October 2014 | VOL. 13, NO. 10 | www.McGowan.pitt.edu
Needle Treatment for Glaucoma Shows Promise: A Monthly Injection That Might Replace Eye Drops Used Twice Daily
 As reported by Ann Lukits of the Wall Street Journal, Pittsburgh scientists led by McGowan Institute for Regenerative Medicine faculty member Steven Little, PhD, chairman of the Department of Chemical and Petroleum Engineering and associate professor and CNG faculty fellow in the Departments of Chemical and Petroleum Engineering, Bioengineering, Immunology, and Ophthalmology, are working to develop new techniques to administer medication for glaucoma patients that replace the usual regimen of twice-daily eye drops, a sometimes unreliable treatment. McGowan Institute for Regenerative Medicine faculty member Joel Schuman, MD, Eye and Ear Foundation professor and chairman of the Department of Ophthalmology, with secondary appointments as professor of bioengineering and of clinical and translational science, and professor at the Center for the Neural Basis of Cognition, a University of Pittsburgh/Carnegie Mellon University collaboration, was a member of the research team as well.
As reported by Ann Lukits of the Wall Street Journal, Pittsburgh scientists led by McGowan Institute for Regenerative Medicine faculty member Steven Little, PhD, chairman of the Department of Chemical and Petroleum Engineering and associate professor and CNG faculty fellow in the Departments of Chemical and Petroleum Engineering, Bioengineering, Immunology, and Ophthalmology, are working to develop new techniques to administer medication for glaucoma patients that replace the usual regimen of twice-daily eye drops, a sometimes unreliable treatment. McGowan Institute for Regenerative Medicine faculty member Joel Schuman, MD, Eye and Ear Foundation professor and chairman of the Department of Ophthalmology, with secondary appointments as professor of bioengineering and of clinical and translational science, and professor at the Center for the Neural Basis of Cognition, a University of Pittsburgh/Carnegie Mellon University collaboration, was a member of the research team as well.
A promising approach, reported in the August issue of Experimental Eye Research, involves a monthly injection of a slow-release drug directly into the eye. The technique, tested on laboratory animals, uses significantly less medication than current treatment and potentially manages the disease better, the researchers said. One injection was comparable to taking eye drops twice a day for 28 days.
Glaucoma, in which eye pressure damages the optic nerve, affects an estimated 2.7 million Americans and puts them at risk for vision loss, according to the National Eye Institute. Treatment usually involves taking eye drops multiple times a day to maintain a healthy eye pressure, but studies have shown as many as 80% of patients forget to take the drops, the researchers said. The drops can also leach into the opposite eye, causing retinal hemorrhages and other complications.
A 28-day pilot study at the University of Pittsburgh tested the injection technique on rabbits with normal eye pressure. One group of animals was injected with a biodegradable, spherical-shaped capsule called a microsphere just below the conjunctiva, or thin membrane covering the white part of the eye. The microsphere contained less than three drops of brimonidine tartrate, a glaucoma medication. Another group was injected with blank microspheres and a third received two brimonidine drops a day in the traditional way.
On average, eye pressure dropped 21.4% in rabbits injected with the slow-release drug and 19.9% in rabbits treated with eye drops. There was no significant change in eye pressure in the animals injected with blank microspheres. The microspheres didn’t irritate other eye tissues and had degraded by day 35, the researchers said.
The Pittsburgh team is testing a topical solution that would enable patients to receive the same month-long treatment without the insertion of a drug-delivery system, said Dr. Little.
To date, the microsphere drug-delivery system hasn’t been tested on humans. Also, the effects of repeated injections aren’t known. More research is necessary before this potential technology may be available to patients.
Upcoming Events
Computational Modeling: From Clinical Needs to Bench Results
 December 15, 2014; 8 AM – 4 PM
December 15, 2014; 8 AM – 4 PM
Room S100-Biomedical Science Tower
Yoram Vodovotz, PhD
The workshop is designed to bring together leading local academic physicians, translational researchers, and members of industry in order to share advances in the clinically-driven applications of computational modeling. Participants will be exposed to cutting-edge presentations regarding advances in computational modeling in biomarker discovery, personalized diagnostics, and drug design.
The program includes:
- Morning Keynote: Translational Systems Biology: Roadmap for the Future of Biomedical Research
- Session I: An Overview of Clinical Questions and Computational Successes
- Session II: Computational Modeling for Pre-Clinical Targets
- Session III: Interdisciplinary collaborations and practical considerations
- Afternoon Keynote: Computational biology and personalized medicine at Pitt and UPMC
Seating is LIMITED; please register HERE to reserve your place for this exciting program.
Special Presentation: Department of Defense and Federal Funding Opportunities for Academic Health Centers
 Ronald Poropatich, MD, Executive Director of the Center for Military Medicine Research, will give a presentation that will provide an overview of the Pitt Center for Military Medicine Research and the approach to applying for DoD funding. Opportunities for McGowan Institute affiliated faculty will be identified. An overview of the difference between NIH and DoD medical research will be presented as well as a description of the military medical research needs. Examples will be cited of successful Pitt research that is funded by the DoD.
Ronald Poropatich, MD, Executive Director of the Center for Military Medicine Research, will give a presentation that will provide an overview of the Pitt Center for Military Medicine Research and the approach to applying for DoD funding. Opportunities for McGowan Institute affiliated faculty will be identified. An overview of the difference between NIH and DoD medical research will be presented as well as a description of the military medical research needs. Examples will be cited of successful Pitt research that is funded by the DoD.
This presentation will be held November 13, 2014 at 9:30 AM in room 1105ABC of Scaife Hall.
RESOURCES AT THE MCGOWAN INSTITUTE
November Special at the Histology Lab
Hematoxylin and Eosin Stain
Hematoxylin is a natural compound extracted from a species of tree found in Mexico and the West Indies. The extracted compound is then oxidized to produce hematein, which is the active staining component of the hematoxylin stain. Hematoxylin stains must therefore be ‘ripened’ by oxidation before they can be used. Hematoxylin staining requires the use of a mordant (most commonly aluminum salts) and stains the nuclear components of cells a dark blue. Hematoxylin is used in combination with eosin because eosin stains the cytoplasmic organelles varying shades of pink, red or orange. The combination of the two stains provides a broad range of morphological information about the section.
The McGowan Institute uses an automated staining machine for all of the H&E staining requests, guaranteeing uniform and reproducible results. The more H&Es you submit to the Histology Core Laboratory the less you will pay for each one.
All of November, the Histology Lab will take an additional 30% off our regularly priced H&E staining when mentioning this ad. You will find quality work at fair prices at the MIRM Histology Core. Take advantage of November specials and get to know what our staff can offer your research team.
Call Lori at (412) 624-5265 or email: perezl@upmc.edu to ask about automated staining, special stains, or immunohistochemistry.
SCIENTIFIC ADVANCES
Pittsburgh Pediatric Mechanical Cardiopulmonary Support Reaches to Florida
 McGowan Institute for Regenerative Medicine faculty member Peter Wearden, MD, PhD, surgical director of Pediatric Heart and Lung Transplantation and director of the Pediatric Mechanical Cardiopulmonary Support Program of the Children’s Hospital of Pittsburgh of UPMC, assistant professor of cardiothoracic surgery at the University of Pittsburgh School of Medicine, and deputy director of pediatrics at the McGowan Institute, is member of a team of medical professionals at Children’s Hospital who are using advanced and innovative technologies to perform life-saving procedures on children in need of cardiac support. Dr. Wearden is the leader in the field of pediatric mechanical cardiopulmonary support and receives numerous clinical patient cases from doctors and hospitals outside the Pittsburgh area. He is widely recognized for his clinical innovations where he has adapted existing technologies and devices to address the needs of these tiny patients.
McGowan Institute for Regenerative Medicine faculty member Peter Wearden, MD, PhD, surgical director of Pediatric Heart and Lung Transplantation and director of the Pediatric Mechanical Cardiopulmonary Support Program of the Children’s Hospital of Pittsburgh of UPMC, assistant professor of cardiothoracic surgery at the University of Pittsburgh School of Medicine, and deputy director of pediatrics at the McGowan Institute, is member of a team of medical professionals at Children’s Hospital who are using advanced and innovative technologies to perform life-saving procedures on children in need of cardiac support. Dr. Wearden is the leader in the field of pediatric mechanical cardiopulmonary support and receives numerous clinical patient cases from doctors and hospitals outside the Pittsburgh area. He is widely recognized for his clinical innovations where he has adapted existing technologies and devices to address the needs of these tiny patients.
In adults, when supplemental cardiac support is needed as an interim measure, surgeons may elect to implant an artificial heart pump. The pumps that work for adults are not suitable for infants and children. On numerous occasions Dr. Wearden has successfully adapted a pediatric heart pump, the Berlin Heart, which underwent a U.S. Food and Drug Administration (FDA) evaluation trial at Children’s and 14 other hospitals nationwide.
The Berlin Heart is an extracorporeal artificial heart pump—or ventricular assist device (VAD)—that keeps children with life-threatening heart failure alive while awaiting a heart transplant. The device has also been used effectively as a bridge to recovery. Mark Roth, Pittsburgh Post-Gazette, reported the story of 2-year-old Ethan Gradowski who was placed on the heart transplant list in early November 2008 due to narrowing of his arteries as a result of acute inflammation or a reaction to medications following a routine repair of an atrial septal defect (a hole in the wall that divides the heart’s two upper chambers). He was placed on the Berlin Heart (then an experimental device) shortly thereafter to wait for a donor heart. As Ethan waited and with the help of the Berlin Heart his tiny body slowly began to heal itself. The Berlin Heart gave his arteries the rest they needed to recover, and in late December 2008 Ethan was successfully weaned from the device.
Announced recently, a partnership effort between St. Joseph’s Children’s Hospital in Florida and the Children’s Hospital of Pittsburgh of UPMC has been established to provide highly specialized cardiovascular care. This would include patients ranging from babies in the womb to adults with congenital heart disease. At St. Joseph’s Heart Institute, a new 35,000-square-foot, state-of-the-art facility is located on the hospital campus in Tampa. In addition to collaborating with St. Joseph’s Children’s Hospital’s multidisciplinary cardiac team on surgical and non-invasive cardiology services, Children’s Hospital of Pittsburgh of UPMC’s experts will provide additional support to patients, families, and caregivers in St. Joseph’s Children’s Hospital’s cardiac intensive care unit via telemedicine.
The MCI group was more likely to have differences in the biologic activity of 24 proteins that are involved in the regulation of immune and inflammatory pathways, intracellular signaling, cell survival, and protein and lipid balance.
Illustration: Dr. Peter Wearden examines 3-year-old congenital heart patient Natali Perez at St. Joseph’s Children’s Hospital in Tampa. (PRNewsFoto/St. Joseph’s Children’s Hospital)
Dr. John Kellum Helps Develop New Kidney Test
 McGowan Institute for Regenerative Medicine affiliated faculty member John Kellum, MD, professor of critical care medicine at the University of Pittsburgh, transplant physician in anesthesiology at the Thomas E. Starzl Transplantation Institute, and co-director at the Mechanisms and Novel Therapies for Resuscitation and Acute Illness (MANTRA) Lab, was the lead investigator of a now FDA-approved lab test that will be used to help determine if critically ill hospitalized patients are at risk of developing moderate to severe acute kidney injury (AKI) in the 12 hours following the administration of the test. Early knowledge that a patient is likely to develop AKI may prompt closer patient monitoring and help prevent permanent kidney damage or death.
McGowan Institute for Regenerative Medicine affiliated faculty member John Kellum, MD, professor of critical care medicine at the University of Pittsburgh, transplant physician in anesthesiology at the Thomas E. Starzl Transplantation Institute, and co-director at the Mechanisms and Novel Therapies for Resuscitation and Acute Illness (MANTRA) Lab, was the lead investigator of a now FDA-approved lab test that will be used to help determine if critically ill hospitalized patients are at risk of developing moderate to severe acute kidney injury (AKI) in the 12 hours following the administration of the test. Early knowledge that a patient is likely to develop AKI may prompt closer patient monitoring and help prevent permanent kidney damage or death.
“I’m delighted at the emergence of new tools that may help improve care for patients at risk for AKI,” said Dr. Kellum. “While more physicians are aware of the need to assess patient risk for AKI, we have lacked a reliable method of performing this risk assessment.”
The first biomarker-based immunoassay, called NephroCheck, was developed by San Diego-based Astute Medical Inc. The 510(k) clearance through the FDA clears the path to commence sales in the coming weeks with its partner Ortho-Clinical Diagnostics Inc., whom Astute Medical has designated as the exclusive sales agent for Astute’s NephroCheck Test.
“Astute is committed to developing innovative tests to provide better health care. We are excited to introduce the first real improvement in renal testing in over 60 years,” said Chris Hibberd, Astute Medical chief executive officer. “Together with our partner, Ortho Clinical Diagnostics, we look forward to making the NephroCheck Test System available. We thank our employees and clinical investigators who helped make this important advancement in AKI.”
Dr. Andrew Duncan Receives NIH Grant Focused on Obtaining a Better Understanding of Liver Biology
 Nearly 25 million Americans are affected by liver dysfunction, and liver diseases are the 10th leading cause of death in the US. There is a clear and urgent need for developing new alternatives to whole organ replacement. A better understanding of liver biology is required to improve existing approaches and to innovate therapies for the treatment of liver diseases, including viral hepatitis and steatohepatitis. McGowan Institute for Regenerative Medicine faculty member Andrew Duncan, PhD, assistant professor in the Department of Pathology at the University of Pittsburgh with a secondary appointment in the Department of Bioengineering of the Swanson School of Engineering, recently received a 5-year, $2,174,309 grant from the National Institute of Health’s National Institute of Diabetes and Digestive and Kidney Diseases. The project title is “Mechanisms of Polyploidy and Aneuploidy in the Liver.”
Nearly 25 million Americans are affected by liver dysfunction, and liver diseases are the 10th leading cause of death in the US. There is a clear and urgent need for developing new alternatives to whole organ replacement. A better understanding of liver biology is required to improve existing approaches and to innovate therapies for the treatment of liver diseases, including viral hepatitis and steatohepatitis. McGowan Institute for Regenerative Medicine faculty member Andrew Duncan, PhD, assistant professor in the Department of Pathology at the University of Pittsburgh with a secondary appointment in the Department of Bioengineering of the Swanson School of Engineering, recently received a 5-year, $2,174,309 grant from the National Institute of Health’s National Institute of Diabetes and Digestive and Kidney Diseases. The project title is “Mechanisms of Polyploidy and Aneuploidy in the Liver.”
Hepatocytes, the primary functional cell type in the liver, display a range of chromosomal diversity resulting from prevalent physiological polyploidy (>90% in mice and 50% in humans) and aneuploidy (60% in mice and 30-90% in humans). In eukaryotic organisms, cells usually contain a diploid genome comprised of pairs of homologous chromosomes.
Polyploidy refers to gains in entire sets of chromosomes, and aneuploidy refers to gains and losses of individual chromosomes. The roles of hepatic polyploidy and aneuploidy represent a major gap in our current understanding of liver biology. Dr. Duncan and the team recently found that aneuploidy enhances the regenerative capacity of the mouse liver. In response to Tyrosinemia-induced injury, that is normally toxic to the liver, they identified a subset of aneuploid hepatocytes that was resistant to the disease. The data suggest that aneuploid hepatocytes are endowed with enhanced capacity for adaptation and regeneration.
Dr. Duncan’s central hypothesis is that aneuploidy functions as an adaptive mechanism in response to hepatic injury. The goals of his project are to identify mechanisms regulating hepatic aneuploidy/polyploidy and to unravel how aneuploidy affects liver function. To investigate these questions, he and the team propose to determine whether polyploid hepatocytes are necessary for development of aneuploid livers. Experiments will characterize hepatic cell divisions, karyotypes, and stress response in E2f7/E2f8 knockout mice, which have normal liver function but are depleted of polyploid hepatocytes. Next, the researchers will dissect the role of a novel regulator of hepatic polyploidy, recently identified in Dr. Duncan’s laboratory, microRNA-122 (miR-122). Experiments will determine how miR-122 alters ploidy and aneuploidy throughout life. The scientists will also identify cellular and molecular mechanisms by which miR-122 regulates hepatic ploidy. Finally, the team will determine how random karyotypes (in aneuploid hepatocytes) affect function in the liver. They will utilize a novel xenotransplantation model to examine clonal nodules of regenerating human hepatocytes. Experiments will measure aneuploidy and determine gene expression profiles in these nodules. Together, these studies will define the extent to which aneuploidy affects liver repair/regeneration as well as the molecular mechanisms that control this process. Understanding how aneuploid hepatocytes arise and function will provide new and crucial insights into liver homeostasis, diseases, and treatments.
Genetic Discovery Yields Prostate Cancer Test, Promise of Future Therapy
 A genetic discovery out of the University of Pittsburgh School of Medicine is leading to a highly accurate test for aggressive prostate cancer and identifies new avenues for treatment. The analysis, published in the American Journal of Pathology, found that prostate cancer patients who carry certain genetic mutations have a 91 percent chance of their cancer recurring. McGowan Institute for Regenerative Medicine affiliated faculty member George Michalopoulos, MD, PhD, professor and chairman of the Department of Pathology at the University of Pittsburgh, was a member of the study’s research team. This research was funded by the National Institutes of Health (NIH), American Cancer Society, and University of Pittsburgh Cancer Institute (UPCI).
A genetic discovery out of the University of Pittsburgh School of Medicine is leading to a highly accurate test for aggressive prostate cancer and identifies new avenues for treatment. The analysis, published in the American Journal of Pathology, found that prostate cancer patients who carry certain genetic mutations have a 91 percent chance of their cancer recurring. McGowan Institute for Regenerative Medicine affiliated faculty member George Michalopoulos, MD, PhD, professor and chairman of the Department of Pathology at the University of Pittsburgh, was a member of the study’s research team. This research was funded by the National Institutes of Health (NIH), American Cancer Society, and University of Pittsburgh Cancer Institute (UPCI).
“Being able to say, with such certainty, that a patient is nearly guaranteed to see a recurrence of his prostate cancer means that doctors and patients can elect to be more aggressive in treating the cancer, knowing that the benefits likely outweigh the risks,” said Jian-Hua Luo, MD, PhD, professor of pathology, Pitt School of Medicine, and member of UPCI. “Eventually, this could lead to a cure for prostate cancer through genetic therapy. With this discovery, we’re at the tip of the iceberg in terms of possibilities for improving patient outcomes.”
Prostate cancer is the second most common cancer among men (behind skin cancer), with one in seven men diagnosed with prostate cancer in their lifetime. The American Cancer Society estimates that this year in the U.S., about 233,000 new cases of prostate cancer will be diagnosed, and 29,480 men will die of prostate cancer.
Despite the high incidence rate, only a fraction of men diagnosed with prostate cancer develop metastases, and even fewer die from the disease.
“In some cases, this can make the treatment more dangerous than the disease, so doctors need more accurate tests to tell them which patients would most benefit from aggressive therapies, such as surgery, radiation, and chemotherapy,” said Dr. Luo.
Dr. Luo and his team sequenced the entire genome of prostate tissue samples from five prostate cancer patients who experienced aggressive recurrence of their cancer and compared them to normal tissue samples from men without cancer.
In the patients with prostate cancer recurrence, they identified 76 genetic fusion transcripts, which are hybrid genes formed from two previously separate genes and often are associated with cancer. After further testing, eight of the genetic fusion transcripts were found to be strongly associated with prostate cancer.
The researchers then screened for the eight fusion transcripts in 127 samples from patients with aggressive prostate cancer recurrence, 106 samples from prostate cancer patients with no recurrence at least 5 years after surgery, and 46 samples from prostate cancer patients with no recurrence less than 5 years after surgery. The samples came from UPMC, Stanford University Medical Center, and University of Wisconsin Madison Medical Center.
In those samples, 91 percent with aggressive recurrence of their prostate cancer were positive for at least one of the fusion transcripts. Two of the fusion transcripts in particular were strongly associated with poor outcomes — none of the patients whose samples contained them survived to 5 years.
In contrast, 68 percent of patients whose samples did not contain at least one of the transcripts remained cancer-free.
Dr. Luo said if continued clinical trials of the test do well, it could be available to all prostate cancer patients in a few years.
$1.25 Million Received from Defense Department to Make Whole-Eye Transplantation a Reality
 University of Pittsburgh School of Medicine researchers co-led by McGowan Institute for Regenerative Medicine faculty members Vijay Gorantla, MD, PhD, associate professor of surgery in the Department of Plastic Surgery at the University of Pittsburgh and the administrative medical director of the Pittsburgh Reconstructive Transplant Program at UPMC, and Joel Schuman, MD, chair of the Department of Ophthalmology, Pitt School of Medicine, and director of the UPMC Eye Center, have been awarded $1.25 million from the U.S. Department of Defense (DOD) to fund two projects that aim to establish the groundwork for the nation’s first whole-eye transplantation program.
University of Pittsburgh School of Medicine researchers co-led by McGowan Institute for Regenerative Medicine faculty members Vijay Gorantla, MD, PhD, associate professor of surgery in the Department of Plastic Surgery at the University of Pittsburgh and the administrative medical director of the Pittsburgh Reconstructive Transplant Program at UPMC, and Joel Schuman, MD, chair of the Department of Ophthalmology, Pitt School of Medicine, and director of the UPMC Eye Center, have been awarded $1.25 million from the U.S. Department of Defense (DOD) to fund two projects that aim to establish the groundwork for the nation’s first whole-eye transplantation program.
Offered through the DOD’s Vision Research Program, the grants support conceptually innovative research that ultimately could lead to critical discoveries or major advancements. The Pitt researchers will lead a multidisciplinary consortium that includes clinicians and scientists from Harvard University and the University of California, San Diego.
Although corneal transplants are routinely performed today, whole-eye transplantation has remained an unrealized goal in vision restoration because of challenges related to immune rejection and reestablishing the connectivity of the optic nerve to the visual centers in the brain.
The Audacious Restorative Goals in Ocular Sciences (ARGOS) Consortium established at Pitt will be the first cross-disciplinary, systematic attempt to explore strategies to enable corneal regeneration, retinal cell survival, long-distance optic nerve regeneration with cortical integration, and whole-eyeball transplantation.
“Recent advances in our understanding of retinal ganglion cell survival and successes with optic nerve regeneration in experimental studies strengthen our hope that whole-eye transplantation is an audacious yet achievable goal,” said Dr. Gorantla. “Our experience with transplanting complex immunogenic tissues, such as the hand, will help us optimize treatments for rejection in eye transplants.”
“This is an aggressive program with very high-risk and high-reward scenarios. We’re excited to be leading the project and honored to be collaborating with global leaders in optic nerve regeneration,” said Dr. Schuman. “By solving one facet of the problem at a time, the long dreamed-of goal of whole-eye transplantation may be possible with the promise of a better life for millions of patients worldwide.”
Sub-awardees of this $1 million award include Jeffrey Goldberg, MD, of the University of California, San Diego; and Larry Benowitz, PhD, of Harvard University.
In a related project led by principal investigator Kia Washington, MD, assistant professor of plastic surgery, University of Pittsburgh School of Medicine, the DOD granted $250,000 to focus on establishing baseline viability and structural integrity in an animal model of whole-eye transplantation. The researchers will examine immune rejection and evaluate the usage of extracellular matrix therapy for improvement of optic nerve function after whole-eye transplantation.
“We have successfully performed an eye transplant in a small animal model,” said Dr. Washington. “This ongoing project may eventually lead to restoration of vision after trauma or degenerative disease.”
The Louis J. Fox Center for Vision Restoration will provide additional key funding for the whole-eye transplantation effort.
Illustration: Randall Mackenzie Illustrations.
Pitt Gets $11 Million from NIH to Lead Center of Excellence in National Big Data Research Consortium
 The National Institutes of Health has awarded the University of Pittsburgh an $11 million, 4-year grant to lead a Big Data to Knowledge Center of Excellence, an initiative that will help scientists capitalize more fully on large amounts of available data and to make data science a more prominent component of biomedical research. McGowan Institute for Regenerative Medicine affiliated faculty member Ivet Bahar, PhD, Distinguished Professor and JK Vries Chair, Department of Computational and Systems Biology, Pitt School of Medicine, is a co-director of the new Center for Causal Modeling and Discovery which will be part of an elite national team addressing the challenges of Big Data in biomedicine.
The National Institutes of Health has awarded the University of Pittsburgh an $11 million, 4-year grant to lead a Big Data to Knowledge Center of Excellence, an initiative that will help scientists capitalize more fully on large amounts of available data and to make data science a more prominent component of biomedical research. McGowan Institute for Regenerative Medicine affiliated faculty member Ivet Bahar, PhD, Distinguished Professor and JK Vries Chair, Department of Computational and Systems Biology, Pitt School of Medicine, is a co-director of the new Center for Causal Modeling and Discovery which will be part of an elite national team addressing the challenges of Big Data in biomedicine.
“As part of a national consortium, this Center of Excellence will put Pitt on the map as a home of Big Data science,” said Arthur S. Levine, MD, senior vice chancellor for the health sciences and John and Gertrude Petersen Dean of the School of Medicine. “Our strengths in this field have stimulated collaborations with leading institutions, including Harvard and Stanford, and now we will be able to further develop such partnerships in many more meaningful ways.”
Much of science focuses on understanding the “why” or “how” in nature, and now the challenge is to find these answers within terabytes and petabytes of data, or what is now known as “Big Data,” said Gregory Cooper, MD, PhD, professor and vice chair of the Department of Biomedical Informatics, Pitt School of Medicine and co-director of the new Center.
A collaboration of researchers at Pitt, Carnegie Mellon University (CMU), the Pittsburgh Supercomputing Center, and Yale University, the new Center will develop and disseminate tools that can find causal links in very large and complex biomedical data. Faculty in CMU’s Department of Philosophy, led by Clark Glymour, PhD, Alumni University Professor and founding chair, are key partners in this data science effort; and Nicholas Nystrom, PhD, director of strategic applications at the Pittsburgh Supercomputing Center, will work to optimize these tools for a high-performance computing environment.
The Center includes a team that will develop and implement causal modeling and discovery algorithms, or processes, to support the data analyses of three separate investigative groups, each focusing on a distinct biomedical problem whose answer lies in a sea of data: cell signals that drive the development of cancer, the molecular basis of lung disease susceptibility and severity, and the functional connections within the human brain (the “connectome”).
“The Center also will be a training ground for the next generation of data scientists who will advance and accelerate the development and broader use of Big Data science models and methods,” said Dr. Bahar. “We will create new educational materials as well as workshops and online tutorials to facilitate the use of causal modeling and discovery algorithms by the broader scientific community and to enable efficient translation of knowledge between basic biological and applied biomedical sciences.”
Other collaborators include the California Institute of Technology, Rutgers University, University of Crete, and the University of North Carolina.
Pitt School of Dental Medicine Team Awarded $2 Million NCI Grant to Study How Cancer Spreads to Bone
 With a $2 million, 5-year grant from the National Cancer Institute (NCI), part of the National Institutes of Health, McGowan Institute for Regenerative Medicine affiliated faculty member Hongjiao Ouyang, DDS, PhD, DMD, and researchers at the University of Pittsburgh School of Dental Medicine will examine the molecular mechanisms that allow certain cancers, particularly multiple myeloma, to spread to the bone. The project could lead to new interventions to prevent such metastases and perhaps slow down primary tumor growth.
With a $2 million, 5-year grant from the National Cancer Institute (NCI), part of the National Institutes of Health, McGowan Institute for Regenerative Medicine affiliated faculty member Hongjiao Ouyang, DDS, PhD, DMD, and researchers at the University of Pittsburgh School of Dental Medicine will examine the molecular mechanisms that allow certain cancers, particularly multiple myeloma, to spread to the bone. The project could lead to new interventions to prevent such metastases and perhaps slow down primary tumor growth.
About 30 percent of multiple myeloma patients are diagnosed after going to the dentist with jaw pain or suspicious lesions in the oral cavity, said the project’s principal investigator Dr. Ouyang, associate professor of the Departments of Restorative Dentistry/Comprehensive Care and Oral Biology, and a member of the Center for Craniofacial Regeneration, Pitt School of Dental Medicine. Multiple myeloma is a cancer of plasma cells that begins in the bone marrow and is known for eating away the bone. Even with treatment, the bone lesions rarely heal.
“This bone destruction is a significant cause of pain and mortality in this disease,” said Dr. Ouyang, an endodontist and bone biologist. “A better understanding of the molecular pathways that underlie this process could lead us to novel targets for treatment.”
Bone marrow stromal cells (BMSCs) reside in the bone marrow and with appropriate stimulation can give rise to bone-forming cells called osteoblasts, fat cells, and other cells. In multiple myeloma, BMSCs produce growth factors and inflammatory proteins that boost tumor cells and activate osteoclasts, which are cells that break down bone while osteoblasts rebuild it as part of normal metabolism. In cancer, osteoclast activation makes holes in the bone that do not heal.
Dr. Ouyang’s team has found that the BMSCs in multiple myeloma patients, unlike those in healthy people, produce much more X-box binding protein (XBP1s), a molecule that has been shown in other tissues to regulate the production of inflammatory proteins. Their lab experiments showed that inducing healthy cells to produce XBP1s leads to changes in the bone microenvironment that support growth of multiple myeloma cells and osteoclast formation. Conversely, knocking out XBP1 production in multiple myeloma patient BMSCs corrected the abnormalities.
For the newly funded project, the team will determine the molecular mechanisms of the stromal XBP1 signaling in altering the bone microenvironment to favor multiple myeloma growth and bone destruction, as well as employ pharmacologic and genetic strategies to repress this molecule as a proof-of-concept for approaches to treat multiple myeloma bone disease.
“This could be helpful not only in treatment of multiple myeloma, but also in other cancers that spread to bone, such as breast, prostate, and lung cancer since BMSCs play a similar role in supporting tumor cell growth in these neoplastic diseases as well,” Dr. Ouyang said. “I am delighted that our research will benefit not only dental patients but also those affected by many other diseases.”
Dr. Keith Cook to Develop Artificial Lungs that Can Be Worn at Home
 Carnegie Mellon University (CMU) professor Keith Cook, PhD, has received a 4-year, $2.4 million grant from the National Institutes of Health (NIH) to support research and development of artificial lungs that patients may use long term in the comfort of their own homes while waiting for a lung transplant. Dr. Cook, an associate professor in the Department of Biomedical Engineering at CMU and a McGowan Institute for Regenerative Medicine affiliated faculty member, will lead the project and collaborate with researchers from the University of Washington, Columbia University, and Allegheny General Hospital in Pittsburgh.
Carnegie Mellon University (CMU) professor Keith Cook, PhD, has received a 4-year, $2.4 million grant from the National Institutes of Health (NIH) to support research and development of artificial lungs that patients may use long term in the comfort of their own homes while waiting for a lung transplant. Dr. Cook, an associate professor in the Department of Biomedical Engineering at CMU and a McGowan Institute for Regenerative Medicine affiliated faculty member, will lead the project and collaborate with researchers from the University of Washington, Columbia University, and Allegheny General Hospital in Pittsburgh.
“Far more people need a lung transplant or an artificial lung than what’s available,” said Dr. Cook, who noted that around 12 million people have chronic lung disease in the United States alone, but less than 2,000 people will be able to receive transplants. As a result, nearly 200,000 die from chronic lung disease every year.
For the last 20 years, Dr. Cook and other researchers have worked on artificial “bridge” lungs that support patients in need of a lung transplant who are placed on a waiting list. These devices have typically lasted days to a couple weeks before failure, and require close monitoring inside a hospital. Dr. Cook, now supported by the NIH grant, has been tasked with developing devices that last much longer and can be worn in homes.
“What got us this grant is that our initial results look very promising,” Dr. Cook said. He said the initial results showed a substantial decrease in the formation of blood clots inside the artificial lung, one of the largest hurdles in building a reliable long-term device.
“Anytime blood comes in contact with artificial material, it starts to clot,” Dr. Cook explained.
Coatings on the device’s artificial surfaces also can help fight clotting, and this will be Dr. Cook’s focus under the NIH grant. To do this, the team must combine several coating techniques that mimic what human blood vessel cells — called endothelial cells — are doing to prevent blood clots. Once this is perfected, Dr. Cook believes the longevity of the artificial lung can increase to 3 months.
“That’s the holy grail — 3 months,” Dr. Cook said. “Two is probably acceptable, but for quality of life, you don’t want a patient having to keep coming back to the hospital.”
Dr. Cook believes that patients could be using one of these artificial lungs in the next 5 to 10 years.
AWARDS AND RECOGNITIONS
Winner of the Michael G. Wells Student Health Care Entrepreneurship Competition Named
 Congratulations are extended to Travis Prest, who is a member of the laboratory of McGowan Institute for Regenerative Medicine faculty member Bryan Brown, PhD, research assistant professor with the Department of Bioengineering (BioE) at the University of Pittsburgh with a secondary appointment in Pitt’s Department of Obstetrics, Gynecology and Reproductive Sciences. Mr. Prest won the Michael G. Wells Student Health Care Entrepreneurship Competition for his project “Neurogel.” Neurogel is an injectable product which both improves and speeds recovery of peripheral nerves following injury.
Congratulations are extended to Travis Prest, who is a member of the laboratory of McGowan Institute for Regenerative Medicine faculty member Bryan Brown, PhD, research assistant professor with the Department of Bioengineering (BioE) at the University of Pittsburgh with a secondary appointment in Pitt’s Department of Obstetrics, Gynecology and Reproductive Sciences. Mr. Prest won the Michael G. Wells Student Health Care Entrepreneurship Competition for his project “Neurogel.” Neurogel is an injectable product which both improves and speeds recovery of peripheral nerves following injury.
Mr. Prest was selected from a group of six highly competitive finalists who presented their ideas at the “First Look” Technology Showcase on October 1, 2014. The “First Look” Technology Showcase is an annual event hosted by the Office of Enterprise Development within the University of Pittsburgh Innovation Institute that provides University student innovators the opportunity to present their ideas to an audience of invited guests including investors, entrepreneurs, regional economic development representatives, local business executives, and alumni of the University. Previous Wells competition winners have progressed within the University entrepreneurial ecosystem and have received additional funding through the Center for Medical Innovation and are currently preliminary candidates in the Coulter Translational Research Partners II Program.
As a winner, Mr. Prest will receive $10,000 to help advance his technology towards commercialization. Mr. Prest was assisted in preparation of his pitch by BioE Undergraduate Mr. Dhruv Srinivasachar. Mr. Srinivasachar, as well as BioE Undergraduate Mr. Yinka Olutoye and BioE Graduate Student Mr. Samuel LoPresti, also contributed to the science behind Neurogel.
Regenerative Medicine Podcast Update
 The Regenerative Medicine Podcasts remain a popular web destination. Informative and entertaining, these are the most recent interviews:
The Regenerative Medicine Podcasts remain a popular web destination. Informative and entertaining, these are the most recent interviews:
#140 – Dr. Kathryn Whitehead is an Assistant Professor in the Department of Chemical Engineering and Biomedical Engineering (courtesy) at Carnegie Mellon University. Dr. Whitehead discusses her research in the oral delivery of macromolecules.
Visit www.regenerativemedicinetoday.com to keep abreast of the new interviews.
Picture of the Month
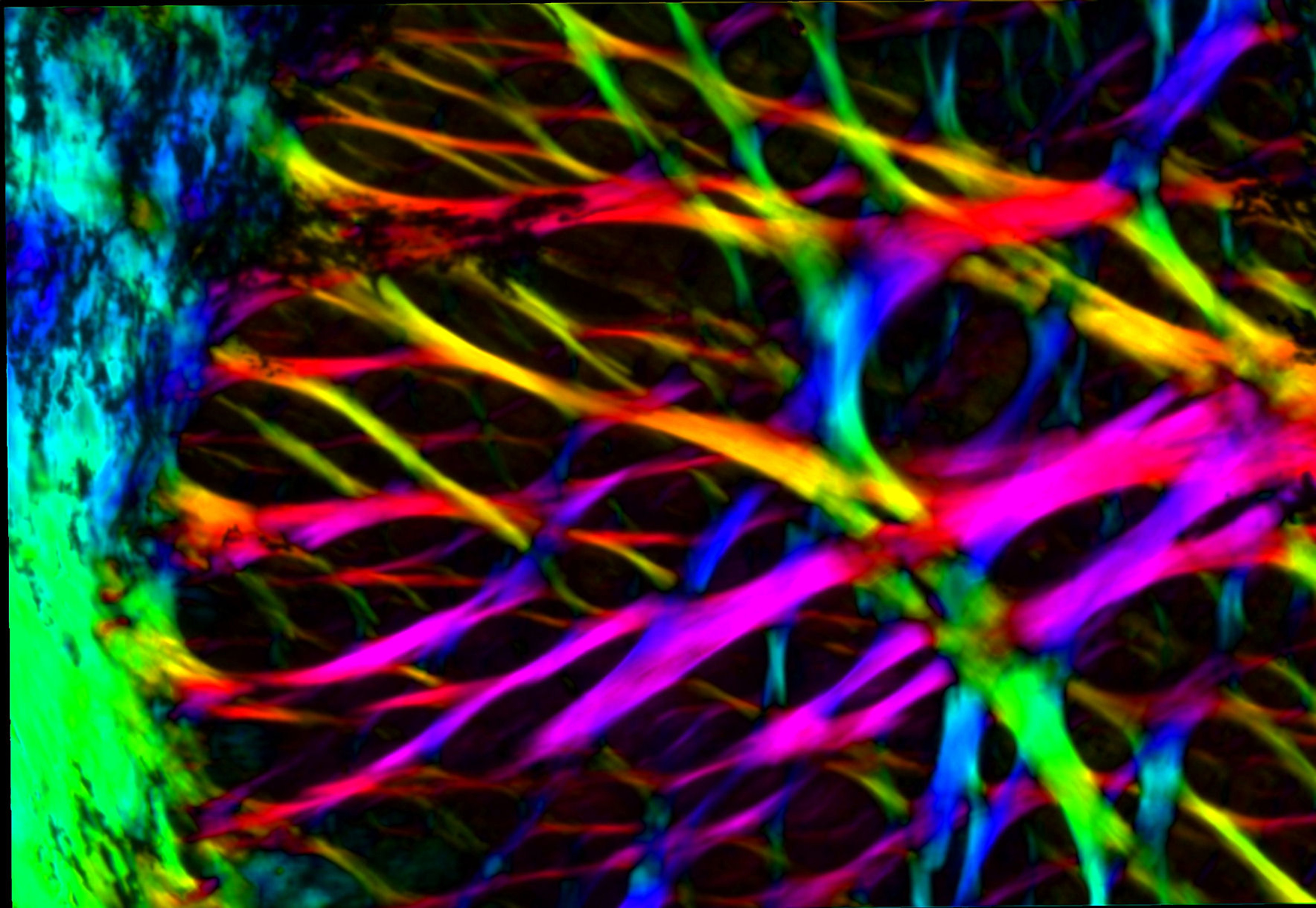
The collagen architecture in the lamina cribrosa, a structure in the optic nerve head or blind spot, of the eye – in this case of the eye of a sheep. The retinal ganglion cell axons pass through the pores visible here as they exit the eye on their way to the brain. The collagen of the lamina cribrosa provides structural support to the axons. The colors of the image represent the local orientation and amount of collagen fibers and provide a means to understand the complex interweaving of the fibers and ultimately of their role in protecting the nerve fibers. We study this structure to develop better preventative and treatment strategies for glaucoma and for optic nerve regeneration.
Image by Ning-Jiun Jan and Ian Sigal, PhD
