January 2017 | VOL. 16, NO. 1| www.McGowan.pitt.edu
NIH Award: Ambulatory Assist Lung for Children

McGowan Institute for Regenerative Medicine faculty members–
- William Federspiel, PhD, William Kepler Whiteford Professor in the Department of Bioengineering with secondary appointments in Chemical Engineering and Critical Care Medicine, and Director of the Medical Devices Laboratory at the McGowan Institute,
- William Wagner, PhD, Director of the McGowan Institute for Regenerative Medicine as well as a Professor of Surgery, Bioengineering and Chemical Engineering at the University of Pittsburgh, and
- Peter Wearden, MD, PhD, congenital cardiothoracic surgeon and Department Chair, Division of Cardiovascular Surgery, Department of Cardiovascular Services at the Nemours Children’s Health System, Orlando, Florida–
are the principal investigators on a recently funded R01 grant from the National Heart, Lung, and Blood Institute of the National Institutes of Health entitled “Ambulatory Assist Lung for Children.”
Acute and chronic lung diseases remain the most life threatening causes of death and hospitalization in the pediatric population. Cystic fibrosis, pulmonary hypertension, and pulmonary fibrosis have been observed to be the most frequent causes of lung failure in pediatric patients. Mechanical ventilation and extracorporeal membrane oxygenation (ECMO) have been used to bridge sick kids to transplant. These procedures can lead to poor post-transplant outcomes by their very restrictive nature on mobility. This project will develop a compact respiratory assist device for pediatric patients, the Pittsburgh Pediatric Ambulatory Lung (P-PAL) to replace ECMO as a bridge to transplant or recovery in kids with acute and chronic lung failure.
The P-PAL is a wearable and fully integrated blood pump and gas exchange module that will be designed for implantation of inflow cannula and outflow cannula/grafts in the right atrium and pulmonary artery, respectively. The P-PAL will be designed for longer-term respiratory support (1-3 months before cartridge change-out) at 70-90% of normal metabolic oxygenation requirements, while pumping blood from 1 to 2.5 Liters/min.
The specific aims of project are:
- To modify the design and operational parameters of the P-PAL to meet requirements for blood pumping, gas exchange, priming volume, and form factor;
- To build P-PAL prototypes along the design development pathway for bench characterization studies of pumping performance, gas exchange, and hemolysis;
- To improve the hemocompatibility of the P-PAL by exploiting novel polymeric zwitterionic coatings that we have already begun to develop for our adult wearable assist lung; and
- To perform acute and chronic studies in preclinical studies to demonstrate the in-vivo performance and hemocompatibility of the P-PAL device and to study its interaction with the cardiopulmonary system.
RESOURCES AT THE MCGOWAN INSTITUTE
February Histology Special
The McGowan Institute Histology Core wants to be your Valentine this month with big savings that will warm your heart. Bring your cardiac tissue to the McGowan Histology Core and receive 25% off your entire order when you mention this ad. From our hearts to yours, you won’t beat these prices, or our rapid turnaround times. Race in today for top quality results!!
Did you know the more samples you submit to the histology lab the less you pay per sample? Contact Lori to find out how!Contact Lori at the McGowan Core Histology Lab and ask about our staining specials by email or call 412-624-5265. As always, you will receive the highest quality histology in the quickest turn-around time.
SCIENTIFIC ADVANCES
News from the Bryan Brown Laboratory

The National Institute of General Medical Sciences (NIGMS) awarded principal investigator Bryan Brown, PhD, Assistant Professor in the Departments of Bioengineering and Obstetrics, Gynecology, and Reproductive Sciences and faculty member at the McGowan Institute for Regenerative Medicine, a 5-year R01 award for his work entitled “Assessing the Impact of Macrophage Polarization Upon the Success of Biomaterial Implants.” Co-investigators on this project include McGowan Institute affiliated faculty members Pamela Moalli, MD, PhD, Associate Professor, Departments of Obstetrics, Gynecology, and Reproductive Sciences and Bioengineering, and Stephen Badylak, DVM, PhD, MD, Professor in the Department of Surgery, deputy director of the McGowan Institute, and Director of the Center for Pre-Clinical Tissue Engineering within the Institute.
Additionally, at the end of 2016, Dr. Brown’s team finalized a 1-year, $174,633 research grant from ACell, Inc. entitled “Assessing the Impact of Aging upon the Host Response to MatriStem®”. This grant will be complementary to a future National Institute on Aging (NIA) R01 award scheduled for Spring 2017.
Lastly, Brown Lab students Dan Hachim and Aimon Ifitikhar were finalists in the TERMIS Business Plan Competition. They were awarded 2nd place at the Annual Meeting in December 2016.
Congratulations, all!
Preclinical Study: Using MRI to Monitor ECM Hydrogel in the Brain Post-Stroke

The research efforts of McGowan Institute for Regenerative Medicine affiliated faculty members Michel Modo, PhD, associate professor in the Department of Radiology at the University of Pittsburgh with secondary appointments in the Department of Bioengineering and the Center for Neural Basis of Cognition, and Stephen Badylak, DVM, PhD, MD, a deputy director of the McGowan Institute, professor in the Department of Surgery, and director of the Center for Pre-Clinical Tissue Engineering within the Institute, were recently published in the journal, Biomaterials. Their paper is entitled “Diamagnetic chemical exchange saturation transfer (diaCEST) affords magnetic resonance imaging of extracellular matrix hydrogel implantation in a rat model of stroke.” This study was funded by a seed grant from the Department of Health of the Commonwealth of Pennsylvania (4100068505) and the National Institute for Neurological Disease and Stroke (R01NS08226).
The paper abstract reads:
Extracellular matrix (ECM) is widely used as an inductive biological scaffold to repair soft tissue after injury by promoting functional site-appropriate remodeling of the implanted material. However, there is a lack of non-invasive analysis methods to monitor the remodeling characteristics of the ECM material after implantation and its biodegradation over time. We describe the use of diamagnetic chemical exchange saturation transfer (CEST) magnetic resonance imaging to monitor the distribution of an ECM hydrogel after intracerebral implantation into a stroke cavity. In vitro imaging indicated a robust concentration-dependent detection of the ECM precursor and hydrogel at 1.8 and 3.6 ppm, which broadly corresponded to chondroitin sulfate and fibronectin. This detection was robust to changes in pH and improved at 37°C. In vivo implantation of ECM hydrogel into the stroke cavity in a rat model corresponded macroscopically to the distribution of biomaterial as indicated by histology, but mismatches were also evident. Indeed, CEST imaging detected an endogenous “increased deposition.” To account for this endogenous activity, pre-implantation images were subtracted from post-implantation images to yield a selective visualization of hydrogel distribution in the stroke cavity and its evolution over 7 days. The CEST detection of ECM returned to baseline within 3 days due to a decrease in fibronectin and chondroitin sulfate in the hydrogel. The distribution of ECM hydrogel within the stroke cavity is hence feasible in vivo, but further advances are required to warrant a selective long-term monitoring in the context of biodegradation.
New Understanding of Pulmonary Hypertension Leads to Promising Drug Targets

A groundbreaking new study led by researchers from the University of Pittsburgh and UPMC—including McGowan Institute for Regenerative Medicine affiliated faculty member Marc Simon, MD, Associate Professor of Medicine, Director of Heart Failure Research, Medical Director of the Montefiore Clinical & Translational Research Center of the Clinical Translational Science Institute, and an Attending Physician in the Advanced Heart Failure and Cardiac Transplantation Section in the Heart and Vascular Institute of the University of Pittsburgh—has identified a new group of compounds that could have robust effects in treating pulmonary hypertension (PH), an enigmatic but sometimes fatal disease of the blood vessels of the lungs that currently has no cure. The findings, which were published in The Journal of Clinical Investigation, highlight the use of these drugs to alter vessel stiffness and its downstream control of metabolism, a link previously unknown for people suffering from the progressive disease.
“If we aim to cure this disease, the next set of medications and treatments should be those that target the origin at the molecular level,” said Stephen Y. Chan, MD, PhD, director of the UPMC Center for Pulmonary Vascular Biology and Medicine at the Vascular Medicine Institute at Pitt, and senior author of the study. “As a community, we are struggling right now to understand those origins of PH, and this study aimed to address that untapped need.”
Affecting tens of millions of people worldwide, PH is high blood pressure in the arteries in the lungs, which makes it difficult for blood to flow from the heart to the lungs. Symptoms of the disease, which can lead to heart failure, include shortness of breath, fatigue, and chest pain; and, in its early stages, might not be noticeable for months or even years. Often a life-threatening condition, it becomes progressively worse, making early and accurate diagnosis important to allow treatments that extend and improve the quality of life for many patients.
To make these discoveries, Dr. Chan and his colleagues used a comprehensive array of tools derived from cells and tissues of animal and humans with PH. Notably, these findings also were relevant to PH caused by human immunodeficiency virus (HIV) infection—a particularly mysterious form of this disease where the underlying molecular processes have remained unknown for decades.
Dr. Chan’s team found that stiffening or hardening of the vessels in the lung is an early event in PH that triggers the activation of two critical signaling molecules called YAP and TAZ. These molecules in turn activate a protein called GLS1, which controls how cells in the vessel produce and use energy.
“The link between vessel hardening and energy production is absolutely central to this disease,” said Dr. Chan. “That discovery offers us so many new ways to design drugs tailor-made to stop PH in its tracks.”
As proof-of-concept, Dr. Chan’s team tested both the YAP inhibitor verteporfin, a Food and Drug Administration-approved medication for macular degeneration, and a GLS1 inhibitor called CB-839, which is in clinical trials for cancer. They found that both of these compounds displayed robust effects in improving PH in a rodent model of disease.
“We are very encouraged by these results,” said Dr. Chan. “We are working to repurpose these drugs for treatment of human PH, which now can include long-neglected disease types such as HIV-related conditions and others. We hope that we can do so without the delay of decades that often happens when developing new compounds from scratch.”
Given that vessel stiffness is prevalent in other diseases—including cancer progression—these results also may be important beyond PH, noted Dr. Chan.
“There is always more work to be done,” he said. “But, we feel this represents a significant milestone in our quest to cure this disease.”
Illustration: UPMC.
New Study Shows Targeted Therapy Needed for Breast Cancer with Brain Metastases

Researchers at the University of Pittsburgh Cancer Institute (UPCI) and Magee-Womens Research Institute (MWRI) have discovered molecular changes in the primary tumor of breast cancer patients who developed brain metastases. The finding is expected to lead to improved diagnosis and targeted therapies.
The results, published in the Journal of the American Medical Association (JAMA) Oncology and presented at the 2016 San Antonio Breast Cancer Symposium, indicate that patients’ treatments should be tailored not only for the original breast cancer, but also the brain tumors, said Adrian Lee, PhD, director of the Institute for Precision Medicine, a joint effort by UPMC and the University of Pittsburgh, and director of the Women’s Cancer Research Center, a collaboration between UPCI and MWRI.
Dr. Lee’s research team—including McGowan Institute for Regenerative Medicine affiliated faculty member Steffi Oesterreich, PhD, Professor of Pharmacology and Chemical Biology at the University of Pittsburgh Cancer Institute and Director of Education at the Women’s Cancer Research Center—set out to determine if there were molecular differences in the primary breast tumors and their patient-matched brain metastatic tumors that would enhance treatment options.
“The brain is a common and catastrophic site of metastasis for breast cancer patients,” said Dr. Lee. “Our study showed that despite the large degree of similarity between the initial breast tumor and the brain metastatic tumor, there were enough alterations to support comprehensive profiling of metastases to potentially alter the course of treatment.”
There are many types of breast cancers, and about 20 percent of them are identified as HER2-positive, meaning the cancer cells have more of a protein called HER2 that causes the cells to grow faster than those with normal levels of the protein. These patients’ cancers typically respond to targeted therapies. However, breast cancer patients with brain metastases who are identified as HER2-negative do not respond favorably to the same therapies.
Little research is available because there are few opportunities to study primary breast tissue with their patient-matched brain metastases. The research team tested tumors from 20 patients from two academic institutions, the University of Pittsburgh and the Royal College of Surgeons in Ireland. Among the discoveries was that the primary tumor initially identified as HER2-negative actually switched to HER2-positive in the metastatic brain tumor.
“This now means we can screen for presence of HER2 so that we can change and target the therapy to improve outcomes for our patients,” said Dr. Lee.
Understanding Why We Should Exercise

Physical activity is good for our overall well-being. Yet, science has never determined exactly why that is. The University of Pittsburgh has been chosen by the National Institutes of Health (NIH) for a large-scale collaboration to investigate why being active is beneficial for our health.
Pitt School of Education faculty member John Jakicic, PhD, will lead Pitt’s contributions to the Molecular Transducers of Physical Activity in Humans program, which seeks to fully understand how physical activity affects the human body at the molecular level. Ultimately, the findings of this NIH endeavor could assist health professionals with physical-activity recommendations and strategies for individuals at various stages of life and with particular health needs.
McGowan Institute for Regenerative Medicine affiliated faculty member Bradley Nindl, PhD, director of the Neuromuscular Research Laboratory in the Department of Sports Medicine and Nutrition within the School of Health and Rehabilitation Sciences, is a member of Dr. Jakicic’s research team.
The Pitt team is 1 of only 19 research centers in the nation to be selected for this collaboration and will share approximately $170 million over the next 6 years for program research. This will be the largest-ever NIH investment into the study of how physical activity improves health and prevents disease.
The research teams will perform individual projects centered around the examination of internal bodily elements—hormones, nucleic acids, proteins, etc.—that transmit the health effects of physical activity. They will then work to determine how certain variables—age, sex, and fitness level, among others—alter these molecular messengers. All of the findings will be housed in a publicly accessible database that future researchers can use to further the field of study.
The Jakicic-led team is one of six research teams that will recruit healthy adults from diverse racial and ethnic backgrounds for an intensive exercise study. The team will collect blood, urine, and tissue samples from active and sedentary volunteers performing resistance and aerobic exercises. These samples will be shared with the other research teams in the collaboration.
“The National Institutes of Health’s latest endeavor to examine the health effects of physical exercise on the human body could potentially transform clinical medicine’s use of physical activity as a treatment and preventive strategy,” said Dr. Jakicic, chair of Pitt’s Department of Health and Physical Activity within the School of Education. “This collaboration will bring together some of the nation’s most advanced researchers in the field of health and physical activity, and the University of Pittsburgh is excited and proud to be a part of it.”
Other prominent Pitt faculty members from various corners of the University community will join Dr. Jakicic for this endeavor: Daniel E. Forman, chair of the Section of Geriatric Cardiology within the Department of Medicine; Erin E. Kershaw, chief of the Division of Endocrinology and Metabolism within the Department of Medicine; Anne B. Newman, chair of the Department of Epidemiology within the Graduate School of Public Health; Lindsay C. Page, an assistant professor of research methodology within the School of Education and a research scientist within the Learning Research and Development Center; and Renee J. Rogers, an assistant professor of health and physical activity within the School of Education.
Pitt’s Center for Medical Innovation Awards Four Novel Biomedical Devices

The University of Pittsburgh’s Center for Medical Innovation (CMI) awarded grants totaling $77,500 to four research groups through its 2016 Round-2 Pilot Funding Program for Early Stage Medical Technology Research and Development. The latest funding proposals include a new technology for treatment of diabetes, a medical device for treating patients requiring emergent intubation, an innovative method for bone regeneration, and a novel approach for implementing vascular bypass grafts. Each project includes McGowan Institute for Regenerative Medicine affiliated faculty members on its team.
CMI, a University Center housed in Pitt’s Swanson School of Engineering (SSOE), supports applied technology projects in the early stages of development with “kickstart” funding toward the goal of transitioning the research to clinical adoption. CMI leadership evaluates proposals rated on scientific merit, technical and clinical relevance, potential health care impact and significance, experience of the investigators, and potential in obtaining further financial investment to translate the particular solution to healthcare.
“This is our fifth year of pilot funding, and our leadership team could not be more excited with the breadth and depth of this round’s awardees,” said McGowan Institute affiliated faculty member Alan D. Hirschman, PhD, CMI Executive Director. “This early-stage interdisciplinary research helps to develop highly specific biomedical technologies through a proven strategy of linking UPMC’s clinicians and surgeons with the Swanson School’s engineering faculty.”
AWARD 1: Intrapancreatic Lipid Nanoparticles to Treat Diabetes
Award for further development and testing of use of lipid nanoparticle technology for the induction of α-to-β-cell transdifferentiation to treat diabetes.
George Gittes, MD
Department of Surgery
University of Pittsburgh School of Medicine
Kathryn Whitehead, PhD
Department of Chemical Engineering
Carnegie Mellon University
AWARD 2: The Esophocclude – Medical Device for Temporary Occlusion of the Esophagus in Patients Requiring Emergent Intubation
Continuation award for further refinement of the Esophocclude Medical Device using human cadaver testing to simulate emergency intubation.
Philip Carullo, MD
Resident, PGY-1
Department of Anesthesiology
University of Pittsburgh Medical Center (UPMC)
Youngjae Chun, PhD
Assistant Professor
Department of Industrial Engineering
Department of Bioengineering (Secondary)
University of Pittsburgh
AWARD 3: RegenMatrix – Collagen-Mimetic Bioactive Hydrogels for Bone Regeneration
Continuation award for fully automating the hydrogel fabrication process, for animal studies and for fine-tuning related innovations.
Shilpa Sant, PhD
Assistant Professor
Department of Pharmaceutical Sciences
Department of Bioengineering
University of Pittsburgh
Akhil Patel, MS
Graduate Student
Department of Pharmaceutical Sciences
University of Pittsburgh
Yadong Wang, PhD
Professor
Department of Bioengineering
University of Pittsburgh
Sachin Velankar, PhD
Associate Professor
Department of Chemical Engineering
University of Pittsburgh
Charles Sfeir, DDS, PhD
Associate Professor
Department of Oral Biology
University of Pittsburgh
AWARD 4: TopoGraft 2.0 – Anti-Platelet Surfaces for Bypass Grafts and Artificial Hearts Using Topo-Graphic Surface Actuation
Continuation award for in-vivo validating of results and developing a new approach for topographic actuation of the inner lumen of synthetic bypass grafts.
Sachin Velankar, PhD
Department of Chemical Engineering
University of Pittsburgh
Luka Pocivavsak, MD, PhD
Department of Surgery
University of Pittsburgh Medical Center
Edith Tzeng, MD
Department of Surgery
University of Pittsburgh Medical Center
Robert Kormos, MD
Department of Cardiothoracic Surgery
University of Pittsburgh Medical Center
Illustration: University of Pittsburgh Swanson School of Engineering.
AWARDS AND RECOGNITION
Tuan Lab Trainee Receives Iris Klarman Women’s Health Fellowship Award

The Foundation for Women’s Wellness has awarded the 2016 Iris Klarman Women’s Health Fellowship Award to Riccardo Gottardi, PhD, a fifth year post-doc fellow in orthopaedic regenerative medicine at the University of Pittsburgh School of Medicine, Department of Orthopaedic Surgery. His research is examining gender differences in osteoarthritis and joint damage, specifically the effect of sex hormones on cartilage in menopausal women (who are disproportionately affected by this disease). He has an impressive list of awards and academic success. In talking to Dr. Gottardi, it is clear that he is not only capable and highly motivated, but also passionate about improving the lives of women suffering from these diseases. His advisor wrote that Dr. Gottardi is “one of the brightest and highly ranked trainees (in our) department.”
Dr. Gottardi works in the laboratory of McGowan Institute for Regenerative Medicine associate director Rocky Tuan, PhD, director, Center for Cellular and Molecular Engineering, the Arthur J. Rooney, Sr. professor and executive vice chair, Department of Orthopaedic Surgery, director, Center for Military Medicine Research, and professor in the Departments of Bioengineering and Mechanical Engineering & Materials Science, University of Pittsburgh.
The Foundation for Women’s Wellness (FWW) is a 501c3 nonprofit public charity dedicated to improving women’s health by raising support for innovative early-stage research and education. Their focus is straight forward — keep FWW’s infrastructure minimal and optimize giving to go directly to research and fellowship awards.
FWW Research Awards focus on start-up funding for small, short-term studies with great promise for advancing women’s health. The FWW primarily targets leading women’s health concerns such as cardiovascular disease, top female cancers, and hormones’ role in disease and stage-of-life health concerns such as pregnancy and menopause. This start-up funding is crucial for moving women’s health forward and yet is scarce in medical funding today. Their selection process is guided by their Medical Advisory Board which emphasizes the promise of both the research and investigator to positively impact women’s health. FWW Fellowship Awards support emergent physician-scientist dedicated to women’s health research.
Regenerative Medicine Podcast Update
The Regenerative Medicine Podcasts remain a popular web destination. Informative and entertaining, these are the most recent interviews:
#167 –– Dr. Jelena Janjic is an Associate Professor of Pharmaceutics at Duquesne University. Dr. Janjic discusses her research in chronic pain.
Visit www.regenerativemedicinetoday.com to keep abreast of the new interviews.
PICTURE OF THE MONTH
The Picture of the Month is a compliment to the longstanding features Grant of the Month and Publication of the Month. Each of these features highlights the achievements of McGowan affiliated faculty and their trainees. As we have always welcomed suggestions for grants and publications, please also consider submitting images that can highlight your pioneering work.
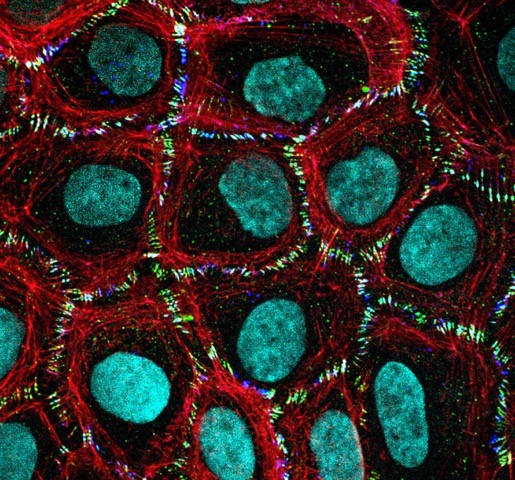
Human dermal epithelial cells, cytoskeletal extraction, stained for F-Actin (red), migfilin (green), e-cadherin (blue), and nucleus (cyan).
Cary Wu (Pathology) and Donna Stolz (CBI).
PUBLICATION OF THE MONTH
Author: Jin T, Nicholls FJ, Crum WR, Ghuman H, Badylak SF, Modo M
Title: Diamagnetic chemical exchange saturation transfer (diaCEST) affords magnetic resonance imaging of extracellular matrix hydrogel implantation in a rat model of stroke
Summary: Extracellular matrix (ECM) is widely used as an inductive biological scaffold to repair soft tissue after injury by promoting functional site-appropriate remodeling of the implanted material. However, there is a lack of non-invasive analysis methods to monitor the remodeling characteristics of the ECM material after implantation and its biodegradation over time. We describe the use of diamagnetic chemical exchange saturation transfer (CEST) magnetic resonance imaging to monitor the distribution of an ECM hydrogel after intracerebral implantation into a stroke cavity. In vitro imaging indicated a robust concentration-dependent detection of the ECM precursor and hydrogel at 1.8 and 3.6 ppm, which broadly corresponded to chondroitin sulfate and fibronectin. This detection was robust to changes in pH and improved at 37 °C. In vivo implantation of ECM hydrogel into the stroke cavity in a rat model corresponded macroscopically to the distribution of biomaterial as indicated by histology, but mismatches were also evident. Indeed, CEST imaging detected an endogenous “increased deposition”. To account for this endogenous activity, pre-implantation images were subtracted from post-implantation images to yield a selective visualization of hydrogel distribution in the stroke cavity and its evolution over 7 days. The CEST detection of ECM returned to baseline within 3 days due to a decrease in fibronectin and chondroitin sulfate in the hydrogel. The distribution of ECM hydrogel within the stroke cavity is hence feasible in vivo, but further advances are required to warrant a selective long-term monitoring in the context of biodegradation.
Source: Biomaterials. 2017 Jan;113:176-190. Epub 2016 Oct 29.
GRANT OF THE MONTH
PI: William Federspiel, William Wagner, Peter Wearden
Title: Ambulatory Assist Lung for Children
Description: Acute and chronic lung diseases remain the most life threatening causes of death and hospitalization in the pediatric population. Cystic fibrosis (CF), pulmonary hypertension and pulmonary fibrosis have been observed to be the most frequent causes of lung failure in pediatric patients. Mechanical ventilation (MV) and extracorporeal membrane oxygenation (ECMO) have been used to bridge sick kids to transplant. These procedures can lead to poor post-transplant outcomes by their very restrictive nature on mobility. This project will develop a compact respiratory assist device for pediatric patients, the Pittsburgh Pediatric Ambulatory Lung (P-PAL) to replace ECMO as a bridge to transplant or recovery in kids with acute and chronic lung failure. The P-PAL is a wearable and fully integrated blood pump and gas exchange module that will be designed for implantation of inflow cannula and outflow cannula/grafts in the right atrium and pulmonary artery, respectively. The P-PAL will be designed for longer-term respiratory support (1-3 months before cartridge change-out) at 70-90% of normal metabolic oxygenation requirements, while pumping blood from 1 to 2.5 Liters/min. The specific aims of project are 1) To modify the design and operational parameters of the P-PAL to meet requirements for blood pumping, gas exchange, priming volume, and form factor, 2) To build P-PAL prototypes along the design development pathway for bench characterization studies of pumping performance, gas exchange, and hemolysis, 3) To improve the hemocompatibility of the P-PAL by exploiting novel polymeric zwitterionic coatings that we have already begun to develop for our adult wearable assist lung, and 4) To perform acute and chronic studies in healthy lambs to demonstrate the in-vivo performance and hemocompatibility of the PAAL device and to study its interaction with the cardiopulmonary system.
Source: National Heart, Lung, and Blood Institute
Term: 01/15/2017 – 12/31/2020
Amount: $2,903,508
