July 2020 | VOL. 19, NO. 7| www.McGowan.pitt.edu
AR3T Receives 5-Year NIH Grant Extension

NIH awarded The Alliance for Regenerative Rehabilitation Research and Training (AR3T) a 5-year renewal of the AR3T grant totaling $5,010,083. AR3T was one of six resource centers selected for this funding, all of which are a part of the Medical Rehabilitation Research Resource (MR3) Network.
Principal Investigators on this renewable effort are McGowan Institute for Regenerative Medicine affiliated faculty members:
- Fabrisia Ambrosio, PhD, MPT, Director of Rehabilitation for UPMC International and an Associate Professor in the Department of Physical Medicine & Rehabilitation at the University of Pittsburgh
- Michael Boninger, MD, Professor in the Department of Physical Medicine & Rehabilitation at the University of Pittsburgh School of Medicine, and Senior Medical Director for Post-Acute Care for the Health Services Division of UPMC and President of UPMC Innovative Homecare Solutions
- Thomas Rando, MD, PhD, Professor, Department of Neurology and Neurological Sciences, Stanford University School of Medicine, and the Director, Glenn Laboratories for the Biology of Aging, and the Deputy Director, Stanford Center on Longevity
An alliance of nationally recognized researchers guides AR3T from the University of Pittsburgh, Stanford University, Mayo Clinic, and the University of Texas at Austin.
AR3T’s overarching goal is to support the expansion of scientific knowledge, expertise, and methodologies across the domains of rehabilitation science and regenerative medicine. In addition to providing research support, education opportunities, mentored collaborative opportunities, and funds for pilot projects and technology development projects, this new cycle will also focus on supporting workforce diversity.
Congratulations and best wishes for continued success!
Illustration: The Alliance for Regenerative Rehabilitation Research and Training (AR3T).
RESOURCES AT THE MCGOWAN INSTITUTE
August Histology Special
Apoptosis, or “programmed cell death”, is a form of cell death designed to eliminate compromised or senescent cells. Apoptosis is controlled by multiple signaling and effector pathways that in turn, are activated in response to external growth, survival, or death factors.
A method for examining apoptosis via DNA fragmentation is the TUNEL assay. This technique can detect early-stage apoptosis in systems where chromatin condensation has begun and strand breaks are fewer, even before the nucleus undergoes recognizable morphologic changes.
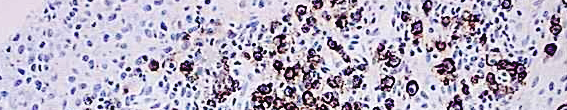
The McGowan Histology Core offers TUNEL staining on paraffin embedded or frozen tissue.
You’ll receive 30% off TUNEL staining in August when you mention this ad. Contact Julia at the McGowan Core Histology Lab by email: Hartj5@upmc.edu or call 412-624-5265.
New Sample Submission Procedures: In response to COVID-19, we ask that you contact us to schedule a drop off time. When you arrive at the building you can call our laboratory at (412)624-5365. Someone will meet you in the lobby to collect your samples. When your samples are completed, you will receive an email to schedule a pickup time.
Save the Date!
ICCAI 2020 Will Be Virtual

The 2020 International Conference on Complex Acute Illness (ICCAI’2020), titled “Meaningful AI in the Age of COVID-19”, will be held on September 10-11, 2020 entirely online, using Zoom teleconferencing technology. ICCAI is the annual meeting of the Society for Complex Acute Illness (SCAI), which, since its inception over 15 years ago, has successfully bridged the domains clinical practice of critical care medicine and quantitative approaches to understanding critical illness, with a strong translational focus. On the methodological side, it has emphasized eclecticism, covering, through its interdisciplinary membership, modeling approaches ranging from pure mechanistic differential equations models and agent-based models to purely data-driven machine learning techniques.
Please view the program and access the free registration site at http://iccai.org/
SCIENTIFIC ADVANCES
Researchers Study a Novel Type of Extracellular Vesicles

Researchers from Sechenov University and the University of Pittsburgh compared the properties of two groups of extracellular vesicles. Either present in a liquid phase or attached to the fibers of the extracellular matrix, these vesicles facilitate metabolism and cell-cell communication. A better understanding of their structure, production and movement can help create new bioengineered materials and repair damaged tissues more quickly.
Findings are published in Science Advances. McGowan Institute for Regenerative Medicine Deputy Director Stephen Badylak, DVM, PhD, MD, Professor in the Department of Surgery and Director of the Center for Pre-Clinical Tissue Engineering within the Institute, and affiliated faculty member Valerian Kagan, PhD, DSc, Professor and Vice-Chairman in the Department of Environmental and Occupational Health as well as a Professor in the Department of Pharmacology and Chemical Biology, the Department of Radiation Oncology, and the Department of Chemistry at the University of Pittsburgh, are co-authors on the study.
The extracellular matrix (ECM) fills the space within tissues, creating a scaffold for separate cells and a dynamic environment for cells, organelles, and molecules to move in. Varying in composition and consistency from tissue to tissue, it performs several key functions such as mechanical support of tissues, division of different types of cells, and transportation of substances, including signaling molecules.
Tissue engineering utilizes materials derived from the ECM, making it critical to preserve its structure and functions. Extracellular vesicles (EVs) are tiny vesicles released by cells and involved in the transfer of part of their content, such as proteins, lipids, enzymes, and microRNAs. Several years ago, researchers discovered that some of the vesicles are attached to the ECM fibers, terming them matrix-bound vesicles (MBVs). Their composition and membrane structure have not been studied so far.
The authors decided to compare the structure and properties of liquid-phase EVs and MBVs. In order to rule out the differences linked to their provenance from different tissues, the researchers used a cell culture environment to control the cell type and medium.
Both groups showed a similar size of no more than 200 nanometers (typical of most of the extracellular vesicles in general), but a different composition of proteins, lipids, and microRNAs they transport. MBVs, just like parent cells, contained a much greater share of cardiolipin, which is almost exclusively found in mitochondrial membranes—an indication that mitochondria and MBVs can have a common origin. Liquid-phase EVs, meanwhile, revealed a higher level of lipoxin, which inhibits inflammation and stimulates tissue regeneration (through the activation of immune cells). The study also highlighted differences in the contents of vesicles obtained from different tissues, although the set of lipids and microRNAs in MBVs is rather consistent.
The findings suggest that the future behavior of the vesicles (whether they will move in a liquid medium or stay attached to the matrix) is determined during their production stage. The fact that MBVs are embedded within a dense network of fibers shows that cells are most likely to release them when tissues are built, or the damaged matrix is repaired. Due to consistency of their lipids and microRNAs, MBVs can be used for both diagnostic and therapeutic purposes as well as the manufacture of new biomaterials.
Infant Heart-Assist Device Gets New Life with $4.7M Grant

The Center for Disease Control estimates that roughly 40,000 infants are born with congenital heart defects (CHDs) each year. Among that population, 25 percent are critical cases that require cardiac surgery. The waitlist for a heart transplant continues to grow; yet, the only FDA-approved life-saving device for CHD has shortcomings and is based on technology from the 1970s.
After being defunded by a company with rights to its intellectual property, development of a pediatric heart-assist device has been revived at Cornell University with the help of a $4.7 million grant from the U.S. Department of Defense. Faculty and students from the Swanson School of Engineering and the McGowan Institute for Regenerative Medicine are a part of this work.
Development and preclinical validation of the PediaFlow heart-assist system will be led by James Antaki, PhD, the Susan K. McAdam Professor of Heart Assist Technology at Cornell’s Meinig School of Biomedical Engineering. Dr. Antaki is an Adjunct Professor at the University of Pittsburgh in the Department of Bioengineering and a McGowan Institute affiliated faculty member. Pitt’s subcontract on this DoD award will include the efforts of McGowan Institute faculty member Marina Kameneva, PhD, research professor of surgery and bioengineering, and William Wagner, PhD, director of the McGowan Institute and distinguished professor of surgery, bioengineering and chemical engineering, who will direct studies assessing the biocompatibility and overall suitability of the PediaFlow pumps developed under the DoD award for clinical use. Salim Olia, PhD (BioE PhD ’18), adjunct assistant professor of surgery at the University of Pennsylvania School of Medicine conducted the crucial bench and preclinical PediaFlow tests as part of his PhD dissertation in the Department of Bioengineering at Pitt and is continuing his work for the DoD award.
“Despite the clinician mantra that ‘babies are not just tiny adults,’ pediatric heart pump development has been historically limited to the miniaturization of existing adult devices with minimal success,” said Dr. Olia. “PediaFlow represents a clean slate approach of designing from the ground up with the primary objective of maximizing patient safety by minimizing blood damage,” he continued.
According to Harvey Borovetz, PhD, Distinguished Professor of Bioengineering and the Robert L. Hardesty Professor of Surgery, “It is our goal at the end of the three-year DoD award to have completed development of the PediaFlow heart-assist pump, in anticipation of submitting an Investigational Device Exemption (IDE) application to the FDA and initiating clinical feasibility studies in these very special patients.”
PediaFlow is an alternative to a heart transplant for infants with congenital heart failure. The device is based on the world’s smallest magnetically levitated rotodynamic blood pump, which was invented by Dr. Antaki and colleagues. Implantation of the device – about the size of a AA battery – can potentially rehabilitate an infant’s heart by stimulating recovery of the muscle.
The system also includes a battery-operated external control unit and peripherals for diagnostics and maintenance.
Dr. Antaki and the Pitt team began work on the device in 2002, when the National Institutes of Health (NIH) issued a call for proposals for pediatric circulatory support because none existed at the time. By 2013, they had found a commercial partner for PediaFlow, as required by the NIH, and was ready to move the device into the next phase of development.
But the research came to an abrupt end when the partner was acquired by a company with no interest in pediatric heart pumps. The project was shelved and the government grant money that had been funding it was returned.
Dr. Antaki reluctantly turned his attention to other projects – until he was contacted by a woman asking to help her granddaughter, who needed a heart transplant.
“My first reaction was, ‘How can I possibly help you? I’m just a professor of biomedical engineering,’” said Dr. Antaki, “but then it occurred to me that if we were still doing the PediaFlow project, then I really could help.”
Dr. Antaki did some research into the Bayh-Dole Act, which allows federal contractors who acquire ownership of inventions made with federal funding to retain that ownership. Two large government contracts from the NIH had made the PediaFlow device possible, yet a private corporation had acquired the license for the technology for the purpose of preventing anyone from developing it further.
In March 2016, Dr. Antaki wrote to the act’s sponsor, former Republican Sen. Robert Dole of Kansas, who had taken a position at a Washington, D.C., law firm following his retirement from the Senate. Sen. Dole arranged an introduction to intellectual property lawyers from the firm; by November 2018, Dr. Antaki had won the right to develop the PediaFlow.
Said Dr. Antaki: “It is a new lease on life for this device for children who have no other alternative.”
Illustration: A rendering of a PediaFlow control unit prototype (top). Various components of the PediaFlow infant heart-assist device (bottom). James Antaki.
A Remote Control for Neurons
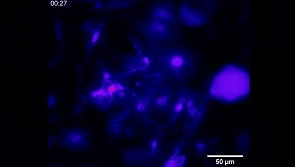
The team led by researchers at Carnegie Mellon University (CMU) has created a new technology that enhances scientists’ ability to communicate with neural cells using light. McGowan Institute for Regenerative Medicine affiliated faculty member Tzahi Cohen-Karni, PhD, CMU’s associate professor of biomedical engineering and materials science and engineering, led a team that synthesized three-dimensional fuzzy graphene on a nanowire template to create a superior material for photothermally stimulating cells. NW-templated three-dimensional (3D) fuzzy graphene (NT-3DFG) enables remote optical stimulation without the need for genetic modification and uses orders of magnitude less energy than available materials, preventing cellular stress. This work was presented in the Proceedings of the National Academy of the Sciences.
Graphene is abundant, cheap, and biocompatible. Dr. Cohen-Karni’s lab has been working with graphene for several years, developing a technique of synthesizing the material in 3D topologies that he’s labeled “fuzzy” graphene. By growing two-dimensional (2D) graphene flakes out-of-plane on a silicon nanowire structure, they’re able to create a 3D structure with broadband optical absorption and unparalleled photothermal efficiency.
These properties make it ideal for cellular electrophysiology modulation using light through the optocapacitive effect. The optocapacitive effect alters the cell membrane capacitance due to rapidly applied light pulses. NT-3DFG can be readily made in suspension, allowing the study of cell signaling within and between both 2D cell systems and 3D, like human cell-based organoids.
Systems like these are not only crucial to understanding how cells signal and interact with each other, but also hold great potential for the development of new, therapeutic interventions. Exploration into these opportunities, however, has been limited by the risk of cellular stress that existing optical remote-control technologies present. The use of NT-3DFG eliminates this risk by using significantly less energy, on a scale of 1-2 orders of magnitude less. Its biocompatible surface is easy to modify chemically, making it versatile for use with different cell types and environments. Using NT-3DFG, photothermal stimulation treatments could be developed for motor recruitment to induce muscle activation or could direct tissue development in an organoid system.
“This is an outstanding collaborative work of experts from multiple fields, including neuroscience through Pitt and UChicago, and photonics and materials science through UNC and CMU,” said Dr. Cohen-Karni. “The developed technology will allow us to interact with either engineered tissues or with nerve or muscle tissue in vivo. This will allow us to control and affect tissue functionality using light remotely with high precision and low needed energies.”
Additional contributions to the project were made by Maysam Chamanzar, PhD, assistant professor of electrical and computer engineering at CMU. His team’s core expertise in photonics and neurotechnologies assisted in developing the much-needed tools to allow both the characterization of the unique hybrid-nanomaterials, and in stimulating the cells while optically recording their activity.
“The broadband absorption of these 3D nanomaterials enabled us to use light at wavelengths that can penetrate deep into the tissue to remotely excite nerve cells. This method can be used in a whole gamut of applications, from designing non-invasive therapeutics to basic scientific studies,” said Dr. Chamanzar.
The team’s findings are significant both for our understanding of cell interactions and the development of therapies that harness the potential of the human body’s own cells. Nanostructures created using NT-3DFG may have a major impact on the future of human biology and medicine.
Other team members involved in the research included Biomedical Engineering PhD student Sahil Rastogi; Materials Science and Engineering PhD students Raghav Garg and Daniel San Roman and master’s student Nicholas Johnson; ECE PhD student Matteo Giuseppe Scopelliti; the University of Chicago’s Francisco Bezanilla, PhD, and Bernardo Pinto, PhD; the University of Pittsburgh’s Jane Hartung, PhD, and Michael Gold, PhD; and the University of North Carolina’s Seokhyoung Kim, PhD, Corban Murphey, and James Cahoon, PhD.
Illustration: Neurons respond to optical stimulus from NT-3DFG nanostructures. Carnegie Mellon University.
Immune System Discovery Could End Chronic Organ Rejection

Chronic rejection of transplanted organs is the leading cause of transplant failure, and one that the field of organ transplantation has not overcome in ok the field to flourish.
Now, a new discovery led by researchers at the University of Pittsburgh School of Medicine and Houston Methodist Hospital suggesting the innate immune system can specifically remember foreign cells could pave the way to drugs that lengthen long-term survival of transplanted organs. The findings, based on results in a mouse model, are published in the journal Science. McGowan Institute for Regenerative Medicine affiliated faculty member Hêth Turnquist, PhD, Associate Professor in the University of Pittsburgh School of Medicine, Department of Surgery, T.E. Starzl Transplantation Institute, is a co-author on the study.
“The rate of acute rejection within one year after a transplant has decreased significantly, but many people who get an organ transplant are likely to need a second one in their lifetime due to chronic rejection,” said Fadi Lakkis, MD, who holds the Frank & Athena Sarris Chair in Transplantation Biology and is scientific director of Pitt’s Thomas E. Starzl Transplantation Institute. “The missing link in the field of organ transplantation is a specific way to prevent rejection, and this finding moves us one step closer to that goal.”
The immune system is composed of innate and adaptive branches. The innate immune cells are the first to detect foreign organisms in the body and are required to activate the adaptive immune system. Immunological “memory” — which allows our bodies to remember foreign invaders so they can fight them off quicker in the future — was thought to be unique to the adaptive immune system. Vaccines, for example, take advantage of this feature to provide long-term protection against bacteria or viruses. Unfortunately, this very critical function of the immune system is also why transplanted organs are eventually rejected, even in the presence of immune-suppressing drugs.
In the new study, Dr. Lakkis, along with co-senior authors Martin Oberbarnscheidt, MD, PhD, assistant professor of surgery at Pitt, and Xian Li, MD, PhD, director of the Immunobiology & Transplant Science Center at Houston Methodist Hospital, used a genetically modified mouse organ transplant model to show that the innate immune cells, once exposed to a foreign tissue, could remember and initiate an immune response if exposed to that foreign tissue in the future.
“Innate immune cells, such as monocytes and macrophages, have never been thought to have memory,” said Dr. Oberbarnscheidt. “We found that their capacity to remember foreign tissues is as specific as adaptive immune cells, such as T- cells, which is incredible.”
The researchers then used molecular and genetic analyses to show that a molecule called paired Ig-like receptor-A (PIR-A) was required for this recognition and memory feature of the innate immune cells in the hosts. When PIR-A was either blocked with a synthetically engineered protein or genetically removed from the host animal, the memory response was eliminated, allowing transplanted tissues to survive for much longer.
“Knowing exactly how the innate immune system plays a role opens the door to developing very specific drugs, which allows us to move away from broadly immunosuppressive drugs that have significant side effects,” said Dr. Lakkis.
The finding has implications beyond transplantation, according to Dr. Oberbarnscheidt. “A broad range of diseases, including cancer and autoimmune conditions, could benefit from this insight. It changes the way we think about the innate immune system.”
Illustration: T.E. Starzl Transplantation Institute.
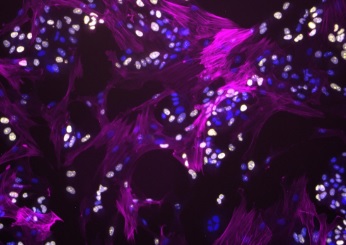
Reversing Drug Resistance in Breast Cancer
Roughly one in eight women in the United States will develop invasive breast cancer over the course of her lifetime, and HER2-positive (HER2+) breast cancers represent about 25 percent of all breast cancer cases. Though multiple therapies exist, most patients will develop metastatic disease and resistance to current treatments.
A collaborative research group from the University of Pittsburgh and Harvard Medical School studied the mechanisms behind tumor cell resistance to therapies targeting metastatic HER2+ breast cancer and recently published their work in the Proceedings of the National Academy of Sciences.
The group examined the tumor microenvironment — a collection of cells, molecules, and blood vessels that surround and influence tumor cells — to get the full picture of what drives this resistance. They found that fibroblasts, a cell type important to tissue regeneration, play a large role.
“Our study shows that fibroblasts promote drug resistance through a parallel signaling pathway in a subset of HER2+ breast cancer cells,” said McGowan Institute for Regenerative Medicine affiliated faculty member Ioannis Zervantonakis, PhD, assistant professor of bioengineering at Pitt’s Swanson School of Engineering.
Cancer cells grow uncontrollably, which is partially driven by continuous activation of proteins called kinases. HER2+ breast tumors have high levels of the HER2 receptor kinase that is important for their growth and is a major drug target. Anti-cancer drugs — like lapatinib — work to interfere with kinases, but because of this parallel signaling, fibroblasts are able to protect tumor cells and counteract the drug’s inhibitory effects.
Compare this cell-to-cell signaling process to communication through a radio. The radio antenna is analogous to a receptor in the cell that is activated when an electrical signal is received. Radios can have multiple antennas to respond to different electrical signals.
“By analogy, cancer cells have multiple receptors (antennas) that can be activated by signals in their environment,” explained Dr. Zervantonakis. “In the radio example, transmission of an electrical signal produces a sound. While in cancer cells, fibroblast-derived signals stimulate cancer growth.”
You can produce a series of sounds, and even amplify the volume, by receiving more than one signal at a time through activation of parallel pathways.
“In this scenario, the HER2+ kinase is one signal that is continuously activating sound, and then there is another pathway through which signals emitted by fibroblasts activate sound,” he continued. “Lapatinib only blocks the HER2+ kinase signal, but through another pathway, the fibroblasts are able to transmit signals to keep the cancer cells alive or allow them to grow.”
The findings in this paper are important for restoring sensitivity to breast cancer therapies and developing treatments that are more effective.
“Sensitivity to these drugs can be re-established through a combination of therapies that inhibit critical proteins in the pathway activated by fibroblasts,” said Dr. Zervantonakis. “Particularly, combination therapies with the FDA-approved drug everolimus and investigational agents targeting anti-apoptotic proteins were effective in restoring drug sensitivity in fibroblast-protected cancer cells.”
The next step for Dr. Zervantonakis and his lab is to create predictive mathematical models to identify the fibroblast density range in tumors that will elicit drug resistance. From there, they can develop personalized therapies to improve outcomes in HER2+ breast cancer.
Illustration: Fibroblasts modulate HER2 therapy sensitivity in breast tumor cells. Fibroblasts (magenta) interacting with proliferating breast tumor cells (yellow). Cell nuclei for both tumor and fibroblasts are stained in blue. (Image Courtesy of Alexis Scott)
‘Cell-Soldiers’ Turn Out to Be More Resistant Than ‘Cell-Combat Medics’

Researchers from Sechenov University and University of Pittsburgh discovered that the resistance of innate immune cells, macrophages, to ferroptosis – a type of programmed cell death – depends on the type of their activation. It turned out that cells helping tissues to recover from inflammation were more vulnerable. The researchers identified the mechanisms underlying the cells’ resistance and explained how this research would help regulate inflammation in a paper published in Nature Chemical Biology. McGowan Institute for Regenerative Medicine affiliated faculty members Valerian Kagan, PhD, DSc, Professor and Vice-Chairman in the Department of Environmental and Occupational Health as well as a Professor in the Department of Pharmacology and Chemical Biology, the Department of Radiation Oncology, and the Department of Chemistry at the University of Pittsburgh, and Ivet Bahar, PhD, Distinguished Professor, the John K. Vries Chair, and the Founding Chair in the Department of Computational & Systems Biology at the University of Pittsburgh’s School of Medicine, are co-authors on the study.
When tissues are damaged and bacteria or viruses enter the body, this triggers a defense reaction and starts inflammation. Vasodilation and increased temperature at the site of injury aim to attract macrophages (cells that can ‘capture’ and digest bacteria and also pick-up and eliminate the remains of dead cells). In this process, macrophages get activated: depending on the stage of inflammation and incoming signals, they can become ‘soldiers’ (M1-type macrophages), which attack foreign particles and damaged cells of the body, or ‘combat medics’ (M2-type), that remove cell fragments and regenerate tissues. At the beginning of inflammation, the actions of the first group of macrophages are required, but if they continue their work longer than necessary, the inflammation becomes chronic and the surrounding tissues can suffer, so it is important that the second team get into action on time.
One of the mechanisms that can prevent the transition of acute pro-inflammatory response to the resolution phase is the different resistance of M1- and M2-macrophages to ferroptosis. As the authors of the study found, M2-type cells are much more susceptible to this type of cell death. In addition, the cells debris accumulating during ferroptosis can further provoke inflammation and even worsen the situation.
Ferroptosis allows the body to get rid of damaged and metabolically defective cells; it is necessary for normal development of the body and can serve as a potential new strategy for fighting cancer (activating ferroptosis and killing the cancer cells can slow down tumor growth), but in many cases, cell death as a result of ferroptosis is undesirable, and chronic inflammation is among them. Therefore, scientists try to explore this process as much as possible to understand what makes cells resistant or, on the contrary, vulnerable to it.
The authors conducted many experiments to determine the mechanisms that cause different sensitivity of cells to ferroptosis. Based on their results and new information, a mathematical model has been developed describing the stability of macrophages of different types under different conditions. The model can both describe the observed processes and predict the results of experiments.
This work deepens our understanding of the cellular processes that determine the resistance/sensitivity of macrophages to ferroptosis, thus facilitating the targeted control of this type of cell death.
UPMC and VA Pittsburgh Partner to Perform VA’s First Living-Donor Living Transplant

VA Pittsburgh Healthcare System (VAPHS) and UPMC partnered to perform VA’s first living-donor liver transplant, in which a portion of a healthy person’s liver is transplanted to someone in need of a liver.
Army veteran Timothy Yablonski received a portion of liver from his daughter, Alyssa Last. Both transplant surgeries were completed at UPMC in mid-March.
One of 13 VA transplant centers, VA Pittsburgh performs in-house liver, kidney and living-donor kidney transplants and now collaborates with UPMC for living-donor liver transplants and small bowel transplants. VA Pittsburgh is the only VA medical center in the nation that now accepts living-donor liver transplant and small bowel transplant referrals. Mr. Yablonski’s surgery at UPMC marks the first time any VA in the nation has participated in a living-donor liver transplant.
“UPMC has a long history of collaboration and support for VA Pittsburgh and its transplant program. We are proud to offer lifesaving liver transplants to veterans both in our region, and across the country,” said Abhinav Humar, MD, chief of transplantation at UPMC and an affiliated faculty member of the McGowan Institute for Regenerative Medicine.
At UPMC, living-donor liver transplant is the first-line option rather than the last resort for patients. According to Dr. Humar, just 5% of all transplants across the country are living donors, while 60% at UPMC are from a living donor. As the most experienced living-donor liver transplant program in the country, UPMC transplant surgeons have saved the lives of hundreds of patients who might have otherwise died while waiting.
Mr. Yablonski lives in New York and receives care at the Syracuse VA Medical Center (VAMC). Ms. Last lives in Florida and although she is not a veteran, she is eligible for transplant follow-up care at the West Palm Beach VAMC. The Veterans Health Administration (VHA) also provides post-transplant care to veterans who have received a transplant at a non-VA medical facility either through dual care or primary care arrangements.
“As soon as we found out he was able to receive a living-donor liver transplant, I wanted to help,” said Ms. Last. “I was the first one who applied and was evaluated for it.”
“It made me feel incredible that my daughter wanted to support me,” said Mr. Yablonski. “What my doctor told me after the transplant is my daughter kept me alive.”
All veterans enrolled for VA health care are eligible for consideration for transplant services. VA transplant centers are designed specifically for veterans and their specific health care needs. VA policies support travel benefits, transplant procedures and follow-up care. VA also uses Clinical Video Telehealth (CVT) to expedite evaluations and follow-up transplant care while allowing veterans to save on travel time and costs.
For more information on transplant services at VA Pittsburgh and UPMC, visit:
Illustration: UPMC/VA Health System.
Pitt, UPMC Team Creates ‘Playbook’ for Return of Youth Athletics

A multidisciplinary team of clinicians and researchers at Pitt and UPMC has developed guidelines to assist coaches, athletic trainers, and organizers with creating a safe environment for youth athletes, fans, and staff as they consider a return to play.
Among the people involved with the creation of the playbook are Jeanne Doperak, DO, assistant professor of orthopaedic surgery and program director for the Primary Care Sports Medicine Fellowship; and McGowan Institute for Regenerative Medicine affiliated faculty member MaCalus Hogan, MD, MBA, associate professor of orthopaedic surgery and vice chairman of education and residency program director.
The UPMC Youth Sports Playbook contains recommendations for establishing a minimal set of standards in several categories for resuming athletic programs, including pre-participation physicals, social distancing, equipment sanitization, personal protective equipment, acclimation phases, practice and competition tactics and illness protocols.
AWARDS AND RECOGNITION
McGowan Institute for Regenerative Medicine 2020 Best Doctors

The McGowan Institute for Regenerative Medicine applauds its affiliated faculty members who were recently recognized by Pittsburgh Magazine. This year’s list was excerpted from The Best Doctors in America 2019-2020 database, which includes close to 40,000 U.S. doctors in more than 450 medical specialty/subspecialty combinations. The Best Doctors in America database is compiled and maintained by Best Doctors, Inc.
This year 26 McGowan Institute affiliated faculty were recognized in the May issue of the magazine with 3 colleagues* receiving dual specialty acknowledgment. Congratulations are extended to:
Anesthesiology: Erin Sullivan, MD
Cardiovascular Disease: Dennis McNamara, MD, John Pacella, MD
Critical Care Medicine: Derek Angus, MD, MPH, John Kellum, MD, FACP
Neurological Surgery: David Okonkwo, MD, PhD
Obstetrics and Gynecology: Stephen Emery, MD, Pamela Moalli, MD, PhD*
Ophthalmology: Ian Conner, MD, PhD
Orthopedic Surgery: Freddie Fu, MD, MaCalus Hogan, MD, Patrick McMahon, MD, Kurt Weiss, MD
Otolaryngology: Carl Snyderman, MD, MBA*
Pathology: Anthony Demetris, MD
Pediatric Cardiology: Jacqueline Kreutzer, MD
Pediatric Surgery: George Gittes, MD, George Mazariegos, MD
Plastic Surgery: Howard Edington, MD,* Michael Gimbel, MD, Ernest Manders, MD, Peter Rubin, MD, Mario Solari, MD
Surgery: Timothy Billiar, MD, Abhinav Humar, MD, Carl Snyderman, MD, MBA
Surgical Oncology: Howard Edington, MD
Thoracic Surgery: James Luketich, MD
Urology: Pamela Moalli, MD, PhD
Congratulations, all!
Parker Lab Student is Winner of ChE Spring Term 2020 Outstanding PhD Paper
 The Department of Chemical and Petroleum Engineering Spring Term 2020 Outstanding PhD Paper winner is Michelle Pressly, BS, a PhD candidate in the laboratory of McGowan Institute for Regenerative Medicine affiliated faculty member Robert Parker, PhD, Professor in the Department of Chemical and Petroleum Engineering at the University of Pittsburgh. She published the journal manuscript titled “Accelerating availability of clinically-relevant parameter estimates from thromboelastogram point-of-care device” in the Journal of Trauma and Acute Care Surgery. McGowan Institute affiliated faculty member Gilles Clermont, MD, Professor of Critical Care Medicine, of Industrial Engineering, and of Mathematics at the University of Pittsburgh, is also a co-author of this publication.
The Department of Chemical and Petroleum Engineering Spring Term 2020 Outstanding PhD Paper winner is Michelle Pressly, BS, a PhD candidate in the laboratory of McGowan Institute for Regenerative Medicine affiliated faculty member Robert Parker, PhD, Professor in the Department of Chemical and Petroleum Engineering at the University of Pittsburgh. She published the journal manuscript titled “Accelerating availability of clinically-relevant parameter estimates from thromboelastogram point-of-care device” in the Journal of Trauma and Acute Care Surgery. McGowan Institute affiliated faculty member Gilles Clermont, MD, Professor of Critical Care Medicine, of Industrial Engineering, and of Mathematics at the University of Pittsburgh, is also a co-author of this publication.
The goal of the work was to predict coagulopathy, which affects 1 in 3 trauma patients. Current test results for these patients can take over an hour to report. Ms. Pressly found that using only the first 3 minutes of data, the team’s algorithm could predict test results with high accuracy (with an area under the receiver operating characteristic curve of 0.95). She took this a step further by using the first 4 and 5 minutes to predict platelet and packed red blood cell transfusion needs, respectively. This work provides actionable results over 50 minutes sooner than was previously possible.
Congratulations, Ms. Pressly!
Illustration: ResearchGate (Pressly) and McGowan Institute (Parker/Clermont).
McGowan Institute Faculty Receive Pitt’s Center for Medical Innovation Awards
 The University of Pittsburgh’s Center for Medical Innovation (CMI) awarded grants totaling $60,000 to three research groups through its 2020 Round-1 Pilot Funding Program for Early Stage Medical Technology Research and Development. Two of the three project teams funded include McGowan Institute for Regenerative Medicine faculty members. They are:
The University of Pittsburgh’s Center for Medical Innovation (CMI) awarded grants totaling $60,000 to three research groups through its 2020 Round-1 Pilot Funding Program for Early Stage Medical Technology Research and Development. Two of the three project teams funded include McGowan Institute for Regenerative Medicine faculty members. They are:
“Targeted removal of cell-free plasma hemoglobin in extracorporeal therapies” – For an extracorporeal hemoperfusion device that removes plasma hemoglobin from a blood column using treated porous beads.
- Nahmah Kim-Campbell, MD, MS, Assistant Professor of Critical Care Medicine and Pediatrics
- William Federspiel, PhD, Professor of Bioengineering and Director of the McGowan Institute Medical Devices Lab
- Ryan Orizondo, PhD, Researcher in Bioengineering and Member of the McGowan Institute Medical Devices Lab
“Individual Biocontainment Unit for Reducing Viral Transmission to Healthcare Workers and Patients” – For the expedited development, approval, and manufacture of a novel device for use with ICU patients to reduce contamination by aerosolized particles.
- David M. Turer, MD, MS, Independent Resident, Department of Plastic Surgery, UPMC
- Heng Ban, PhD, Professor, Mechanical Engineering and Material Science, Swanson School of Engineering
- Peter Rubin, MD, Chairman, Department of Plastic Surgery, UPMC
CMI, a University Center housed in Pitt’s Swanson School of Engineering (SSOE), supports applied technology projects in the early stages of development with “kickstart” funding toward the goal of transitioning the research to clinical adoption. Proposals are evaluated on the basis of scientific merit, technical and clinical relevance, potential health care impact and significance, experience of the investigators, and potential in obtaining further financial investment to translate the particular solution to healthcare.
“This is our eighth year of pilot funding, and our leadership team could not be more excited with the breadth and depth of this round’s awardees,” said Alan Hirschman, PhD, CMI Executive Director and a McGowan Institute for Regenerative Medicine affiliated faculty member. “This early-stage interdisciplinary research helps to develop highly specific biomedical technologies through a proven strategy of linking UPMC’s clinicians and surgeons with the Swanson School’s engineering faculty.”
Congratulations, Drs. Federspiel, Rubin, and Orizondo!
Illustration: University of Pittsburgh’s Center for Medical Innovation.
Regenerative Medicine Podcast Update
The Regenerative Medicine Podcasts remain a popular web destination. Informative and entertaining, these are the most recent interviews:
#211 –– Dr. Ioannis Zervantonakis discusses his work in microfluidic and microfabricated assays to model the tumor microenvironment and to study tumor, fibroblast, and immune cell infiltration.
Visit www.regenerativemedicinetoday.com to keep abreast of the new interviews.
PUBLICATION OF THE MONTH
Author: Mark Asher Schusterman, MD, Robert D. Rehnke, MD, John M. Clarke, MD, Brent Price, MD, Uzma Waheed, MD, Richard Debski, PhD, Stephen F. Badylak, MD, PhD, DVM, and J. Peter Rubin, MD, FACS
Title: Breast Reconstruction Using a 3-dimensional Absorbable Mesh Scaffold and Autologous Fat Grafting: A Composite Strategy Using Tissue Engineering Principles
Summary: BACKGROUND: Breast reconstruction remains an important field in plastic surgery, with most procedures utilizing implants and/or autologous tissue. Few series report on experience with fat grafting as the primary form of breast reconstruction. This study reports a novel method of breast reconstruction using a 3-dimensional absorbable mesh scaffold and subsequent autologous fat grafting (AFG).
METHODS: A retrospective review was performed for all patients who underwent breast reconstruction using Lotus scaffold and AFG. Postoperative mammogram and magnetic resonance imaging were analyzed, and tissue specimens collected at subsequent procedures were harvested and stained with H&E for histologic evaluation. Finally, compression testing of the scaffold was performed using a tensiometer and digital tracking technology.
RESULTS: Twenty-two patients underwent reconstruction of 28 breasts using Lotus scaffold and AFG between February 2015 and February 2018. Average follow-up was 19 months. All patients were satisfied with final breast shape and size. Mean patient age was 60.5 years, and average body mass index was 28. Patients required on average 2 fat grafting sessions to achieve a successful result (range, 1–4). Postoperative mammogram and magnetic resonance imaging revealed robust adipose tissue in the breast with a slowly resorbing mesh and no oil cysts or calcifications. Histologic evaluated revealed no capsule formation with ingrowth of fat tissue around the scaffold. Compression testing revealed that the Lotus scaffold is a compliant construct with a high resilience profile.
CONCLUSIONS: The Lotus scaffold with AFG is a viable method for breast reconstruction, giving the patient an autologous reconstruction with less morbidity than free tissue transfer.
Source: Plast Reconstr Surg Glob Open. 2019 Aug; 7(8 Suppl): 23-24. Published online 2019 Sep 10.
GRANT OF THE MONTH
PI: Fabrisia Ambrosio
PI: Thomas Rando
Title: Alliance for Regenerative Rehabilitation Research & Training (AR3T)
Description: The advancement of regenerative medicine principles and technologies holds great potential to drive progress in the prevention and treatment of individuals with a host of pathologies resulting from injury, disease or aging. The long-term goal of regenerative medicine is to promote the repair, replacement, or regeneration of tissues. Likewise, rehabilitation seeks to harness the body’s innate regenerative potential in order to maximize function. Both fields hold great potential to drive progress in the treatment of a host of acute and chronic pathologies. We propose that these two fields are inextricably intertwined; an intersection of disciplines known as Regenerative Rehabilitation. To fully realize the tremendous potential of Regenerative Rehabilitation, we must promote the interaction of basic scientists with rehabilitation specialists. We must also train rehabilitation clinicians who can help oversee the quality, safety, and validity of these innovative Regenerative Rehabilitation technologies. The overarching goal of the Alliance for Regenerative Rehabilitation Research & Training (AR3T) is to establish a national network that will expand scientific knowledge, expertise and methodologies across the domains of regenerative medicine and rehabilitation. AR3T will provide didactic training that exposes rehabilitation researchers to cutting-edge investigations and state-of-the-art technologies in the field of regenerative medicine (Specific Aim 1). AR3T will drive the science underlying Regenerative Rehabilitation by: cultivating collaborative opportunities among rehabilitation researchers and internationally renowned investigators in the field of regenerative medicine (Specific Aim 2); launching a pilot funding program to support novel lines of Regenerative Rehabilitation investigations (Specific Aim 3); developing and validating technologies to elucidate the stem cell response to extrinsic mechanical signals (Specific Aim 4). Careful monitoring and evaluation of the effectiveness of our program, as outlined in Specific Aim 5, will ensure that these aims achieve the greatest success possible. These five aims have been organized to parallel each of the P2C grant proposal components.
Source: NIH
Term: 5 years
Amount: $5,010,083
