has high potency, high selectivity, and is highly stable, with a plasma half-life of over 120 hours. Published in Nature Communications, the study is the result of an extensive collaboration with three other labs in Switzerland and the US. McGowan Institute for Regenerative Medicine affiliated faculty member Keith Cook, PhD, Professor and Associate Department Head for Graduate Education at Carnegie Mellon University (CMU) in its Department of Biomedical Engineering, is a co-author on the study.
“The FXII inhibitor is a variation of a cyclic peptide that we identified in a pool of more than a billion different peptides, using a technique named phage display,” says Dr. Heinis. The researchers then improved the inhibitor by painstakingly replacing several of its natural amino acids with synthetic ones. “This wasn’t a quick task; it took over six years and two generations of PhD students and post-docs to complete.”
With a potent FXII inhibitor in hand, Dr. Heinis’s group wanted to evaluate it in actual disease models. To do this, they teamed up with experts in blood and disease-modeling at the University Hospital of Bern (Inselspital) and the University of Bern.
Working with the group of Professor Anne Angellillo-Scherrer (Inselspital), they showed that the inhibitor efficiently blocks coagulation in a thrombosis model without increasing the bleeding risk. Then they assessed the inhibitor’s pharmacokinetic properties with the group of Professor Robert Rieben (University of Bern). “Our collaboration found that it is possible to achieve bleeding-free anti-coagulation with a synthetic inhibitor,” says Dr. Heinis.
Artificial lungs
“The new FXII inhibitor is a promising candidate for safe thromboprotection in artificial lungs, which are used to bridge the time between lung failure and lung transplantation,” says Dr. Heinis. “In these devices, contact of blood proteins with artificial surfaces such as the membrane of the oxygenator or tubing can cause blood clotting.” Known as ‘contact activation’, this can lead to severe complications or even death and limits the use of artificial lungs for longer than a few days or weeks.
To test the effectiveness of the FXII inhibitor in artificial lungs, Dr. Heinis’s group turned to Dr. Cook, an expert for artificial lung system engineering. Dr. Cook’s group tested the inhibitor in an artificial lung model, and found that it efficiently reduced blood clotting, all without any bleeding side-effects.
The only problem is that the inhibitor has a relatively short retention time in the body: it is too small, and the kidneys would filter it out. In the context of artificial lungs, this would mean constant infusion, since suppressing blood clotting for several days, weeks or months requires a long circulation time.
But Dr. Heinis is optimistic: “We’re fixing this; we’re currently engineering variants of the FXII inhibitor with a longer retention time.”
Surgical Benefits of Artificial Intelligence

As a Senior Staff Writer for Orthopedics Today, Ms. Casey Tingle recently reviewed the impact of artificial intelligence (AI) in the health care sector. In her article she highlighted where AI may provide benefits to patients with musculoskeletal injuries. Her work included input from McGowan Institute for Regenerative Medicine deputy director Stephen Badylak, DVM, PhD, MD, professor of surgery at the University of Pittsburgh, who said AI may be able to identify whether tissues that are healing from a traumatic injury will have a good or bad outcome based on certain biomarkers.
“For example, the more scar tissue and adhesions you get, the worse the clinical outcome is going to be,” Dr. Badylak said. “If we knew early on that certain things were starting to go down the wrong pathway, we could intervene either surgically or in other ways that could change the direction of wound healing,” he said.
Once the healing biomarkers are identified, Dr. Badylak noted AI, in the form of smart bandages, can be developed that, when used on wounds following a traditional repair procedure, can indicate in which direction the wound is healing and provide guidance on how surgeons can intervene, and when. Currently, Dr. Badylak and his colleagues are using AI to develop methods “that will decrease the time to functional healing of injured musculoskeletal tissue by 50%.”
“In other words, someone comes in with a traumatic injury, they have a fractured femur and they lost 50% of their quadriceps muscle. That is a pretty serious injury. Right now the expectation is we are going to do the best we can to salvage this and we are going to get to a point where we have scar tissue with a lot of morbidity and limited function,” Dr. Badylak told Orthopedics Today. “Well, the AI tells us that does not have to be the case. We can not only restore most of the functional tissue that was gone, but we could do it in half of the time if we only knew when to intervene and how to intervene both surgically and medically.”
Barriers of AI
Because not every hospital will have the resources to run and upkeep predictive models, Dr. Badylak said the models must be developed in an understandable and user-friendly way, especially in light of the current barriers to implementing AI as a standard of care. He said regardless of whether they are user-friendly, the more advanced AI designed for treatment of musculoskeletal injuries may be disadvantaged by regulatory barriers, as well as funding issues.
“The only way you are going to [obtain] third-party reimbursement is if you can show that, in the long run, this is more cost effective,” Dr. Badylak said. “That takes years to do because the cost benefits are going to be realized over years,” he said.
‘Keep an open mind’
Although Dr. Badylak said surgeons should keep an open mind regarding AI use in their practices, he also advises them to educate themselves and not believe everything they hear.
“If we are feeding the wrong information into a device that works on AI, then we are not going to get the information out of it that we want,” Dr. Badylak said. “In addition to keeping an open mind, we have to put forth the effort to understand how to take full advantage of what is being developed, otherwise we are going to either not make any advances or even have worse outcomes because we have provided the wrong information and expect a machine to fix it for us, and that does not happen.”
UPMC Children’s and Pitt Researcher Receives $2.59 Million NIH Grant for Diabetes Gene Therapy

The National Institutes of Health (NIH) National Institute of Diabetes and Digestive and Kidney Diseases has awarded a grant of $2.59 million to George Gittes, MD, director of the Richard King Mellon Foundation Institute for Pediatric Research, co-scientific director at UPMC Children’s Hospital of Pittsburgh, and affiliated faculty member of the McGowan Institute for Regenerative Medicine, to support continuing development of gene therapy technology that may have the potential to cure Type 1 and Type 2 diabetes, which affects approximately 10% of the U.S. population, or more than 34 million people.
“Diabetes gene therapy could eliminate the need for insulin replacement for people with diabetes and holds the potential to provide long-term effectiveness, or even be a cure for diabetic patients,” said Dr. Gittes, also chief of pediatric surgery at UPMC Children’s.
Developed by Dr. Gittes, the gene therapy technology has restored normal glucose levels in the blood of mice by reprogramming beta cells in the pancreas.
“This funding will continue to support our research in diabetic primates as we move toward human clinical trials,” said Dr. Gittes. “We saw encouraging data in our preclinical mice studies, where the gene therapy reprogrammed pancreatic cells to restore normal blood glucose levels in diabetic mice for approximately four months, which could translate to decades in humans. More recently, preliminary results in monkeys also have been very promising.”
This technology is the subject of an exclusive license agreement entered between Genprex and the University of Pittsburgh in February of 2020. Dr. Gittes is the inventor of the gene therapy for diabetes, and he is eligible to receive royalties on this technology in the future.
Project on Analyzing Cells for Future Biomedical Devices Awarded 5-Year NIH Grant

McGowan Institute for Regenerative Medicine Yu-li Wang, PhD, and his team are researching cell migration while using the technologies developed for potential applications in artificial organs and other devices. The team has been awarded a five-year research grant from the National Institute of General Medical Sciences (NIGMS), one of the National Institutes of Health, to support this work.
“We are one of the world leaders in the field of mechanobiology, and we show that if you want to design an artificial material or organ to work properly, you need to pay attention to how living cells interact with materials,” said Dr. Wang, professor of biomedical engineering at Carnegie Mellon University. “You have to look not only at chemical interactions but also the mechanical properties.”
Cell migration is the directed movement of a cell or group of cells in response to chemical or mechanical signals. It is a fundamental cellular process throughout life, from embryonic development to death. Given the complexity of even the simplest tissue, it is almost impossible to put correct types of cells directly in the correct location to form a native or artificial organ without involving some forms of migration.
This research toes the fuzzy line between foundational and translational research. Dr. Wang and his team have developed both tools and fundamental knowledge that are critical for the successful design of biomedical devices and interventions.
At the center of the research is how cells communicate with each other mechanically. Cells have been observed to generate forces and apply them both to the environment and the neighboring cells — they tug on their neighbors to communicate with them.
“We need to know how they conduct that kind of communication and then how we can design artificial materials that facilitate communication in order to direct cells to their destination,” Dr. Wang said.
For example, experiments have been conducted in biomedicine to inject stem cells in part of a diseased heart where cells have died. If the stems cells are injected without paying attention to the surrounding environment (i.e. changes in the diseased tissue), they will never turn themselves into normally functioning heart cells. Researchers need to observe the environment and figure out how to modify it so the injected stem cells can successfully transform into part of a healthy heart. The same idea applies for artificial organs and engineered tissue, meaning Dr. Wang’s work is general knowledge capable of affecting many kinds of translational research.
“In order to facilitate our research, we are developing new methods specifically designed to help solve problems,” Dr. Wang said. “From my experience, the highest impact often comes with the introduction of a new technology that not only enables us to solve our problems but contributes to broad, general applications.”
One of the technologies Dr. Wang and his team are developing is 3D printing for polymeric hydrogel, a material that could potentially be a base for artificial organs. This particular hydrogel is permeable and non-sticky and has been used in some types of contact lenses. However, wider applications have not been possible, in part because there was no technology for forming complex shapes. The team has successfully developed 3D printing technology to solve this problem.
The team has also developed a machine learning approach that tracks migrating cells over time and distance. With part of their research focused on detecting the mechanical forces generated by cells in the environment, machine learning allows the team to streamline the calculation and bypass common limitations and caveats of conventional methods.
The broad accessibility and impact of Dr. Wang’s research is why it is supported by NIGMS, which specifically funds research that carries the potential for general advances in human health and medicine. This type of grant is specifically designed to support senior researchers with strong track records in funded research. Instead of the typical period of four years, it provides five years of funding and gives researchers a significant degree of freedom to develop the direction of their work within a general frame of purpose. This is particularly important in a rapidly developing field.
“We can modify the approach and direction of research to take advantage of any new developments,” Dr. Wang said. That is particularly important in our field, which is developing very quickly. The grant encourages us to maximize our creativity and imagination.”
Drs. Jelena Janjic and Vijay Gorantla Receive Department of Defense Research Grant

McGowan Institute for Regenerative Medicine affiliated faculty members Jelena Janjic, PhD, Associate Professor of Pharmaceutics in the Graduate School of Pharmaceutical Sciences and Mylan School of Pharmacy at Duquesne University and the Founder and Co-Director of the Chronic Pain Research Consortium at Duquesne, and Vijay Gorantla, MD, PhD, Associate Professor of Surgery, Ophthalmology and Bioengineering at the Wake Forest School of Medicine, will serve as co-principal investigators on a recent grant funded through the Department of Defense Congressionally Directed Medical Research Program. Dr. T. Kevin Hitchens, Director of Preclinical MRI, UPMC Hillman Cancer Center; Director of the Animal Imaging Center, University of Pittsburgh School of Medicine; and Research Associate Professor of Neurobiology, University of Pittsburgh is also a co-principal investigator. The award of $1.5M over three years from the Reconstruction Transplant Research Program will focus on the project entitled “Nanoimaging for Noninvasive Monitoring of Donor and Recipient Immune System Contribution to Acute and Chronic Rejection in VCA.” The abstract of this work follows:
Despite two decades of experience with clinical vascularized composite allotransplantation (VCA), the sensitivity and specificity of skin rash as a marker of acute rejection (AR) and the sampling error on skin biopsies remains unknown. The extent and intensity of skin involvement by clinically apparent AR (e.g., papular or erythematous rash) is usually the initial driver of management. One of the key reasons for treatment failure and recurrent rejection in VCA is lack of non-invasive deep tissue-penetrating imaging modalities that can provide the clinician true state of the graft during the course of treatment and lifetime of the graft. Many of these cases with chronic rejection (CR) have been associated with medication non-compliance (NC) that causes wide fluctuations of systemic drug levels with periodic or persistent under-immunosuppression, leading to smoldering immune responses in the graft. Thus, missed interpretation or underreporting of CR (as current Banff criteria do not include CR) across all VCA may be more prevalent than assumed, given the constraints of access to protocol/for-cause deep tissue biopsies. There is thus an imminent need for non-invasive tools that enable reliable and reproducible, quantitative, and objective diagnosis and monitoring of rejection in deep graft tissues otherwise inaccessible by skin biopsy.
Novel nanoprobes capable of immunolabeling multiple cell populations in vivo can shed critical insights into the dynamics, kinetics, intensity, and spatial patterns or progression of cellular VCA rejection across deep and superficial tissues, as well as objectively quantify responsiveness or resistance of rejection to immunosuppressive therapy. The proposed nanoimaging probes will be produced by GMP-scalable methodologies using only materials already deemed safe for use in humans. Proposed nanoimaging agents are designed for safe and effective immune cell tracking in small and large animals and, as such, are readily translatable to clinical VCA. Our proposed technology meets the needs of VCA recipients and clinicians for accurate, non-invasive diagnosis and monitoring of rejection in superficial and deep tissues, assessment of therapeutic response, and, consequently, true treatment personalization and assessment of patient compliance.
We hypothesize that our proposed cellular nanoimaging biomarker approach using multispectral probes will noninvasively detect, quantify, and monitor macrophage-driven immune processes in superficial and deep tissues of VCA during AR or CR and assess therapeutic efficacy and treatment response to immunosuppressive interventions. Proposed tools also offer unbiased quantitative patient-reporting independent compliance assessment. To test this hypothesis, in Aim 1, we will manufacture and validate in small animals multispectral and multimodal nanoimaging agents. In Aim 2, we will test these agents in rodent models of VCA to establish immune cell distribution patterns in acute and chronic rejection and, in Aim 3, we will validate this technology as a therapeutic response biomarker in acute and chronic rejection in a non-human primate VCA model. Additionally, we will evaluate in a non-human primate model, for the first time, nanoimaging methods as unbiased biomarkers for non-invasive monitoring of patient compliance.
Proposed strategies are an unprecedented advancement in VCA, where standard-of-care diagnosis and management are guided by invasive graft monitoring and where for-cause superficial skin biopsies are not reliable indicators of deeper tissue rejection. Presented nanoimaging methods can potentially provide unique first insights into the cellular dynamics of deep tissue rejection, its spatiotemporal association with skin AR, and overall therapeutic responses of superficial versus deep tissue rejection in VCA. Such noninvasive immune surveillance can facilitate precise immunosuppression management in Service members as well as civilians receiving VCA for devastating tissue loss, enabling optimization/personalization of immunosuppression as well as unbiased monitoring of patient adherence to medications. The long-term vision of this technology lies in its scope and scalability to wider populations beyond combat/civilian subjects seeking VCA, such as immunotherapy monitoring, inflammatory diseases (IBD, RA, lupus), and autoimmune diseases.
Congratulations, Drs. Janjic and Gorantla!
The American Liver Foundation and UPMC Partner to Raise Awareness of Living-Donor Liver Transplants

The American Liver Foundation (ALF), in partnership with the University of Pittsburgh Medical Center (UPMC), launched “The Greatest Gift Initiative” to raise awareness about lifesaving living-donor liver transplantation. This initiative teaches Americans with end-stage liver disease strategies for finding a living donor and inspires more people to consider donating a part of their liver.
While the COVID-19 pandemic has created additional challenges for patients on the liver transplant waiting list, there has long been a large disparity between the number of deceased donor organs available for transplant and the growing number of patients on the liver transplant waiting list. Currently, over 1,500 people die in the U.S. each year while waiting for a deceased-donor liver to become available for transplantation. The “Greatest Gift Initiative” aims to bring that number to zero.
“If you look at the history of liver transplantation, it really started here in Pittsburgh. Fast forward 30 years and we are still at the forefront. Our philosophy is that anyone who needs a liver transplant can potentially qualify for a living-donor liver transplant. That is why our partnership with the American Liver Foundation and on-going efforts to raise awareness and increase education about living-donor liver transplants is so important,” says Abhinav Humar, MD, Chief of Transplantation at UPMC and an affiliated faculty member of the McGowan Institute for Regenerative Medicine. “Our mission is to help patients reduce time spent on the transplant waiting list so they can get back to enjoying life with their families and loved ones.”
A survey conducted by WebMD, in partnership with UPMC, in 2019 found that 80% of respondents reported a lack of understanding about the liver transplant process, and only 1 in 10 liver patients and caregivers stated that living donation was discussed by their physician. Increased awareness of living-donor liver transplant is key to addressing this gap. UPMC and the ALF are committed to working together to offer education about the benefits of living-donor liver transplant among waiting list patients and potential living donors to increase access to living-donor liver transplant and ultimately decrease waiting list mortality.
“At ALF, we envision a future in which no one must die waiting for a liver, and the general public is aware of the miracle of living-donor liver transplantation. If Americans want to know what they can do to help others during this unprecedented time, one option is to consider become a living donor,” said Lynn Seim, MSN, RN, Executive Vice President and Chief Operating Officer, American Liver Foundation. “There are many reasons living-donor liver transplantation is underutilized; however, most often it has to do with a lack of information about this option, compounded by the stigma that surrounds liver disease. That is why we are proud to partner with UPMC, one of the leading transplant centers in the country on this initiative. We are thankful to New York-Presbyterian/Weill Cornell Medical Center/Columbia University Medical Center and Jim and Jeannie Goldberg, who are donating their time, expertise, and support to ensure the initiative’s success.”
As part of “The Greatest Gift Initiative,” ALF launched an online hub, called the Living-Donor Liver Transplant Information Center, with reliable and comprehensive information for individuals interested in becoming living donors and for those in need of a liver transplant.
Liver disease affects over 30 million people in the U.S. each year, including children and adults. Many liver diseases have silent symptoms, sometimes leading to late diagnoses and the need for liver transplantation. In 2016, the most common reasons for needing a liver transplant were alcoholic liver disease, liver cancer, fatty liver disease (nonalcoholic steatohepatitis NASH), and cirrhosis caused by chronic hepatitis. For children, the most common cause was biliary atresia. The ALF works to promote education, advocacy, awareness, and funding of research for liver disease.
Illustration: American Liver Foundation and the University of Pittsburgh Medical Center.
U.S. Department of Transportation Grants $1 Million to Pitt’s New University Transportation Center

The U.S. Department of Transportation recently announced $4.925 million in grants to fund four new Tier 1 University Transportation Centers (UTCs) to advance research and education programs that address critical transportation challenges facing our nation. The University of Pittsburgh was named one of the recipients of $1 million for its project entitled “Implications of Accessible Automated Vehicles and Mobility Services for People with Disabilities.”
Pitt will work with the Uniformed Services University of Health Sciences in Bethesda, MD, The Catholic University of America in Washington D.C., the Toyota Mobility Foundation, and the Merlin Mobility Corporation. McGowan Institute for Regenerative Medicine affiliated faculty member Rory Cooper, PhD, FISA & Paralyzed Veterans of America Professor and Distinguished Professor of the Department of Rehabilitation Science and Technology, professor of Bioengineering, Physical Medicine and Rehabilitation, and Orthopedic Surgery at the University of Pittsburgh, and Founding Director and VA Senior Research Career Scientist of the Human Engineering Research Laboratories, will serve as the lead researcher and director of the study. As reported by Kiley Koscinski, WESA, Pitt’s UTC will complete three projects within the study according to Dr. Cooper:
- A review of the scientific and public literature about accessibility transportation and autonomous vehicles
- A survey of consumers and car manufacturers including focus groups and mobility mapping
- A consumer market model where data and guidelines for autonomous vehicle developers will be displayed
Pittsburgh has become a hub for autonomous vehicle development, with companies like Uber and Argo AI testing vehicles on the streets. But Dr. Cooper worries developers aren’t thinking about accessibility as a standard to meet alongside the safety and efficiency metrics.
“There’s been autonomous vehicles on the streets of Pittsburgh for several years, and yet they’re not accessible,” Dr. Cooper said.
He expects the study to help developers and manufacturers be more mindful, “So that as these systems eventually become ubiquitous, they’re enabling for individuals with disabilities rather than creating artificial barriers.”
However, some autonomous vehicle developers in Pittsburgh have contacted Dr. Cooper with interest in the study, he said. He declined to identify which companies expressed interest.
According to Dr. Cooper, the project marries Pittsburgh’s innovative autonomous vehicle sector with its history of developing disability technology.
“I’m glad to see that we’re able to marry those two domains together,” he said. “We have the potential to change the world.”
Each UTC will conduct its research over the next 18 to 24 months. UTCs advance U.S. technology and expertise in the many modes and disciplines composing transportation and address workforce needs for the next generation of transportation leaders.
SBIR Grant to Address Microvascular Obstructions

McGowan Institute for Regenerative Medicine affiliated faculty member John Pacella, MD, Associate Professor in the School of Medicine at the University of Pittsburgh and an Interventional Cardiologist within the UPMC Heart and Vascular Institute, is partnering with Microvascular Therapeutics (MVT), a biotechnology company based in Tucson, Arizona, and a leader in microbubble technology. MVT recently was awarded a Phase I Small Business Innovation Research (SBIR) grant from the National Heart, Lung, and Blood Institute (NHLBI), a part of the National Institutes of Health.
MVT has developed a new, improved ultrasound contrast agent, MVT-100, which is currently in clinical testing and is being supported by the NHLBI. MVT is modifying the MVT-100 microbubble into smaller bubbles called nanodroplets for microvascular clot detection by incorporating a targeting molecule to bind the microbubbles to the clots.
Cardiovascular disease is a leading cause of death in the United States, with over 1 million Americans estimated to have a new or recurrent heart attack this year. Although mortality from heart attack has decreased in recent years, post-myocardial infarction (MI) congestive heart failure is increasing due to blockage of the small blood vessels in the heart by small clots. This SBIR is leveraging the unique expertise of MVT and the University of Pittsburgh Center for Ultrasound Molecular Imaging and Therapeutics.
“The team at MVT will work with Dr. John Pacella, MS, MD, Interventional Cardiologist and Associate Professor of Medicine at the University of Pittsburgh Vascular Medicine Institute, who developed animal models for microvascular obstruction, and key opinion leader, Dr. Matthias Wilson, MD, Professor at the University of Sao Paulo School of Medicine, who conducted a prospective, randomized clinical trial of treating heart attack patients using transthoracic ultrasound coupled with administration of microbubbles. At MVT we have developed smaller targeted microbubbles, called nanodroplets which will permeate the clots more easily and upon activation by ultrasounds dissolve the clots. The grant is for the in vivo proof of concept studies and translate our in vitro observations into small animal models of microvascular obstruction,” said Emmanuelle Meuillet, PhD, VP of Research and Development at MVT.
AWARDS AND RECOGNITION
Dr. Eni Halilaj Receives Post-Doctoral Young Scientist Award from American Society of Biomechanics

McGowan Institute for Regenerative Medicine affiliated faculty member Eni Halilaj, PhD, received the Post-Doctoral Young Scientist Award from the American Society of Biomechanics (ASB). This award recognizes early achievements by promising young scientists who are within five years of receiving their PhD. Selection is based upon the scientific quality of the submitted material. The awardee must attend the annual ASB meeting to present their work in a special awards session. It is expected that the awardee will submit a full-length manuscript for publication in the Journal of Biomechanics. The award consists of an engraved plaque, a check for $500, and a waiver of conference fees for the annual ASB meeting.
At Carnegie Mellon University (CMU), Dr. Halilaj is an assistant professor in mechanical engineering, with joint appointments in Biomedical Engineering and the Robotics Institute. She also holds an appointment in Orthopaedic Surgery at the University of Pittsburgh School of Medicine. Dr. Halilaj obtained her undergraduate and graduate degrees at Brown University. Before joining CMU in 2018, she was a distinguished postdoctoral fellow at Stanford University’s Mobilize Center—an NIH Big Data to Knowledge (BD2K) Center. She currently directs the CMU Musculoskeletal Biomechanics Lab, an interdisciplinary group of mechanical engineers, bioengineers, and computer scientists seeking to advance precision rehabilitation in orthopaedics. Their experimental, computational, and clinical research, primarily on osteoarthritis, integrates wearable technologies, medical imaging, and machine learning.
Congratulations, Dr. Halilaj!
Dr. Stephen Badylak Extracellular Matrix Work Receives Two More U.S. Patents
McGowan Institute for Regenerative Medicine deputy director Stephen Badylak, DVM, PhD, MD, professor of surgery at the University of Pittsburgh, is an inventor of 71 U.S. patents and over 300 patents worldwide. In August 2020, Dr. Badylak received two new patents relative to his work with extracellular matrix (ECM). They are:
Non-Gelling Soluble Extracellular Matrix with Biological Activity, U.S. Patent No. 10736991 (issued on August 11, 2020)
Soluble forms of ECM, which form a hydrogel at body temperature, are increasingly used in anatomic locations that do not permit the use of an ECM scaffold. Similar to ECM scaffolds, ECM hydrogels have biological effects that include immune modulation and recruitment of stem cells, among others. However, all therapeutics applications do not require a hydrogel, and a non-gelling soluble form of ECM may be preferred in many instances. This patent describes methods of preparation and the biologic effects of a non-gelling soluble form of ECM.
Extracellular Matrix-Derived Gels and Related Methods, U.S. Patent No. 10729813 (issued on August 4, 2020)
This application was originally filed in 2012 and has since branched into a family of U.S. and international patents. This patent describes an acidic solution comprised of an acid protease and solubilized, non-dialyzed, non-crosslinked ECM (from a tissue selected from the group consisting of heart, pancreas, liver, ovary, spleen, and urinary bladder removed of submucosa) that, when neutralized, forms a gel at a temperature greater than 25oC.
These latest two patents join Dr. Badylak’s other most recently received patents:
- Extracellular Matrix Mesh Coating, U.S. Patent No. 10286119 (issued on May 14, 2019)
- Methods for Preparation of a Terminally Sterilized Hydrogel Derived from Extracellular Matrix, U.S. Patent No. 10213526 (issued on February 26, 2019)
- Biohybrid Composite Scaffold, U.S. Patent No. 10092676 (issued on October 9, 2018)
Dr. Badylak was inducted into the National Academy of Inventors as a Fellow in 2018.
Congratulations, Dr. Badylak!
Illustration: Image of solubilized dermal ECM, manufactured under cGMP conditions, that is easily injectable and forms a gel state at body temperature. The Badylak Laboratory.
Fellows Elected IAMBE Class 2020

The International Academy of Medical and Biological Engineering (IAMBE) recently announced that 28 distinguished colleagues have been elected to its 2020 Class of Fellows. Two of these notable honorees are McGowan Institute for Regenerative Medicine affiliated faculty members: Sanjeev Shroff, PhD, and Yu-li Wang, PhD.
The election to the Academy represents a highly selective process. The IAMBE Fellows are nominated by current Fellows (~150 individuals), evaluated by the Membership Committee, and elected by all active Fellows. The election to the Academy is in recognition of a colleague’s distinguished contributions to and leadership in the field of medical and biological engineering in an international level.
Dr. Shroff is the Distinguished Professor of and Gerald E. McGinnis Chair in the Department of Bioengineering, Swanson School of Engineering and Professor of Medicine in the School of Medicine at the University of Pittsburgh. He is also the Chair of the Department of Bioengineering. Prior to joining the University of Pittsburgh in April 2000, Dr. Shroff was a faculty member at the University of Chicago in the Department of Medicine (Cardiology Section). Trained as an electrical engineer (B.Tech., Indian Institute of Technology, Kanpur, India and M.Eng., McMaster University, Canada), Dr. Shroff obtained his doctoral degree in Bioengineering from the University of Pennsylvania.
Dr. Shroff’s research is in the cardiovascular arena, with two main focus areas: (1) Contractile and regulatory proteins and post-translational regulation of cardiac contraction. (2) Role of vascular stiffness in cardiovascular function and potential therapeutic applications of vascular stiffness-modifying drugs and/or hormones (e.g., relaxin). His research efforts have been supported by numerous grants from NIH (continuous funding since 1986), AHA, NSF, and industry partners. He was the recipient of the Established Investigator Award from the AHA (1986-1991) and was elected as a Fellow of the American Physiological Society (1988), Fellow of the American Institute for Medical and Biological Engineering (1999), and Fellow of Biomedical Engineering Society (2007). Recognized by his colleagues and peers as a consummate teacher and mentor, Dr. Shroff received the Carnegie Science Center Award for Excellence (University Educator) in 2007, the Swanson School of Engineering’s Outstanding Educator Award in 2010, and University of Pittsburgh Chancellor’s Distinguished Teaching Award in 2011. He has mentored 37 students (15 post-doctoral and 22 graduate/pre-doctoral), most of whom are pursuing independent research careers in academia or industry. Dr. Shroff has been serving as the Principal Investigator on a NIH-NHLBI pre-doctoral T32 training grant (Cardiovascular Bioengineering Training Program) since 2005 and on the Coulter Translational Research Partnership II grant since 2013. In 2012 Dr. Shroff was named the Distinguished Professor at the University of Pittsburgh, a designation that constitutes the highest honor that the University can accord a member of the professoriate.
Dr. Wang is the R. Mehrabian Professor of Biomedical Engineering at Carnegie Mellon University. Prior to this he was a Professor in the Departments of Physiology, Cell Biology, and Biomedical Engineering and Medical Physics at the University of Massachusetts Medical School. Dr. Wang received his BS in Physics from National Taiwan University in Taipei. He earned his PhD in Biophysics from Harvard University, where he then served as a postdoctoral associate.
Dr. Wang is a member of the American Society for Cell Biology, the Biophysical Society, and the Biomedical Engineering Society. He is an Associate Editor of Molecular Biology of the Cell and Cellular & Molecular Bioengineering, and a Fellow of the American Institute for Medical and Biological Engineering.
Dr. Wang’s research areas include mechanisms of cell division and migration, mechanics of cell-matrix interactions, cellular responses to mechanical forces, mechanotransduction, live cell imaging, and micromanipulation.
The new Fellow Induction will be held in a Virtual Induction Ceremony, in conjunction with Carnegie Mellon Forum on Biomedical Engineering to be held on September 18, 2020.
Congratulations, Drs. Shroff and Wang!
Top Dentists Honored in Pittsburgh Magazine

“If you had a patient in need of a dentist, which dentist would you refer them to?”
This is the question asked to thousands of dentists to help determine who the topDentists should be. Dentists and specialists are asked to take into consideration years of experience, continuing education, manner with patients, use of new techniques and technologies, and of course physical results. Pittsburgh Magazine’s annual list, which in 2020 contains 414 dentists across 10 specialties, includes three McGowan Institute for Regenerative Medicine affiliated faculty members:
Endodontics
- Herbert Ray, Jr., DMD, Associate Professor of Endodontics and the Department Chair and Director, Graduate Endodontic Residency Program, University of Pittsburgh School of Dental Medicine
Oral and Maxillofacial Surgery
- William Chung, MD, DDS, Professor in the Department of Oral and Maxillofacial Surgery, School of Dental Medicine, University of Pittsburgh
- Bernard Costello, MD, DMD, FACS, Dean of the University of Pittsburgh School of Dental Medicine, Professor of Oral and Maxillofacial Surgery, Chief of Pediatric Oral and Maxillofacial Surgery at the Children’s Hospital of Pittsburgh, and a surgeon with the Cleft and Craniofacial Center
The nomination pool of dentists consists of dentists listed online with the American Dental Association as well as all dentists listed online with their local dental societies, thus allowing virtually every dentist the opportunity to participate. Dentists are also given the opportunity to nominate other dentists that they feel should be included in the list.
topDentists was started with the intent to identify the best dentists and specialists in the country and is the only list of its kind, chosen by the dental profession themselves. topDentists has over fifty years in combined experience compiling peer-review referral guides in the legal, dental, and medical fields. Using this experience along with the input of several prominent dentists in the United States, topDentists has created a selection process that has been refined and improved over previous superlative guides. Because dentists must be voted in strictly by their peers, and virtually every dentist is given an opportunity to participate, and listings cannot be purchased (and no payment is required to be listed), inclusion in topDentists is considered a distinct honor.
Congratulations, all!
Dr. Michael Pinsky Receives 2020 ATS Assembly on CC Annual Lifetime Achievement Award

McGowan Institute for Regenerative Medicine affiliated faculty member Michael Pinsky, MD, Professor and Vice Chair Emeritus of the Department of Critical Care Medicine, University of Pittsburgh, has been selected to receive the 2020 American Thoracic Society Assembly on Critical Care Annual Lifetime Achievement Award. The award is given in recognition and appreciation of Dr. Pinsky’s career dedicated to research and teaching of the science and practice of critical care medicine. He will receive his award during the Critical Care Assembly virtual membership meeting to be held August 5-10, 2020.
“The Lifetime Achievement Award is a wonderful and fitting way for Michael’s peers to recognize the impact of his 40-year career devoted to academic critical care medicine,” said Douglas White, MD, MAS, who is Vice Chair for Faculty Development in the Department of Critical Care Medicine.
Dr. Pinsky’s research focuses on the effects of ventilation on cardiovascular function, functional hemodynamic monitoring as a precision diagnostic tool for cardiorespiratory insufficiency, left ventricular pump function and dysfunction in the setting of regional wall motion abnormalities and, more recently, machine learning to glean knowledge from data in defining health and disease. He is also recognized internationally as the co-editor of Hemodynamic Monitoring, the definitive textbook on major principles in managing critical care patients.
He holds many roles across the Pitt campus, including director of the Cardiopulmonary Research Laboratory, senior advisor for the Center for Military Medicine Research, and a faculty member of the Center for Critical Care Nephrology. Among his many honors, Dr. Pinsky is a fellow of the American Physiological Society, Docteur honoris causa from the University of Paris V (2002), a Master of the College of Critical Care Medicine (2011), and he received the SMART Award for Distinguished Achievement in Critical Care (2014).
Congratulations, Dr. Pinsky!
Dr. Derek Angus Appointed UPMC’s Chief Healthcare Innovation Officer

McGowan Institute for Regenerative Medicine affiliated faculty member Derek Angus, MD, MPH, has been named the UPMC chief healthcare innovation officer. His appointment comes at a time when healthcare is undergoing unparalleled change. The acceleration of biomedical discovery, together with advances in technology and data science, is spurring revolutionary changes in the entire relationship a person has with her health and wellness.
As an organization committed to healthcare transformation, UPMC seeks to simultaneously provide the best care today and learn how to provide better care tomorrow. Dr. Angus will lead UPMC’s efforts to ensure patients and doctors make every care decision as wisely as possible, while also learning as fast as possible such that tomorrow’s care arrives sooner.
The way forward requires fusion of many different disciplines and skills, blending expertise in clinical care delivery with medical research, artificial intelligence and computer science, and social and behavioral sciences. Dr. Angus is ideally positioned to lead this effort. Dr. Angus is an internationally renowned and highly prolific scientist who has developed and led many successful multidisciplinary collaborations of basic scientists, clinicians, data scientists, economists, and behavioral and social scientists.
In recent years, he has focused on approaches that facilitate smarter decision-making and faster learning in healthcare, including novel Bayesian adaptive trial designs, the application of machine learning to large-scale data, and use of behavioral economics and decision psychology to support optimal decision-making. Dr. Angus’ interests dovetail with the applied machine learning work that has been spearheaded to date by the clinical analytics group and will help advance this work to the next level.
Also, Dr. Angus will work closely with Oscar Marroquin, MD, UPMC’s newly appointed chief healthcare data and analytics officer. In addition, Drs. Angus and Marroquin will partner with the Wolff Center to support UPMC’s commitment to the learning health system. Dr. Angus will also work closely with the Health Plan’s Center for High Value Health, and with executive leadership across UPMC’s Health Services, Health Plan and Enterprise Divisions to coordinate cross-division digital and clinical innovation, such as the Learning While Doing program, that will accelerate UPMC’s capacity to bring life-changing medicine.
In addition to his chief healthcare innovation officer role, Dr. Angus will continue to serve as chair of the UPP Department of Critical Care Medicine and physician director of the UPMC ICU Service Center. He also holds the rank of Distinguished Professor and the Mitchell P. Fink Endowed Chair in Critical Care Medicine at the University of Pittsburgh School of Medicine.
Dr. Angus completed medical school and internal medicine training at the University of Glasgow and affiliated teaching hospitals. He completed a fellowship in critical care medicine at the University of Pittsburgh and attained a Master of Public Health in Health Services Administration. He is a Fellow of the Royal College of Physicians of the United Kingdom, a member of the American Association of Physicians, and has received multiple national and international honors.
Congratulations, Dr. Angus!
Dr. Stefanie Sydlik Joins World Economic Forum’s Community of Young Scientists

McGowan Institute for Regenerative Medicine affiliated faculty member Stefanie Sydlik, PhD, has been selected by the World Economic Forum (WEF) as part of the 2020 Community of Young Scientists.
Dr. Sydlik, an assistant professor of chemistry at Carnegie Mellon University (CMU), joins the forum’s Young Scientists community, which recognizes scientific rising stars under the age of 40 who are pursuing high-impact research.
Created in 2008, the Young Scientists community engages researchers from a variety of disciplines and geographies to integrate scientific knowledge into society for the public good.
Dr. Sydlik, a 2007 Mellon College of Science (MCS) graduate, synthesizes innovative new polymers for biological uses. By modifying graphite with calcium phosphate, for example, her lab has been able to turn the material found in pencil lead into a scaffold that can be used to help heal traumatic bone injuries and then naturally degrade away. In a similar vein, her lab has also worked to develop additives for medical adhesives that can allow those adhesives to carry small-molecule drugs such as analgesics for pain or anti-inflammatories.
“I’m really excited to be involved in this global network and to be in a group where we can talk about the big problems and affect policy going forward,” Dr. Sydlik said of the honor. In particular, she said she’s hoping to engage with non-scientists from around the world and learn about the problems they hope scientists will work to solve.
One particular issue Dr. Sydlik is interested in tackling is microplastics, minute fragments of plastic that can accumulate in the environment and affect animals and even humans.
Dr. Sydlik joined the faculty at CMU in 2015 after completing a postdoctoral fellowship in chemical engineering and a PhD in organic chemistry at Massachusetts Institute of Technology. As an MCS undergraduate, she was selected as a Beckman Scholar in 2006 and received the 2007 Judith A. Resnik Award.
Congratulations, Dr. Sydlik!
Nindl Lab Student Receives the 2020 Colonel Mary Lipscomb Hamrick Army Medical Specialist Corps Manuscript Award

Major Will Conkright, currently enrolled in the Doctor of Philosophy in Rehabilitation Science program at the University of Pittsburgh’s School of Health and Rehabilitation Sciences, was the recipient of the 2020 Colonel Mary Lipscomb Hamrick Army Medical Specialist Corps Manuscript Award for his publication titled “Differential Recovery Rates of Fitness Following U.S. Army Ranger Training.” The article was published in the May 2020 issue of the Journal of Science and Medicine in Sports. Major Conkright’s main academic advisor is McGowan Institute for Regenerative Medicine affiliated faculty member Brad Nindl, PhD (COL, USAR), who is the director of the University of Pittsburgh Neuromuscular Research Lab/Warrior Human Performance Research Center.
Selection for this award is highly competitive and is given to researchers demonstrating excellence in publishing peer-reviewed literature with profound relevance and impact to the military. Major Conkright is an active duty Army officer and is currently engaged in his doctoral studies at the NMRL/WHPRC. He was presented this award via a virtual awards presentation hosted by Colonel John Balser (SP Corps Chief).
Illustration: University of Pittsburgh School of Health and Rehabilitation Sciences.
Regenerative Medicine Podcast Update
The Regenerative Medicine Podcasts remain a popular web destination. Informative and entertaining, these are the most recent interviews:
#212 –– Drs. Macalus Hogan and Jeanne Doperak discuss guidelines to assist coaches, athletic trainers, and organizers with creating a safe environment for youth athletes, fans, and staff amidst CoVid-19.
Visit www.regenerativemedicinetoday.com to keep abreast of the new interviews.
PUBLICATION OF THE MONTH
Author: Jonas Wilbs, Xu-Dong Kong, Simon J Middendorp, Raja Prince, Alida Cooke, Caitlin T Demarest, Mai M Abdelhafez, Kalliope Roberts, Nao Umei, Patrick Gonschorek, Christina Lamers, Kaycie Deyle, Robert Rieben, Keith E Cook, Anne Angelillo-Scherrer, Christian Heinis
Title: Cyclic peptide FXII inhibitor provides safe anticoagulation in a thrombosis model and in artificial lungs
Summary: Inhibiting thrombosis without generating bleeding risks is a major challenge in medicine. A promising solution may be the inhibition of coagulation factor XII (FXII), because its knock-out or inhibition in animals reduced thrombosis without causing abnormal bleeding. Herein, we have engineered a macrocyclic peptide inhibitor of activated FXII (FXIIa) with sub-nanomolar activity (Ki = 370 ± 40 pM) and a high stability (t1/2 > 5 days in plasma), allowing for the preclinical evaluation of a first synthetic FXIIa inhibitor. This 1899 Da molecule, termed FXII900, efficiently blocks FXIIa in mice, rabbits, and pigs. We found that it reduces ferric-chloride-induced experimental thrombosis in mice and suppresses blood coagulation in an extracorporeal membrane oxygenation (ECMO) setting in rabbits, all without increasing the bleeding risk. This shows that FXIIa activity is controllable in vivo with a synthetic inhibitor, and that the inhibitor FXII900 is a promising candidate for safe thromboprotection in acute medical conditions.
Source: Nat Commun. 2020 Aug 4;11(1):3890.
GRANT OF THE MONTH
PI: George Gittes
Title: Alpha Cells Conversion to Beta Cells in Non-Human Primates
Description: An ideal solution to the treatment or cure of type 1 diabetes mellitus would be the formation of new functioning β-cells from the patient’s own tissues that are not attacked by the autoimmunity, thereby avoiding the need for any immunosuppression. Abundant recent data have suggested that α-cells are a viable potential source for endogenous transdifferentiation into β-cells. Here, we describe a pancreatic intraductal viral delivery system in the mouse, wherein a single infusion of an adeno-associated virus (AAV), carrying a pdx1/mafA expression vector, is given to a toxin-induced (alloxan) diabetic mouse. This AAV gene therapy induced robust and durable α-cell transdifferentiation into β-cell-like cells through neogenesis, with recovery of over 60% of the β-cell mass within 4 weeks, and with persistent, durable euglycemia. Serendipitously, when this β-cell-like cell neogenesis was similarly induced in the autoimmune NOD mouse model, the mice became euglycemic for 4 months or more, without any additional therapy or immunosuppression. To our knowledge, no clinically applicable β-cell replacement therapy in NOD mice has been successful without immunosuppression. We suspect that the neogenic β-cell-like cells may not be attacked by the autoimmunity because they are “imperfect” β-cells by RNA-seq analysis. Since pancreatic duct injection is routinely performed in humans as a relatively simple, non-surgical procedure, and since numerous viral gene therapy trials are currently ongoing for several diseases, we feel that our approach may be rapidly translatable to humans with type 1 diabetes mellitus. In this proposal, we will perform important proof-of-principle studies in non-human primates as last steps in preparation for human gene therapy clinical trials. The primate pancreas has a very different texture and consistency than the mouse pancreas (and is very similar to the human pancreas). Thus, the mechanics of the viral delivery will likely require substantial alterations. In addition, a glucagon promoter is preferable to the CMV promoter for expression of pdx1 and mafA, so we will strive to develop and optimize a glucagon promoter vector that is effective in primates. Further studies will investigate this pancreatic ductal infusion approach in the context of AAV neutralizing antibodies. We will also perform detailed analyses of the new β-cell-like cells, including physiology, gene expression phenotype, and anatomy. In summary, we feel that the proposed studies, if successful, should position us well in preparation for clinical trials in humans with type 1 diabetes mellitus.
Source: NIDDK
Amount: $2.59 Million
