October 2016 | VOL. 15, NO. 10| www.McGowan.pitt.edu
Preclinical Research: Potential Treatment of IBD
Results of a study recently published online in the Journal of Crohn’s and Colitis indicated positive results from a preclinical study to assess the effectiveness of a hydrogel for the treatment of Inflammatory Bowel Disease (IBD). The study was led by McGowan Institute for Regenerative Medicine deputy director Stephen Badylak, DVM, PhD, MD, in collaboration with Asana Medical, Inc. The study focused on effectiveness of an ExtraCellular Matrix Hydrogel (ECMH) in the treatment of IBD. Asana has a field-limited exclusive license to certain patent rights for this technology from the University of Pittsburgh.
The preclinical design leveraged earlier proof-of-concept studies. The results confirm that ECMH inhibits inflammation and promotes the rapid regeneration of healthy tissue in a rodent model of Ulcerative Colitis (UC). Specifically, the use of ECMH established:
- proof of mucosal regeneration and reduction in inflammation and ulceration;
- reduction in stool blood and weight loss;
- restoration of the cell barrier function, and;
- reduction in inflammatory biomarkers
Dr. Badylak, professor in the Department of Surgery, deputy director of the McGowan Institute, and director of the Center for Pre-Clinical Studies within the Institute, said, “The study results suggest an alternative to traditional therapies for Inflammatory Bowel Disease. The study shows the ability of ECMH to rapidly transform the inflammatory flares of UC into a healing state in this rodent model. These findings are consistent with the known mechanisms by which ECM provides constructive signals in other body tissues.”
McGowan Institute for Regenerative Medicine affiliated faculty member Neill Turner, PhD, Research Assistant Professor in Pitt’s Department of Surgery, was a member of the research team.
“Results from this study suggest a new method of potential treatment for Ulcerative Colitis,” said Steven Wexner, MD, PhD (Hon), Director of the Digestive Disease Center and Chairman of the Department of Colorectal Surgery at Cleveland Clinic Florida, and current member of Asana’s Medical Advisory Board and a paid consultant. “Traditional therapy attempts to ameliorate clinical symptoms by reducing inflammation. This approach with ECMH seems to try to provide a local scaffold to allow healing.” Additional research is planned to further assess this approach to treat IBD.
Illustration: Asana Medical, Inc.
RESOURCES AT THE MCGOWAN INSTITUTE
November Special at the Histology Lab
Reticular fibers, or reticulin is a type of fiber in connective tissue, composed of type III collagen secreted by reticular cells. Reticular fibers crosslink to form a fine meshwork (reticulin). This network acts as a supporting mesh in soft tissues such as liver, bone marrow, and the tissues and organs of the lymphatic system.
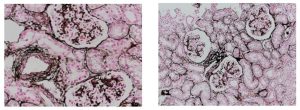
Jones Silver Stain is used to identify basement membrane and reticulin.
You’ll receive 25% off Jones Silver Stain in November when you mention this ad.
Contact Lori at the McGowan Core Histology Lab and ask about our staining specials by email or call 412-624-5265.
As always, you will receive the highest quality histology in the quickest turn-around time.
Did you know the more samples you submit to the histology lab the less you pay per sample?
Contact Lori to find out how!
SCIENTIFIC ADVANCES
In a First, Pitt-UPMC Team Help Paralyzed Man Feel Again Through a Mind-Controlled Robotic Arm

Imagine being in an accident that leaves you unable to feel any sensation in your arms and fingers. Now imagine regaining that sensation, a decade later, through a mind-controlled robotic arm that is directly connected to your brain.
That is what 28-year-old Nathan Copeland experienced after he came out of brain surgery and was connected to the Brain Computer Interface (BCI), developed by researchers at the University of Pittsburgh and UPMC. In a study published online in Science Translational Medicine, a team of experts led by Robert Gaunt, PhD, assistant professor of physical medicine and rehabilitation at Pitt, demonstrated for the first time ever in humans a technology that allows Mr. Copeland to experience the sensation of touch through a robotic arm that he controls with his brain.
“The most important result in this study is that microstimulation of sensory cortex can elicit natural sensation instead of tingling,” said study co-author McGowan Institute for Regenerative Medicine affiliated faculty member Andrew Schwartz, PhD, distinguished professor of neurobiology and chair in systems neuroscience, Pitt School of Medicine, and a member of the University of Pittsburgh Brain Institute. “This stimulation is safe, and the evoked sensations are stable over months. There is still a lot of research that needs to be carried out to better understand the stimulation patterns needed to help patients make better movements.”
This is not the Pitt-UPMC team’s first attempt at a BCI. Four years ago, study co-author Jennifer Collinger, PhD, assistant professor, Pitt’s Department of Physical Medicine and Rehabilitation, and research scientist for the VA Pittsburgh Healthcare System, and the team demonstrated a BCI that helped Jan Scheuermann, who has quadriplegia caused by a degenerative disease. The video of Ms. Scheuermann feeding herself chocolate using the mind-controlled robotic arm was seen around the world. Before that, Tim Hemmes, paralyzed in a motorcycle accident, reached out to touch hands with his girlfriend.
But the way our arms naturally move and interact with the environment around us is due to more than just thinking and moving the right muscles. We are able to differentiate between a piece of cake and a soda can through touch, picking up the cake more gently than the can. The constant feedback we receive from the sense of touch is of paramount importance as it tells the brain where to move and by how much.
For Dr. Gaunt and the rest of the research team, that was the next step for the BCI. As they were looking for the right candidate, they developed and refined their system such that inputs from the robotic arm are transmitted through a microelectrode array implanted in the brain where the neurons that control hand movement and touch are located. The microelectrode array and its control system, which were developed by Blackrock Microsystems, along with the robotic arm, which was built by Johns Hopkins University’s Applied Physics Lab, formed all the pieces of the puzzle.
In the winter of 2004, Mr. Copeland was in a car accident that snapped his neck and injured his spinal cord, leaving him with quadriplegia from the upper chest down, unable to feel or move his lower arms and legs, and needing assistance with all his daily activities.
For the trial, study co-investigator, McGowan Institute for Regenerative Medicine faculty member, and UPMC neurosurgeon Elizabeth Tyler-Kabara, MD, PhD, assistant professor, Department of Neurological Surgery, Pitt School of Medicine, implanted four tiny microelectrode arrays each about half the size of a shirt button in Nathan’s brain. Prior to the surgery, imaging techniques were used to identify the exact regions in Mr. Copeland’s brain corresponding to feelings in each of his fingers and his palm.
“I can feel just about every finger—it’s a really weird sensation,” Mr. Copeland said about a month after surgery. “Sometimes it feels electrical and sometimes its pressure, but for the most part, I can tell most of the fingers with definite precision. It feels like my fingers are getting touched or pushed.”
At this time, Mr. Copeland can feel pressure and distinguish its intensity to some extent, though he cannot identify whether a substance is hot or cold, explains Dr. Tyler-Kabara.
McGowan Institute for Regenerative Medicine affiliated faculty member Michael Boninger, MD, professor of physical medicine and rehabilitation at Pitt, and senior medical director of post-acute care for the Health Services Division of UPMC, recounted how the Pitt team has achieved milestone after milestone, from a basic understanding of how the brain processes sensory and motor signals to applying it in patients
“Slowly but surely, we have been moving this research forward. Four years ago we demonstrated control of movement. Now Dr. Gaunt and his team took what we learned in our tests with Tim and Jan—for whom we have deep gratitude—and showed us how to make the robotic arm allow its user to feel through Nathan’s dedicated work,” said Dr. Boninger, also a co-author on the research paper.
Dr. Gaunt explained that everything about the work is meant to make use of the brain’s natural, existing abilities to give people back what was lost but not forgotten.
“The ultimate goal is to create a system which moves and feels just like a natural arm would,” says Dr. Gaunt. “We have a long way to go to get there, but this is a great start.”
The lead author on the research publication is Sharlene N. Flesher, of Pitt. Additional authors on this research are Stephen T. Foldes, PhD, Jeffrey M. Weiss, and John E. Downey, all of Pitt; and Sliman J. Bensmaia, PhD, of the University of Chicago.
Primary support for the study was provided by the Defense Advanced Research Projects Agency’s (DARPA) Revolutionizing Prosthetics program through contract N66001-10-C-4056. Additional support was provided by the Office of Research and Development, Rehabilitation Research and Development Service, U.S. Department of Veterans Affairs, grant numbers B6789C, B7143R, and RX720, and the National Science Foundation Graduate Research Fellowship grant DGE-1247842.
Illustration: UPMC.
Pitt Scientists Identify How Repair Protein Finds DNA Damage

Researchers at the University of Pittsburgh School of Medicine and University of Pittsburgh Cancer Institute (UPCI)—including McGowan Institute for Regenerative Medicine affiliated faculty member Simon Watkins, PhD, founder and director of the Center for Biologic Imaging at the University of Pittsburgh—have demonstrated how Rad4, a protein involved in DNA repair, scans the DNA in a unique pattern of movement called “constrained motion” to efficiently find structural faults in DNA. The findings, reported recently in the journal Molecular Cell, could lead to therapies that boost existing drug treatments and counter drug-resistance.
“Rad4 is like the cop who is the first responder at an accident,” said senior author Bennett Van Houten, PhD, Richard M. Cyert Professor of Molecular Oncology, Pitt School of Medicine, and co-leader of UPCI’s Molecular and Cellular Cancer Biology Program. “The cop can move quickly to recognize where the incident is, and regulate traffic while directing the paramedics arriving in an ambulance.”
Constrained motion allows Rad4 to be fast enough to scan large lengths of DNA quickly yet slow enough that it does not miss structural errors in DNA that could be caused by chemicals or ultraviolet (UV) light.
Mutations in Rad4, called XPC in humans, and other proteins in the DNA repair machinery are known to cause a genetic condition called xeroderma pigmentosum, where individuals have sensitivity to sunlight and are at an extremely high risk for developing skin cancer.
Muwen Kong, a graduate student in Dr. Van Houten’s laboratory, along with his collaborators, tagged normal and mutant Rad4 molecules with light-emitting quantum dots. They then watched them move across strands of DNA suspended between beads using a fluorescence microscope.
The results obtained suggest that the first responder, consisting of Rad4 and another protein, Rad23, quickly scans the DNA for accidents by attempting to bend it. Alterations in the structure of DNA, such as those caused by chemicals or UV light, change the ease with which DNA can be bent. Once a potential accident is recognized, the Rad4-Rad23 first-responder team slows down to a ‘constrained motion’ pattern to more carefully examine a smaller region of 500-1,000 base pairs in the DNA.
When structural damage is confirmed, Rad4-Rad23 stays near the scene and flags down the ‘paramedics,’ comprised of the rest of the DNA repair machinery, to fix the damage. This mechanism, which Dr. Van Houten calls ‘recognition-at-a-distance,’ allows Rad4 to be near the error without impeding the rest of the DNA repair crew.
Though much work is needed before these results can be translated to the clinic, the results provide new avenues to improve treatment methods, especially in cancer. Resistance is a major problem with current treatments, such as the drug cisplatin, which kills cancer cells by introducing DNA crosslinks similar to UV light. By developing drugs that target Rad4/XPC or other repair proteins, it could be possible to enhance the effects of current treatments when they are used together, and also reduce the chances of tumor cells developing resistance, Dr. Van Houten said.
Activity Trackers Are Ineffective at Sustaining Weight Loss
 Wearable devices that monitor physical activity are not reliable tools for weight loss, says a new study from the University of Pittsburgh School of Education’s Department of Health and Physical Activity. McGowan Institute for Regenerative Medicine affiliated faculty member Steven Belle, PhD, MScHyg, Professor in the Department of Epidemiology, Graduate School of Public Health at the University of Pittsburgh and a Co-Director in the Epidemiology Data Center in the Graduate School of Public Health, was a member of the research study team. The study specifically investigated whether regular use of commercially available activity trackers is effective for producing and sustaining weight loss.
Wearable devices that monitor physical activity are not reliable tools for weight loss, says a new study from the University of Pittsburgh School of Education’s Department of Health and Physical Activity. McGowan Institute for Regenerative Medicine affiliated faculty member Steven Belle, PhD, MScHyg, Professor in the Department of Epidemiology, Graduate School of Public Health at the University of Pittsburgh and a Co-Director in the Epidemiology Data Center in the Graduate School of Public Health, was a member of the research study team. The study specifically investigated whether regular use of commercially available activity trackers is effective for producing and sustaining weight loss.
At the conclusion of a 24-month trial, researchers observed that usage of a wearable device in combination with a behavioral weight loss program resulted in less weight loss when compared to those receiving only the behavioral weight loss program. In fact, participants without physical activity trackers showed nearly twice the weight loss benefits at the end of the 24 months. Participants who utilized wearable devices reported an average weight loss of 7.7 pounds, while those who partook only in health counseling reported an average loss of 13 pounds.
Through these observations, researchers concluded that devices that monitor and provide feedback on physical activity do not offer an advantage over standard weight loss approaches that include behavioral counseling on physical activity and diet. Thus, while these devices allow for ease of tracking of physical activity along with feedback and encouragement, they may not enhance adherence to the tenets of a healthy lifestyle, which is the most important aspect of any weight loss regiment.
“While usage of wearable devices is currently a popular method to track physical activity—steps taken per day or calories burned during a workout—our findings show that adding them to behavioral counseling for weight loss that includes physical activity and reduced calorie intake does not improve weight loss or physical activity engagement. Therefore, within this context, these devices should not be relied upon as tools for weight management in place of effective behavioral counseling for physical activity and diet,” said John Jakicic, PhD, the study’s lead researcher and chair of Pitt’s Department of Health and Physical Activity.
The study, “Effect of Wearable Technology Combined with a Lifestyle Intervention on Long-Term Weight Loss: the IDEA Randomized Clinical Trial,” was released recently in JAMA: The Journal of the American Medical Association.
“The findings of our study are important because effective long-term treatments are needed to address America’s obesity epidemic,” said Dr. Jakicic. “We’ve found that questions remain regarding the effectiveness of wearable devices and how to best use them to modify physical activity and diet behaviors in adults seeking weight loss.”
Illustration: Griffin.
Grant Received from Hillman Foundation to Advance Brain Research

A lead gift of $600,000 from the Henry L. Hillman Foundation will help the University of Pittsburgh Brain Institute (UPBI) launch groundbreaking interdisciplinary projects to advance research on normal brain function and its impairment in a range of disorders. With matching funds from internal sources, the UPBI is distributing a total of $925,000 to five projects managed by Pitt faculty that address paralysis, Parkinson’s disease, adolescent substance use, obsessive compulsive disorder, and brain computation. The funding also will support the growth of a UPBI Brain Bank to further research. Two of the selected projects will be managed by McGowan Institute for Regenerative Medicine affiliated faculty members:
- Enhanced Neural Prosthetics Using Shared-Mode Control: This project builds on the world’s most advanced program in brain-controlled robotic arms and hands for paralyzed individuals. Led by Andrew Schwartz, PhD, distinguished professor of neurobiology at Pitt, a group of Pittsburgh scientists and engineers will enhance the performance of neural prosthetics, allowing a paralyzed person to manipulate an object. A prosthetic limb will operate under an individual’s brain control, with a boost from an artificial intelligence component designed to predict what the individual intends to do. This shared-mode control will enable people who have quadriplegia to dexterously handle objects with a robotic arm and hand, and thus increase their independence in daily life.
- The Impact of Variability on Neuronal Processing: Neurons are an unpredictable lot, varying their response from moment to moment within any given experiment. They also generate large amounts of data. Coping with such large-scale data and characterizing variability across brain regions will be essential to constructing models of how the brain processes sensory signals, and ultimately for developing an overarching theory of cognition. Through their work, Brent Doiron, PhD, associate professor of mathematics, and Matthew Smith, PhD, assistant professor of ophthalmology, both of Pitt, will aim to understand the functional consequences of variability on specific brain computations and the behaviors that they generate.
“Collectively, these projects have the potential to significantly advance our understanding of brain function and shed light on disorders that affect millions of people. New therapies are urgently needed, and the road to these cures begins with this kind of fundamental research,” said Peter Strick, PhD, scientific director of the UPBI, who also is a distinguished professor and holds the Thomas Detre Endowed Chair in the Pitt School of Medicine Department of Neurobiology. “These projects will enable us to better frame the national research agenda and compete more effectively for large-scale federal funding opportunities.”
Pitt Team Receives $2.5 million in DOD Funds for First Retrievable Vascular Stent

Researchers at the UPMC Division of Vascular Surgery and the University of Pittsburgh Swanson School of Engineering have been awarded a 4-year, $2.5 million contract from the U.S. Department of Defense (DOD) for further development of a retrievable stent to treat noncompressible hemorrhages, a major cause of mortality among servicemen and women, as well as civilian gunshot victims.
“A well-known principle of first aid for bleeding is to apply pressure to the bleeding site,” said McGowan Institute for Regenerative Medicine faculty member Bryan Tillman, MD, PhD, assistant professor of vascular surgery at Pitt’s School of Medicine, and principal investigator on the project. “Unfortunately, for injuries within the chest and abdomen, this maneuver is not effective, and death rates exceed 80 percent due to massive bleeding in a very short time.”
The proposal, “A Rapid, Temporary Stent for Hemorrhagic Injuries of the Torso,” allows for refinement of a novel stent that can be rapidly placed by non-vascular physicians with minimal training. The system uses radiofrequency tags similar to the microchips used to identify pets. A handheld device is used by physicians to help place the tagged stent in a blood vessel, simplifying positioning and replacing bulky and often unavailable X-ray equipment.
“The absence of immediate vascular expertise and X-ray imaging on the battlefield or even in some hospitals remains a major obstacle to treat hemorrhages,” Dr. Tillman said. “What is needed is a way to rapidly control massive bleeding until a patient can be transported to a proper medical facility or to a properly equipped vascular hybrid room.”
As the first fully retrievable vascular stent, the proposed device can be removed at the time of permanent repair by skilled vascular surgeons with dedicated imaging equipment. A prototype of the stent was developed and successfully tested by UPMC as a part of the DOD application for funding.
“This device will be used to save the lives of critically wounded soldiers until they can be transported to a major medical facility,” Dr. Tillman said. “We expect that this same technology also will be used in trauma bays when vascular expertise or state-of-the-art hybrid operating suites are not immediately available to rescue civilian patients with life-threatening traumatic injuries of the liver and large vessels.”
Researchers Receive NIH Grant to Develop Better Methodology to Treat Rotator Cuff Tears

Rotator cuff tears are one of the most common injuries seen by orthopedic surgeons, resulting in 30 percent of all visits to orthopaedic surgeons and over 150,000 surgical procedures per year in the United States. The preferred initial treatment is 6 to 12 weeks of physical therapy (PT), but 25-50 percent of those cases still require surgery. Researchers at the University of Pittsburgh’s Swanson School of Engineering recently received a $2.79 million award from the National Institutes of Health to develop diagnostic methods to allow surgeons to determine whether PT or surgery is the most effective initial treatment.
Principal investigator of the 5-year study, “Predicting the Outcome of Exercise Therapy for Treatment of Rotator Cuff Tears,” is McGowan Institute for Regenerative Medicine affiliated faculty member Richard Debski, PhD, associate professor of bioengineering and co-director of the Orthopaedic Robotics Laboratory at Pitt. Co-principal investigators are James J. Irrgang, PhD, professor and chair of the Department of Physical Therapy in Pitt’s School of Health and Rehabilitation Sciences and vice chair of clinical outcomes research in the Department of Orthopaedic Surgery; and Scott Tashman, PhD, professor of orthopaedic surgery at the University of Texas Health Science Center at Houston.
“Rotator cuff injuries are one of the most common injuries for people aged 40-70, and can be caused by an injury but often occur simply from wear and tear as we age,” Dr. Debski explained. “Over the age of 50, chances increase that 40-50 percent of people have a tear and many don’t know it. Although physical therapy is the first preferred treatment, most patients still require surgery, which prolongs recovery time and increases costs. Our goal is to utilize new methods to perform a biomechanical analysis to determine whether a patient is more suited for PT or surgery, and thereby improve overall recovery.”
Over the first 2 years, Dr. Debski and his group plan to enroll 100 patients with isolated full thickness tears of the supraspinatus tendon – the most basic tear. The biomechanics analysis will measure shoulder motion and tear size before and after physical therapy. He noted the technology that they will use, a bi-planar x-ray system that Dr. Tashman developed, is unique in such a study. The x-ray images provide quantitative measurements of shoulder motion during activities of daily living and are representative of rotator cuff function. The group will also track the tear size longitudinally out to 1 year, a first for such a study. Their long-term goal is to perform a clinical trial to determine whether the predictions make a difference in treatment outcomes.
“Determining the characteristics of treatment versus surgery will be critical, but for the first time we’ll be gathering comprehensive, quantitative data to make these predictions,” he said. “The genesis of this study began in 2011 with Volker Musahl, MD, medical director of the UPMC Rooney Center for Sports Medicine and orthopaedic surgeon for Pitt’s football team. Our rotator cuff research has now grown to include bioengineering, radiology, physical therapy, and orthopaedic surgery, which is a tremendous interdisciplinary effort to attack this problem.”
Illustration: Biodynamics Laboratory, Department of Orthopaedic Surgery, University of Pittsburgh.
World-Renowned Expert in Research and Therapies for Blindness and Vision Impairment Joins McGowan Institute

One of the world’s top experts in retinal diseases, who is developing an artificial retina as well as other regenerative therapies to treat blindness and vision impairments, has been named as the chair of the Department of Ophthalmology at the University of Pittsburgh School of Medicine, director of the UPMC Eye Center, and the Eye and Ear Foundation Chair of Ophthalmology.
José-Alain Sahel, MD, founder and director of the Vision Institute in Paris and currently a professor at the Sorbonne’s medical school Université Pierre-et-Marie-Curie, has joined UPMC and Pitt’s faculty. Dr. Sahel will also serve as an affiliated faculty member of the McGowan Institute for Regenerative Medicine.
Dr. Sahel is known worldwide for his expertise in vision restoration techniques. He has developed several interventions— including stem cell implantation, gene therapy, innovative pharmacologic approaches, and the artificial retina—for retinitis pigmentosa, age-related macular degeneration, vascular eye disease, and other vision impairments that currently are untreatable. Over the past decade he has led pioneering efforts in optogenetic vision restoration, a technique in which cells in the retina are genetically modified to express light sensitive proteins. This therapeutic technique has the potential to help patients who are blind or visually impaired as a result of a genetic defect.
He also brings a strong neuroscience perspective to ophthalmology research, such as exploring the application of brain-computer interface technology, for which Pitt and UPMC are well known.
Dr. Sahel will sustain his engagement with the Vision Institute, one of the world’s largest centers of integrated research on eye diseases, through a partnership formed between the Institute, the Sorbonne, Pitt School of Medicine, and UPMC.
As director of UPMC Eye Center, which is rated among the top ophthalmology programs in the United States in the delivery of care, Dr. Sahel will see patients, particularly those with complex diseases, and will lead UPMC clinicians in the diagnosis and treatment of both common and unusual eye disorders. The UPMC Eye Center is home to an outstanding ophthalmology teaching program that trains residents and clinical fellows in their pursuit of clinical, research, and clinician-scientist careers.
Dr. Sahel is a co-inventor on more than 40 patents, several of which have led to start-up companies including Fovéa Inc., which Dr. Sahel founded and later became the Ophthalmologic Division of Sanofi Aventis. He also is a scientific co-founder of GenSight Biologics Inc. and Pixium Vision Inc. He is a member of 11 editorial boards, including the Journal of Clinical Investigation and Science Translational Medicine.
He has been the recipient of numerous honors and awards, which include membership in the Legion of Honour (2008), the National Order of Merit (2002), and induction into the Collège de France, the highest honor awarded to a French scientist. He is an elected member of the Académie des sciences-Institut de France, the Académie des technologies, the Academia Ophthalmologia Internationalis, and the German National Academy of Sciences. He has also received the Grand Prix for Neurosciences of the Foundation NRJ, Institut de France; the CNRS Medal of Innovation; the Foundation for Fighting Blindness Trustee Award; and the Liura Liggett-Gund Award, among others. Dr. Sahel holds an honorary degree from the University of Geneva.
Illustration: UPMC.
AWARDS AND RECOGNITION
Brown Lab Students Take 2nd Place in Michael G. Wells Student Healthcare Entrepreneurship Competition

The Michael G. Wells Competition is sponsored by the Michael G. Wells Entrepreneurial Scholars Fund, and is for University of Pittsburgh students who are developing innovations related to the healthcare field.
Second place in the 2016 competition went to a team from the lab of McGowan Faculty member Bryan Brown, PhD. See the video of Dan Hachim and Aimon Iftikhar presenting their technology “LayerUp,” a nanoscale immunomodulatory coating system for enhancing biomaterial integration and long term performance. A paper describing this work was just published in Biomaterials (“Shifts in macrophage phenotype at the biomaterial interface via IL-4 eluting coatings are associated with improved implant integration”; Daniel Hachim, Samuel T. LoPresti, Cecelia C. Yates, Bryan N. Brown). The team was selected as one of seven finalist groups from 35 initial applicants. They won 2nd place ($10,000) based upon their pitch to a group of local VC’s, consultants, and Michael Wells. The prize was awarded in front of a large crowd at SCIENCE 2016.
Students (individually or as a team) were given the opportunity to present their ideas at the “First Look” Technology Showcase in October 2016, and compete for the $20,000 Michael G. Wells Entrepreneurial Scholars Award to help move their idea towards commercialization. A $10,000 second place and $6,000 third place were also awarded. The “First Look” Technology Showcase is an annual event hosted by the Innovation Institute that provides University innovators the opportunity to present their ideas to an audience of investors, entrepreneurs, regional economic development representatives, local business executives and Pitt alumni. Details about the program are available here.
Dr. Billiar Receives Highest Honor from Chinese Government

Timothy Billiar, MD, George Vance Foster Professor and chair in the Department of Surgery at the University of Pittsburgh School of Medicine, associate medical director UPMC International, and McGowan Institute for Regenerative Medicine faculty member has received the Friendship Award, the highest recognition bestowed by the People’s Republic of China to foreign experts who have made outstanding contributions to the country’s economic and social progress.
Dr. Billiar was nominated by Central South University in Changsha, Hunan, China for his work in developing student and physician exchange programs and for his role in developing a Chinese medical student research training program at the Pitt School of Medicine. Every year, 50 foreign experts are selected for the Friendship Award. This year, recipients came from 18 foreign countries and included experts in the fields of industry, science and technology, medicine, agriculture, energy, environmental protection, and education.
“Our collaboration with colleagues in China has been of great mutual benefit as we work to advance medical science. I am truly honored to receive this recognition from the Chinese government,” Dr. Billiar said.
He traveled to Beijing to accept the award from the State Administration of Foreign Experts Affairs at a ceremony hosted by Vice-Premier Ma Kai on Sept. 29 at Zhongnanhai, the seat of government in China.
In addition to Pitt’s academic partnerships, UPMC International currently works with several partners in China to provide world-class care close to patients’ homes, including second-opinion telepathology services for KingMed Diagnostics, operational support for Xiangya International Medical Center in Changsha, and development of oncology services on Hainan Island in collaboration with First Chengmei Medical Industry Group.
Dr. Brown Laboratory Student Accepted Into CTS TL1 Program

Aimon Iftikhar, graduate student researcher in the Department of Bioengineering at the University of Pittsburgh and in the laboratory of McGowan Institute for Regenerative Medicine faculty member Bryan Brown, PhD, assistant professor in the Department of Bioengineering with a secondary appointment in the Department of Obstetrics, Gynecology, and Reproductive Sciences at the University of Pittsburgh, has been accepted for appointment as a Pre-doctoral Fellow in the Clinical and Translational Science (CTS) Fellowship TL1 program. Ms. Iftikhar began her initial 12-month project period on October 1, 2016.
The TL1 CTS Fellowship is offered by the Institute for Clinical Research Education (ICRE) and the Clinical and Translational Science Institute (CTSI). It provides rigorous translational research training to qualified pre- and post-doctoral students who are able to effectively create innovative ways to advance research from the initial discovery through improved patient outcomes and health policy. The overarching goal of this program is to speed the translation of discoveries and improve the efficiency with which these discoveries are implemented in practice.
Congratulations, Ms. Iftikhar!
Regenerative Medicine Podcast Update
The Regenerative Medicine Podcasts remain a popular web destination. Informative and entertaining, these are the most recent interviews:
#164 –– Dr. William Federspiel is the William Kepler Whiteford Professor in the Department of Bioengineering with secondary appointments in Chemical Engineering and Critical Care Medicine. He is also the Director of the Medical Devices Laboratory at the McGowan Institute. Dr. Federspiel discusses his research in the design and development of novel artificial lung devices.
Visit www.regenerativemedicinetoday.com to keep abreast of the new interviews.
PICTURE OF THE MONTH
The Picture of the Month is a compliment to the longstanding features Grant of the Month and Publication of the Month. Each of these features highlights the achievements of McGowan affiliated faculty and their trainees. As we have always welcomed suggestions for grants and publications, please also consider submitting images that can highlight your pioneering work.
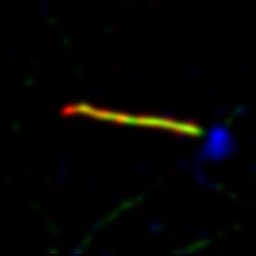
Super-resolution (Stimulated Emission Depletion, STED) image of a primary cilium. Blue is gamma-tubulin, green is acetylated tubulin (cilia cytoskeleton) and red is Arl13b (ciliary membrane). Scale: 20 nm/pixel. Super-resolution capabilities available at the Center for Biologic Imaging.
Image from Tim Feinstein and Cecilia Lo, Developmental Biology
PUBLICATION OF THE MONTH
Author: Abboud A, Mi Q, Puccio A, Okonkwo D, Buliga M, Constantine G, Vodovotz Y
Title: Inflammation Following Traumatic Brain Injury in Humans: Insights from Data-Driven and Mechanistic Models into Survival and Death.
Summary:
Inflammation induced by traumatic brain injury (TBI) is a complex mediator of morbidity and mortality. We have previously demonstrated the utility of both data-driven and mechanistic models in settings of traumatic injury. We hypothesized that differential dynamic inflammation programs characterize TBI survivors vs. non-survivors, and sought to leverage computational modeling to derive novel insights into this life/death bifurcation. Thirteen inflammatory cytokines and chemokines were determined using Luminex™ in serial cerebrospinal fluid (CSF) samples from 31 TBI patients over 5 days. In this cohort, 5 were non-survivors (Glasgow Outcome Scale [GOS] score = 1) and 26 were survivors (GOS > 1). A Pearson correlation analysis of initial injury (Glasgow Coma Scale [GCS]) vs. GOS suggested that survivors and non-survivors had distinct clinical response trajectories to injury. Statistically significant differences in interleukin (IL)-4, IL-5, IL-6, IL-8, IL-13, and tumor necrosis factor-α (TNF-α) were observed between TBI survivors vs. non-survivors over 5 days. Principal Component Analysis and Dynamic Bayesian Network inference suggested differential roles of chemokines, TNF-α, IL-6, and IL-10, based upon which an ordinary differential equation model of TBI was generated. This model was calibrated separately to the time course data of TBI survivors vs. non-survivors as a function of initial GCS. Analysis of parameter values in ensembles of simulations from these models suggested differences in microglial and damage responses in TBI survivors vs. non-survivors. These studies suggest the utility of combined data-driven and mechanistic models in the context of human TBI.
Source: Front Pharmacol. 2016 Sep 27;7:342. eCollection 2016.
GRANT OF THE MONTH
PI: Andrew Schwartz
Title: Enhanced Neural Prosthetics Using Shared-Mode Control
Description:
This project builds on the world’s most advanced program in brain-controlled robotic arms and hands for paralyzed individuals. A group of Pittsburgh scientists and engineers will enhance the performance of neural prosthetics, allowing a paralyzed person to manipulate an object. A prosthetic limb will operate under an individual’s brain control, with a boost from an artificial intelligence component designed to predict what the individual intends to do. This shared-mode control will enable people who have quadriplegia to dexterously handle objects with a robotic arm and hand, and thus increase their independence in daily life.
Source: Henry L. Hillman Foundation
