September 2017 | VOL. 16, NO. 9| www.McGowan.pitt.edu
Findings About Immune System Could Stop Allergic Skin Reactions at the Cellular Source

Recently reported by PittWire, many people have suffered through an itchy skin rash after a brush with poison oak, wearing jewelry containing nickel, or using latex gloves.
That rash is one of the several symptoms caused by allergic contact dermatitis (ACD), a common skin condition that also causes blistering, ulcers, and cracking skin, among other ailments.
Topical creams and ointments can relieve symptoms, but they do not treat the underlying causes.
Researchers at the University of Pittsburgh may have hit upon a better treatment. In a paper recently published in the Journal of Controlled Release, McGowan Institute for Regenerative Medicine affiliated faculty members Steven Little, PhD, chair of Pitt’s Department of Chemical and Petroleum Engineering, and Louis Falo, MD, PhD, chair of Pitt’s Department of Dermatology, and colleagues propose that the underlying causes of ACD can be remedied by manipulating T cells, which control inflammation.
“The technology here coaxes the body’s own cells to address inflammation that leads to these kinds of diseases,” said Dr. Little. “We are essentially using strategies like this to convince the immune system into not attacking something that it would normally attack. When we administer our treatment at the same time as the allergen, it teaches the body to not become inflamed to that specific thing.”
The researchers manipulated cells to release proteins, immune system molecules, and other compounds to suppress destructive hypersensitivity responses to allergens that cause skin rashes, effectively preventing or reversing ACD in previously sensitized mice.
Dr. Little said other researchers are trying to solve this problem by administering drugs that suppress the immune system, but side effects are a concern.
Another treatment method under investigation takes cells out of the body, manipulates them, and then reinjects them.
“This is really tough, because it is inefficient and we don’t know what happens to the cells when you put them back into the body,” Dr. Little said. “The FDA is wary of these kinds of things.”
The difference between these methods and the one proposed by Dr. Little and his colleagues is that it appears possible to induce the body’s own cells to treat the disease by manipulating cells inside the body with proteins that promote T cells to divide and react to allergens more quickly and aggressively to better control inflammation.
Researchers also said this approach to what is known as in vivo T cell induction could also aid in the development of new therapies for transplant rejection and autoimmune diseases.
“It has the potential to do all of this without the side effects you’d normally see,” Dr. Little said.
Illustration: PittWire.
RESOURCES AT THE MCGOWAN INSTITUTE
October Histology Special
Mucins are a family of high molecular weight, heavily glycosylated proteins (glycoconjugates) produced by epithelial tissues in most metazoans. Mucins’ key characteristics include their ability to form gels; therefore, they are a key component in most gel-like secretions, serving functions that range from lubrication to cell signaling to forming chemical barriers. For example, mucins often serve a modulatory role by controlling mineralization and bone formation in vertebrates. Mucins bind to pathogens as part of the immune system, and overexpression of mucin proteins, especially MUC1, is associated with many types of cancer.
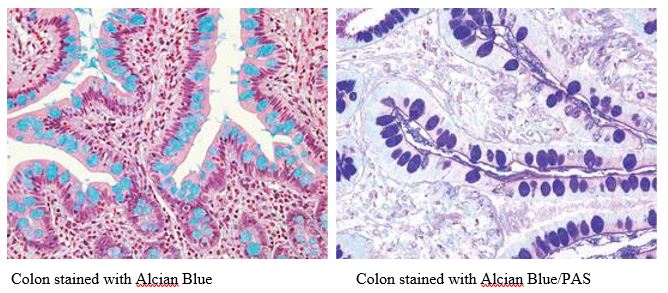
Alcian Blue / PAS
The combination of the alcian blue and various PAS techniques can be used to distinguish neutral mucins from acid mucins. In most protocols, sections are stained with the standard alcian blue (pH 2.5) method followed by the PAS technique. The alcian blue at a pH of 2.5 will stain all acid mucins deep blue but will not color the neutral mucins. The subsequent application of the PAS technique will stain the neutral mucins bright magenta. Tissues or cells that contain both neutral and acidic mucins may show a dark blue or purple coloration. The combined application of alcian blue and PAS is useful for several reasons. Changes in the distribution or pattern of expression of neutral and acid mucins are indicative of certain pathological conditions. In addition, the combined alcian blue/PAS technique is perhaps the most sensitive and comprehensive method for detection since all mucins should react regardless of the charge nature of the mucin.
You will receive 30% off your Alcian Blue or PAS staining, or both, when you mention this article any time in October.
Contact Lori at the McGowan Core Histology Lab and ask about our staining specials by email or call 412-624-5265. As always, you will receive the highest quality histology in the quickest turn-around time.
Did you know the more samples you submit to the histology lab the less you pay per sample? Contact Lori to find out how!
FACS Analysis of Stem Cell Membrane Fluidity
Jihee Sohn, PhD, Center for Cellular and Molecular Engineering, Department of Orthopaedic Surgery, is a postdoctoral associate in the laboratory of McGowan Institute for Regenerative Medicine associate director Rocky Tuan, PhD. Dr. Sohn conducts research to determine how adult mesenchymal stem cells (MSCs) can be manipulated for regenerative medical applications in the field of orthopaedics. She explains how flow cytometry helped her research project:
My current research focuses on how the composition and structure of the cell membrane affects the biology of MSCs, particularly in terms of the cell’s interaction with its physical environment. The number and distribution of lipid molecules, in particular cholesterol, in the plasma membrane may play a key role in governing stem cell phenotype, behaviors, and membrane properties. So, I have investigated the role of membrane cholesterol in regulating cell membrane fluidity.
The level of membrane cholesterol was modified in MSCs by either depleting or supplementing free cholesterol, and the resultant change in membrane fluidity was measured by a fluorescent labeling method (Membrane Fluidity kit, Abcam 189819). Briefly, cells were stained at room temperature with the fluorescent lipid reagent containing pyrenedecanoic acid, which exists as either a monomer or an excimer, the latter being formed upon monomer spatial interaction, resulting in a substantial red shift of the emission spectrum of the pyrene probe. Quantitative monitoring of the membrane fluidity can be attained by measuring the ratio of monomer to excimer fluorescence.
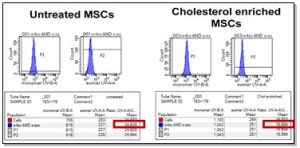
The manufacturer’s instructions provided are for adherent cells and for measurement of fluorescence intensity using a plate reader. To better describe the active movement of molecules within the lipid bilayer of cells in a dynamic environment, I have adopted the method for cells in suspension, with measurement of fluorescence intensity using a fluorescence-activated cell sorting (FACS) instrument. The protocol was developed with input from Abcam and Lynda Guzik, manager at the McGowan Institute Flow Cytometry Core Facility. The findings in Figure 1 show that cholesterol enrichment in MSCs resulted in decreased excimer:monomer ratio compared to untreated cells, which is an indication of decreased cell membrane fluidity.
Figure 1. Fluorescence intensities for excimer (450 nm excitation) and monomer (395 nm excitation) were measured. The ratio of excimer to monomer fluorescence provides a quantitative monitoring of the membrane fluidity of untreated- and cholesterol enriched-MSCs.
If you have a research project where flow cytometry may benefit your efforts, please feel free to contact Lynda Guzik, guzilj@upmc.edu, for more information.
UPCOMING EVENTS
Injury, Repair, and Regenerative Medicine Seminar Series Schedule Announced

McGowan Institute faculty affiliated members Drs. Cecelia Yates and Lauren Kokai have organized an exciting program for the Fall 2017 sessions of the McGowan Institute Injury, Repair, and Regenerative Medicine Seminar Series. This semester marks the first Faculty-Trainee Joint Lecture Series in ‘Biomaterials in Tissue Repair’. This series is co-organized by the newly established Society for Biomaterials Student Chapter at the University of Pittsburgh and combines the broad interests of the regenerative medicine community for both investigators and trainees.
This seminar series was created to give a forum for investigators, post-doctoral fellows, and graduate students studying tissue and cellular response to injury, repair, and remodeling. Our mission is to develop a community of researchers interested in wound healing, biomechanics, and biomedical engineering, as well as trauma and critical care medicine to promote the development of novel therapeutic strategies.
Also, new this year, selected lectures will be lived streamed and posted online, and selected lectures will be offer CME credits.
This seminar series is schedule for the 1st and 3rd Tuesday in Board Room – 5th Floor Eye and Ear Institute from 1-2pm. We invite you and your research lab to participate in this series. The current fall schedule can be found here.
International Symposium on Regenerative Rehabilitation

Medical advances in the field of Regenerative Medicine are accelerating at an unprecedented rate. Re-growing a lost limb, Restoring function to a diseased organ and harnessing the body’s natural ability to heal itself are becoming part of our reality. Technologies using biomaterials and cellular therapies to heal the body are in clinical trials and in clinical practice across the country. Regenerative Rehabilitation combines these discoveries with rehabilitation research in order to achieve synergy, propelling the translation into functionally relevant treatments and transforming the future of healthcare.
The Keynote Speaker is David J. Mooney, PhD of Harvard University. Dr. Mooney will speak on exploiting mechanics to promote regeneration.
The symposium will be held on November 1-3, 2017 at the University of Pittsburgh.
This symposium encourages participation of scientists, clinicians and physical therapists that are in the fields of regeneration and / or physical medicine and rehabilitation. This gathering is a platform for bridging these two areas of expertise in a setting that fosters discussion, interaction, cross discipline pollination intersection and networking.
For more information and to register, please click here.
SCIENTIFIC ADVANCES
Medical Implants and Aging

McGowan Institute for Regenerative Medicine faculty member Bryan Brown, PhD, is an Assistant Professor in the Department of Bioengineering with a secondary appointment in the Department of Obstetrics, Gynecology, and Reproductive Sciences at the University of Pittsburgh. Recently, he discussed with Liz Reid of National Public Radio’s WESA his current research with different bioactive coatings to modulate the immune response to implanted devices.
Dr. Brown is trying to figure out how to harness the immune system’s natural inflammatory response to better integrate these devices into the body. In his lab, researchers develop materials for different surgical applications and observe how the body responds using animal trials.
“Think about some of the cells that come in there [during inflammation]: they clean up debris, they kill bacteria. Those are important functions,” said Dr. Brown. “And so, any time we have an implant we create a surgical injury, so we have inflammation whether we want it or not.”
“Inflammation can actually be productive in terms of helping an implant to integrate within the body,” Dr. Brown said. But in older mammals, including those people most likely to wind up with hip replacements and pacemakers, the response becomes less effective.
Dr. Brown said figuring out what is different about the inflammation that happens following the implantation of a medical device in an aged animal versus young could help researchers find ways to make devices work better in the body. This is important, he said, because while we are living longer, we aren’t necessarily staying healthy longer.
Illustration: Liz Reid/WESA.
An Eye Towards Islets

Tiny packets of cells called islets throughout the pancreas allow the organ to produce insulin. Type 1 diabetes – also known as juvenile diabetes – tricks the immune system into destroying these islets. Patients must take insulin daily to maintain blood sugar, or too much sugar will build up in the blood stream and lead to hyperglycemia, diabetic ketoacidosis, and, if left untreated, death. Patients must self-regulate their blood sugar for their entire lives, unless there were some way to restore the pancreatic islets.
To explore that potential, the National Science Foundation has funded a multi-university study led by researchers at the University of Pittsburgh Swanson School of Engineering who are investigating the use of human pluripotent stem cells (hPSCs) to engineer pancreatic islets in the lab. A major goal of the research is to develop a method of vascularizing islets in vitro—literally “in glass”—which studies suggest will result in higher viability and enhanced function after the transplant.
“This the first attempt to generate in vitro vascularized pancreatic islet organoids from hPSCs,” explains McGowan Institute for Regenerative Medicine affiliated faculty member Ipsita Banerjee, PhD, associate professor of chemical engineering at Pitt and principal investigator of the study. “Through collaborative efforts, we have developed a method of implanting blood vessel fragments into the islets. By vascularizing the islets before they are transplanted to the body, they are more likely to survive and can begin regulating blood glucose more quickly.”
Pancreatic islets have a very high oxygen demand. Once inside the body, they need to connect to the host vessels quickly, otherwise they start dying and lose their ability to regulate blood glucose levels. Researchers began looking for new techniques to speed up vascularization after tests began to show high vascularity ultimately improved the transplantation outcome.
In addition to developing vascularized islets inside the lab, the study – “Engineering a functional 3D vascularized islet organoid from pluripotent stem cells” – will use a novel hydrogel system to create a three-dimensional cell culture configuration that mimics the way the body forms pancreatic cells naturally.
“The hydrogel is like a scaffold, and it helps to configure the cells in a 3D space,” says Dr. Banerjee. “The status quo is hPSCs randomly arranged in uncontrolled configurations with varying size and structure; however, by using the hydrogel developed by our collaborator at Arizona State, we can create a precise, multicellular architecture called ‘spheroids.’ Unlike a 2D culture grown in a petri dish, islet spheroids grown on the hydrogel look the same as the ones made by the body.”
Although Dr. Banerjee’s research will most directly impact cell therapy for diabetics, creating a procedure for developing working islets outside of the body could also serve as a valuable tool for testing the efficacy and toxicity of new drug compounds for pancreatic disease. The general implications of in vitro vascularization of cells show even more promise.
“The principles behind pre-designing vascularization before transplantation apply to any type of tissue, not just pancreatic,” Dr. Banerjee says. “Even when donor islets are used for a transplant, a fraction of the islets survive the procedure. We expect the advanced measures we are taking in the lab, before the new cells enter the patient’s body, to have tremendous application to the next generation of regenerative medicine.”
Dr. Banerjee’s team of researchers includes McGowan Institute for Regenerative Medicine affiliated faculty member Prashant Kumta, PhD, professor of bioengineering at Pitt; Kaushal Rege, professor of chemical engineering at Arizona State University; and James Hoying, professor of surgery at the University of Louisville.
Pitt Receives NIH Grant to Develop 3-D Tissue Chips that Mimic Human Joints
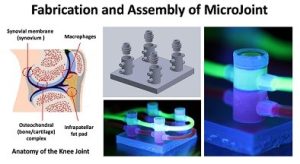
The National Institutes of Health (NIH) has awarded a $1.2 million grant to a multi-institutional project led by the University of Pittsburgh to engineer a 3-dimensional joint-on-a-chip called the “microJoint,” to replicate a human joint on a small scale. The microJoint will be used to study and test drugs for the treatment of arthritic joint diseases. McGowan Institute for Regenerative Medicine associate director Rocky Tuan, PhD, is the principal investigator of the award.
Trauma, inflammation, infection, and aging can cause damages to joint tissues, ultimately leading to arthritic disorders, including osteoarthritis, that cause physical disability and compromise quality of life. Despite being the most common degenerative joint disease, affecting almost 1 in 6 people, osteoarthritis has no effective treatments, largely because the disease is poorly understood, and animal models that are commonly used in research do not sufficiently replicate human disease due to anatomical and functional differences.
The microJoint project led by Pitt, with collaborators at Stanford University and Tulane University, is part of a national initiative by the National Center for Advancing Translational Sciences (NCATS) of the NIH to develop 3-D microphysiological system platforms that model human disease. The Tissue Chip for Disease Modeling and Efficacy Testing program intends to enable researchers to better recognize and identify disease and more accurately predict how patients will respond to treatment drugs. The Pitt project is the only one in the program that is focusing on degenerative joint diseases.
Pitt is one of 13 institutions receiving NIH awards to support research aimed at improving the success rate of drugs that are tested in clinical trials. Currently, more than 60 percent of investigational drugs fail in clinical trials due to a lack of effectiveness, despite showing promise in cell and animal research models.
“We’re building what will be the first joint-on-a-chip that we hope will accurately replicate arthritic diseases in humans, and thus allow in-depth understanding of the disease process that will lead to discovery of potential therapies,” said Dr. Tuan, director of the Center for Cellular and Molecular Engineering, and Distinguished Professor in the Department of Orthopaedic Surgery at the University of Pittsburgh School of Medicine. Dr. Tuan is internationally known for his research in stem cell biology, musculoskeletal tissue engineering, regenerative medicine, and for his innovative leadership role in biomedical education. He also is director of Pitt’s Center for Military Medicine Research.
In addition to Dr. Tuan, the co-investigators on the award are Bruce Bunnell, PhD, director of the Tulane Center for Stem Cell Research and Regenerative Medicine and professor of pharmacology at Tulane University School of Medicine, and Stuart Goodman, MD, professor of orthopedic surgery at Stanford University.
The researchers will engineer the chip by creating and combining multiple joint tissues, including cartilage, bone, the joint capsule, and fat. The tissues will be generated using a specific type of adult stem cells called mesenchymal stem cells, encapsulated within a light-activated gel that will act as a scaffold. In addition, the model will include macrophages, which are immune cells that are linked to inflammation commonly seen in joint disorders. The biological interaction among these joint tissues and cells will be modeled by culturing them in individual custom-fabricated micro-bioreactors that are connected to each other.
The first microJoint models will seek to replicate normal joints and those affected by injury, inflammation, and diabetic complications. Once that is accomplished, the researchers plan to test different drug compounds for their ability to heal diseased joints.
The awards are supported by the NCATS Tissue Chip for Drug Screening program, which leads this effort in collaboration with other NIH Institutes and Centers.
Illustration: Tissue components of the knee joint (left) modeled by the human microJoint, consisting of tissues housed in individual microbioreactors that are connected (right). Credit: Rocky Tuan/University of Pittsburgh.
Dr. Eric Beckman Receives NSF Award

The National Science Foundation recently awarded a 3-year, $300K grant entitled, “SusChEM: machine learning blueprints for greener chelants.” McGowan Institute for Regenerative Medicine affiliated faculty member Eric Beckman, PhD, Bevier Professor of Engineering in the Chemical Engineering Department at the University of Pittsburgh and a Co-Director of the Mascaro Center for Sustainable Innovation, is the project co-principal investigator along with principal investigator John Keith, PhD, R.K. Mellon Faculty Fellow in Energy, Department of Chemical Engineering. The award begins August 1, 2017, and extends through July 31, 2020.
From the project abstract: Chelating agents have recently been identified as a key category of chemical products that are ripe for greener design. It is hypothesized that identifying better alternatives will require far broader explorations of chemical compound space than what is possible with conventional trial and error experimentation. In this project, accurate quantum chemistry calculations will be used to train state-of-the-art machine learning methods that will allow prediction of structures of greener chelating agents.
The machine learning method that will be developed promises a novel route to rapidly predict properties of chelant/metal complexes, not only with higher accuracy but with six orders of magnitude less computational time than conventional predictive quantum chemistry methods (e.g. Kohn-Sham Density Functional Theory). With this computational tool, it will be possible to rapidly screen through about 100,000 hypothetical chelant structures to predict those that would bind strongly to different metal ions. The project will also screen these complexes to see which have high propensities to degrade in reasonable timeframes, and which have low probabilities of being toxic. The top candidates from this novel screening approach will then be experimentally synthesized and tested. This will validate if quantum chemistry-based machine learning would be a transformative tool for environmental sustainability and green chemical design by being a more predictive supplement and/or alternative to conventional QSAR models.
Pitt Researcher Receives NIH Funding Aimed at Preventing Bed Sores

Heat, moisture, and force, in addition to other factors, can lead to pressure injuries — or bed sores — which are common among immobile patients and individuals who use wheelchairs. Many technologies and guidelines exist to help prevent and treat pressure injuries, but there is little evidence to prove which technologies are most effective for patients with different risk factors.
McGowan Institute for Regenerative Medicine affiliated faculty member David Brienza, PhD, professor in the University of Pittsburgh School of Health and Rehabilitation Sciences, was recently awarded a $2.6 million grant from the National Institutes of Health to study the technologies used in hospital beds aimed at preventing pressure injuries.
One technology, called low air loss, redistributes pressure and establishes a healthy microclimate for the patient’s skin by wicking away moisture and circulating air under the bed where the patient is lying.
“Some low air loss mattresses transmit moisture and heat better than others,” said Dr. Brienza. “The information currently provided to caregivers to help them select pressure injury prevention devices does not account for performance variability among the different technologies.”
Dr. Brienza’s randomized trial, which will take place entirely at UPMC Presbyterian and UPMC Montefiore, will enroll more than 800 patients over the course of 3 years. The study is designed to determine if and when low air loss is effective at preventing pressure injuries, and what level of heat and moisture control performance are necessary for prevention effectiveness.
“Patients have different risk factors for pressure injuries, so it is important to determine which measurable mattress characteristics benefit which patients,” said Dr. Brienza. “When the study concludes, we hope to have scientific evidence to back up a performance-based algorithm that would guide caregivers to the mattress systems that best accommodate a patient’s individualized needs.”
Illustration: University of Pittsburgh School of Health and Rehabilitation Sciences.
Pitt Researchers Awarded Funding in Neuroscience Research Grants

McGowan Institute for Regenerative Medicine affiliated faculty members at the University of Pittsburgh have been awarded grants from the National Science Foundation (NSF) and the National Institutes of Health (NIH) to study diverse aspects of how the brain works.
The grants were awarded as part of the federal BRAIN Initiative, a large-scale effort announced in 2013 aimed at gaining a deeper understanding of the brain and applying the knowledge to prevent and treat brain disorders.
The following researchers and projects were awarded grants:
Matthew Smith, PhD, associate professor of ophthalmology at the Pitt School of Medicine, and Byron Yu, PhD, associate professor of electrical and computer engineering and biomedical engineering at Carnegie Mellon University, were awarded a 4-year, $1 million grant by the NSF to understand how the sensory environment and state of mind combine to affect our perception and interpretation of the world around us. The researchers will use brain computer interfaces with implications for treatment of psychiatric disorders.
Pitt’s Avniel Singh Ghuman, PhD, assistant professor of neurosurgery, and Mark Richardson, MD, PhD, associate professor of neurosurgery, will collaborate with Carnegie Mellon University researchers Max G’Sell, PhD, assistant professor of statistics, and Louis-Philippe Morency, PhD, assistant professor of computer science, to understand how our brain perceives and understands the actions, emotions, and communication of others. The NSF-funded, 3-year grant totaling approximately $1 million will allow researchers to understand brain circuits in a real-world setting. Researchers will record electrical brain activity in patients undergoing neurosurgical treatment for epilepsy while they have natural interactions with friends, family, doctors, and hospital staff. Ultimately, the researchers hope to provide much greater insight into neural processes that become dysfunctional in debilitating brain disorders such as autism and post-traumatic stress disorder.
AWARDS AND RECOGNITION
American Health Council Names Dr. William Federspiel to Education Board

McGowan Institute for Regenerative Medicine faculty member William Federspiel, PhD, William Kepler Whiteford Professor of Bioengineering at the University of Pittsburgh, has been selected to join the Education Board at the American Health Council. He will share his knowledge and expertise in healthcare education, workforce development, business, leadership, management, chemistry, biology, biomedical engineering, and medical devices.
Dr. Federspiel has been active in the healthcare industry for the last 28 years. He earned his Bachelor of Science degree in 1978 and his PhD in Chemical Engineering in 1983 from the University of Rochester. Inspired to pursue his profession by a desire to help people, Dr. Federspiel has now been a bioengineering professor at the University of Pittsburgh for 21 years. His day-to-day responsibilities include teaching and mentoring, administrative duties, and research grants.
Dr. Federspiel has been published in over 100 peer-reviewed journals and has held academic positions in biomedical engineering at Johns Hopkins University and Boston University prior to joining the University of Pittsburgh. Awards and honors he has received include Fellow of the Biomedical Engineering Society (BMES), Certificate of Appreciation for Distinguished Service to the Rehabilitation Research and Development Service Scientific Merit Review Board from the Department of Veterans Affairs, and an Honorable Mention in the Start-Up Entrepreneur category of the 2014 Carnegie Science Awards. Dr. Federspiel is also a Fellow of the American Institute for Medical and Biomedical Engineering (AIMBE) and member of the American Society for Artificial Internal Organs (ASAIO). His long-term professional goals include helping his start-up company be successful and seeing his respiratory assist devices treating patients.
Among his many achievements, Dr. Federspiel is most proud to be a founder of ALung Technologies, a Pittsburgh-based medical start-up company, for which he serves as the head of the scientific advisory board.
Anna Balazs, PhD, Named the John A. Swanson Endowed Chair in Engineering

Recognizing her contributions to the fields of chemical engineering and computational modeling, the University of Pittsburgh has appointed McGowan Institute for Regenerative Medicine affiliated faculty member Anna Balazs, PhD, as the John A. Swanson Chair in Engineering at the Swanson School of Engineering. University of Pittsburgh Chancellor Patrick Gallagher made the appointment on the recommendation of Provost Patricia Beeson and U.S. Steel Dean of Engineering Gerald Holder.
“Anna’s appointment to the John A. Swanson Chair in Engineering in the Swanson School of Engineering recognizes and rewards the quality and impact of her work to date, which has earned deep and widespread respect,” said Chancellor Gallagher. “This designation is well deserved—and one of the highest honors that any university can bestow upon a member of its faculty.”
“Anna’s award-winning contributions to her field have been tremendous, and she is one of the most valued and respected members of our faculty,” Dean Holder added. “But most importantly, she has been and continues to be a mentor to so many students and post-doctoral researchers who have been impacted by her innovative research, creativity, and wonderful personality.”
Dr. Balazs is also the Distinguished Professor of Chemical Engineering and previously held the Robert v.d. Luft Professor at the Swanson School. She received her BA in physics from Bryn Mawr College in 1975 and PhD in materials science from the Massachusetts Institute of Technology in 1981. Her research involves developing theoretical and computational models to capture the behavior of polymeric materials, nanocomposites, and multi-component fluids, with funding awarded by the National Science Foundation, Department of Energy, Department of Defense, and the Charles E. Kaufman Foundation.
She is a Fellow of the American Physical Society, the Royal Society of Chemistry, and the Materials Research Society, and was a Visiting Fellow at Corpus Christi College, Oxford University. She has served on a number of editorial boards including Macromolecules, Langmuir, Accounts of Chemical Research, and Soft Matter, and currently serves as an Associate Editor for the journal Science Advances. She was Chair of the American Physical Society Division of Polymer Physics in 1999-2000, and received a Special Creativity Award from the National Science Foundation. Her other awards include the Maurice Huggins Memorial Award of the Gordon Research Conference for outstanding contributions to Polymer Science (2003), the Mines Medal from the South Dakota School of Mines (2013), the American Chemical Society Langmuir Lecture Award (2014), and the Royal Society of Chemistry S F Boys-A Rahman Award (2015). Most recently, she was the first woman to receive the prestigious American Physical Society Polymer Physics Prize (2016).
Dr. Robert Parker Earns Endowed Professorship in Pitt’s ChemE

McGowan Institute for Regenerative Medicine affiliated faculty member Robert Parker, PhD, Professor in the Department of Chemical and Petroleum Engineering at the Swanson School of Engineering at the University of Pittsburgh, Member of the Molecular Therapeutics/Drug Discovery Program at the University of Pittsburgh Cancer Institute, and Member of the Clinical Research, Investigation, and Systems Modeling of Acute Illness Center in the Department of Critical Care Medicine at the University of Pittsburgh, will become a William Kepler Whiteford Endowed Professor in Engineering for a term of 5 years.
As noted by McGowan Institute faculty member Steven Little, PhD, Chairman of the Department of Chemical and Petroleum Engineering and the William Kepler Whiteford Endowed Professor in the Departments of Chemical and Petroleum Engineering, Bioengineering, Immunology, and Ophthalmology:
“I am very proud that Bob’s leadership is being recognized by this Endowed Professorship. Amongst many other things, Bob has created the Systems Medicine focus in the Department with multiple collaborators through his grants from the NIH, DoE, and NSF. Bob’s total funding to date is now over $16M. He has published a number of papers in impressive journals. He is also responsible for multiple Departmental group grants including REU’s and GAANN’s. Bob is also one of the most beloved teachers by our students, and this past year scored an absolutely phenomenal 4.75 in ChemE 0500 (which was a course that he originally wasn’t even scheduled to teach), not to mention his new, highly innovative, ChemE 1933, Engineering a Craft Brewery. And BTW, Bob also was appointed as Vice Chair of Graduate Education in the Department this past year.”
Congratulations, Dr. Parker!
Dr. Shilpa Sant Named 2017 Young Innovator of Cellular and Molecular Bioengineering

McGowan Institute for Regenerative Medicine affiliated faculty member Shilpa Sant, PhD, was named one of eleven of the 2017 Young Innovators of Cellular and Molecular Bioengineering (CMBE) and her work is featured in the October 2017 issue of the journal. As a 2017 CMBE Young Innovator, Dr. Sant will present her research in a special, two-part invited platform session at the 2017 Annual Meeting of BMES in Phoenix, Arizona.
Dr. Sant is an Assistant Professor in Pharmaceutical Sciences, with a secondary appointment in Bioengineering. Research in her lab focuses on developing physiologically relevant in vitro disease models and developing novel therapies to combat these diseases.
In the 2017 CMBE Young Innovator issue, Dr. Sant, along with Pitt Pharmacy graduate student, Yingfei Xue, and collaborators report their work on reactive oxygen species (ROS)-modulating shape-specific cerium oxide nanoparticles (CNPs) as a therapeutic strategy to inhibit oxidative stress-induced heart valve calcification. Indeed, elevated levels of reactive oxygen species in valve tissue have been identified as a prominent hallmark and driving factor for valvular calcification, which still remains a significant clinical challenge without effective pharmacological treatments. To address this, Dr. Sant and colleagues developed an oxidative stress-induced valve calcification model using valve interstitial cells (hVICs) isolated from two patients with stenotic valves and a healthy donor. They demonstrated that hVICs derived from calcified valves exhibited impaired antioxidant defense mechanisms and were more susceptible to oxidative stress than normal hVICs. They further demonstrated therapeutic effect of ROS-modulating shape-specific cerium oxide nanoparticles (CNPs) to inhibit oxidative stress-induced valvular calcification. CNPs are a class of self-regenerative ROS-modulating agents, which can switch between Ce3+ and Ce4+ in response to the surrounding oxidative microenvironment.
Other Pitt co-authors of this work include Cynthia St. Hilaire, PhD, and Luis Hortells, DVM, PhD (Cardiology & Vascular Medicine Institute); McGowan Institute for Regenerative Medicine affiliated faculty member Julie Phillippi, PhD (Cardiothoracic Surgery); and Vinayak Sant, PhD (Pharmaceutical Sciences). Read the full research article here.
Building on this work, Dr. Sant and co-authors envision to create a three-dimensional valve calcification model using human cells so that it can be further extended for testing potential therapies. They also envision that self-regenerative cerium oxide nanoparticles will be a useful platform to balance ROS for many other diseases where excessive oxidative stress is implicated in disease progression.
Cellular and Molecular Bioengineering, an official journal of the Biomedical Engineering Society (BMES), publishes research that advances the study and control of mechanical, chemical, and electrical processes of the cell. Each year, the editors of CMBE dedicate the Young Innovators special issue to highlighting the work of assistant professors conducting innovative bioengineering research at the molecular, cellular, and multi-cellular level.
Regenerative Medicine Podcast Update
The Regenerative Medicine Podcasts remain a popular web destination. Informative and entertaining, these are the most recent interviews:
#175 –– Dr. Lorenzo Soletti is the Co-Founder and CEO of Renerva, LLC. Dr. Bryan Brown is also the Co-Founder of Renerva, as well as an Assistant Professor in the Department of Bioengineering with a secondary appointment in the Department of Obstetrics, Gynecology, and Reproductive Sciences at the University of Pittsburgh. Drs. Soletti and Brown discuss Renerva, a company that is developing surgical solutions to improve peripheral nerve repair, accelerate healing following a nerve injury, and improve nerve function following injury and repair.
Visit www.regenerativemedicinetoday.com to keep abreast of the new interviews.
PICTURE OF THE MONTH
We encourage submissions of images that highlight your research. If you have images, or have an idea for an interesting image, please contact Donna Stolz, PhD at donna.stolz@pitt.edu.
PUBLICATION OF THE MONTH
Author: Balmert SC, Donahue C, Vu JR, Erdos G, Falo LD Jr, Little SR
Title: In vivo induction of regulatory T cells promotes allergen tolerance and suppresses allergic contact dermatitis.
Summary: Allergic contact dermatitis (ACD) is a common T-cell mediated inflammatory skin condition, characterized by an intensely pruritic rash at the site of contact with allergens like poison ivy or nickel. Current clinical treatments use topical corticosteroids, which broadly and transiently suppress inflammation and symptoms of ACD, but fail to address the underlying immune dysfunction. Here, we present an alternative therapeutic approach that teaches the immune system to tolerate contact allergens by expanding populations of naturally suppressive allergen-specific regulatory T cells (Tregs). Specifically, biodegradable poly(ethylene glycol)-poly(lactic-co-glycolic acid) (PEG-PLGA) microparticles were engineered to release TGF-β1, Rapamycin, and IL-2, to locally sustain a microenvironment that promotes Treg differentiation. By expanding allergen-specific Tregs and reducing pro-inflammatory effector T cells, these microparticles inhibited destructive hypersensitivity responses to subsequent allergen exposure in an allergen-specific manner, effectively preventing or reversing ACD in previously sensitized mice. Ultimately, this approach to in vivo Treg induction could also enable novel therapies for transplant rejection and autoimmune diseases.
Source: J Control Release. 2017 Sep 10;261:223-233. Epub 2017 Jul 8.
GRANT OF THE MONTH
PI: Rocky Tuan
Title: Tissue Chip Modeling of Synovial Joint Pathologies: Effects of Inflammation and Adipose-Mediated Diabetic Complications
Description: Trauma, inflammation, infection, and aging can cause damages to joint tissues, ultimately leading to arthritic disorders, such as osteoarthritis (OA), septic arthritis, and inflammatory arthritis, resulting in physical disabilities that compromise quality of life; however, no efficacious therapies are currently available. The limited progress in the development of disease-modifying medications (DMMs) is principally because of: (1) insufficient mechanistic understanding of disease onset/progression; (2) inability to encompass the 3-dimensional (3D) and multi-tissue nature of the synovial joint in early phase in vitro drug discovery; and (3) limited utility of pre-clinical animal studies for early stage clinical efficacy and toxicity prediction (lacking “fail early/fail fast” capabilities), resulting in unanticipated and costly clinical trial failures. Also, patient-specific etiology, progression, and drug sensitivity profiles underscore the need for personalizable therapy development. To address these needs, we propose engineering a 3D human micro-joint chip (mJoint), physiologically analogous to the native joint and capable of modeling pathogenesis of joint diseases for DMM screening/development.
UG3 – Aim 1: Engineering joint components – The osteochondral complex, synovium, and adipose, will be engineered using primary cells, human mesenchymal stem cells (MSCs) or induced pluripotent stem cell (iPSC) derived MSCs encapsulated in a photocrosslinked hydrogel scaffold, with macrophages included to evaluate their critical function in mediating/regulating inflammation, and phenotype-characterized using molecular, biochemical and histological analyses.
Aim 2: Generating normal and diseased mJoint – A bioreactor will be designed to house all of the joint elements (mJoint), simulating the respective in vivo tissue conditions, and exposed to various pathogenic agents and conditions to model OA, inflammatory arthritis, and adipose-mediated diabetic joint complications, which will be assessed based on changes in histology and structure in each individual joint component as well as biomarkers.
UH3 – Aim 3: Investigating tissue interactions and developing specific biomarkers using mJoint – We will assess the contribution of and the interactions among the joint tissue components under normal and diseased conditions. Joint diseases with different etiologies (see Aim 2) will be simulated, tissue interactions analyzed, and potential biomarkers developed to predict joint health.
Aim 4: Testing known drugs and screening candidate DMMs – We will assess the efficacy of known and candidate DMMs using the mJoint disease models, including interleukin-4, NF-κB decoy oligonucleotides, statins, metalloproteinases inhibitors, and others, focusing also on the applicability of biomarkers identified in Aim 3.
Aim 5: Testing potential of cell- based therapy – The therapeutic efficacy of human MSCs and their products, such as exosomes and conditioned media, and other biologics, will be examined, in order to explore the scientific basis of the widely perceived utility of stem cell-based therapy for musculoskeletal disorders.
In summary, the mJoint represents a high-utility in vitro platform to model synovial joint pathologies and to screen therapeutics for the treatment of joint diseases.
Source: National Center for Advancing Translational Sciences
Term: 15 July, 2017 – 30 June, 2019
Amount: $1,129,655