May 2016 | VOL. 15, NO. 5| www.McGowan.pitt.edu
Pitt Innovation Institute 11th Annual Celebration of Innovation Honors McGowan Institute Affiliated Faculty Members

The University of Pittsburgh Innovation Institute honored Pitt Innovators whose inventions were recently licensed at its 11th Annual Celebration of Innovation. Also recognized were Pitt faculty, students, and staff who received patents or submitted invention disclosures since July 1, 2014.
The McGowan Institute congratulates those innovative faculty, staff, post-docs, graduate students, and undergraduate students who were recognized by the Innovation Institute.
Those honored include:
2016 Academic Year Innovator Award Winners
- Stephen Badylak, DVM, PhD, MD
- Xinyan Tracy Cui, PhD
- Rory Cooper, PhD with Dan Ding, MD
- Louis Falo, MD, PhD
- Paulo Fontes, MD, FACS
- John Kellum, MD, FACP, FCCM
- Kacey Marra, PhD
- Rocky Tuan, PhD
2016 Academic Year Patents
- Steven Little, PhD
- Charles Sfeir, DDS, PhD
- David Vorp, PhD
- William Wagner, PhD
- Yadong Wang, PhD
- Douglas Weber, PhD
2016 Academic Year Invention Disclosures
- Elia Beniash, PhD
- David Brienza, PhD
- Bryan Brown, PhD
- Xinyan Tracy Cui, PhD
- William Federspiel, PhD
- Thomas Gleason, MD
- Marina Kameneva, PhD
- Prashant Kumta, PhD
- Steven Little, PhD
- Julie Phillippi, PhD
- Charles Sfeir, DDS, PhD
- Rocky Tuan, PhD
- David Vorp, PhD
- William Wagner, PhD
- Justin Weinbaum, PhD
During the 20 months of accomplishments celebrated, Pitt Innovators overall have:
- Had 185 inventions licensed or optioned
- Received 115 patents
- Submitted 480 invention disclosures
The University has also witnessed more than 120 startup companies launched based on Pitt-developed inventions since the Innovation Institute and its predecessor offices were established in 1996.
“Pitt has a long history of fostering innovation and entrepreneurship. The Pitt Innovators we are honoring at the Celebration of Innovation are emblematic of our forward momentum as we seek to strengthen that culture,” said Innovation Institute Founding Director Marc Malandro.
Congratulations to all!
Illustration: Innovation Institute.
RESOURCES AT THE MCGOWAN INSTITUTE
June Special at the Histology Lab
There are two ways that a cell can die: necrosis and apoptosis. Necrosis occurs when a cell is damaged by an external force, such as poison, a bodily injury, an infection or loss of the blood supply (which might occur during a heart attack or stroke).
Apoptosis, on the other hand, is when a cell commits suicide. It’s also referred to as “programmed cell death.”
Apoptosis is a form of cell death designed to eliminate compromised or senescenet cells. Apoptosis is controlled by multiple signaling and effector pathways that in turn, are activated in response to external growth, survival, or death factors.
A method for examining apoptosis via DNA fragmentation is the TUNEL assay. This technique can detect early-stage apoptosis in systems where chromatin condensation has begun and strand breaks are fewer, even before the nucleus undergoes recognizable morphologic changes.
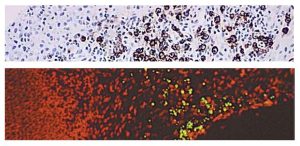 The McGowan Histology Core offers Tunel staining on paraffin embedded or frozen tissue. Light microscopy or fluorescent staining is also available.
The McGowan Histology Core offers Tunel staining on paraffin embedded or frozen tissue. Light microscopy or fluorescent staining is also available.
The Histology Core Laboratory is offering 30% off TUNEL staining for the month of June, when mentioning this ad. Contact Lori at the McGowan Core Histology Lab and ask about our staining specials. Email perezl@upmc.edu or call 412-624-5265. As always, you will receive the highest quality histology in the quickest turn-around time.
Did you know the more samples you submit to the histology lab the less you pay per sample? Contact Lori to find out how!
Flow Cytometry Helps Hone Treatment for Sepsis
Flow cytometry is a laser-based, biophysical technology employed in cell counting, cell sorting, biomarker detection, and protein engineering, which achieves the desired outcome by suspending cells in a stream of fluid and passing them by an electronic detection apparatus. It allows simultaneous multiparametric analysis of the physical and chemical characteristics of up to thousands of particles per second.
Flow cytometry is routinely used in the diagnosis of health disorders, especially blood cancers, but has many other applications in basic research, clinical practice, and clinical trials. A common variation is to physically sort particles based on their properties, so as to purify populations of interest.
Alexander Malkin, a graduate student researcher in the laboratory of McGowan Institute for Regenerative Medicine faculty member William Federspiel, PhD, recently worked with Lynda Guzik, McGowan Institute Flow Cytometry Laboratory Manager, to develop novel treatments for sepsis and other inflammatory conditions using dialysis-like medical devices to filter or modulate patient’s blood. Mr. Malkin explains how his experience with the Flow Cytometry Lab was instrumental in his investigations:
Sepsis is a severe inflammatory response to infection that affects nearly 1 million Americans per year. Despite improvements in care, hospital mortality from sepsis remains high and is currently nearly 20%. Sepsis is now the leading cause of in-hospital death in the United States, yet there are currently no FDA-approved specific treatments for sepsis. The Medical Devices Laboratory, headed by Dr. Federspiel, works to develop novel treatments for sepsis and other inflammatory conditions using dialysis-like medical devices to filter or modulate patient’s blood. The NeRD (Neutrophil Reprogramming Device) is able to reprogram overactive white blood cells, which cause organ damage in acute inflammatory conditions, to treat septic patients.
Development of any medical device requires rapid iteration of the device design and construction. The Medical Devices Laboratory has relied on support from the McGowan Institute Flow Cytometry Laboratory to evaluate and optimize development of the NeRD. Specifically, flow cytometry is used to measure the change in relevant surface receptor expression on activated neutrophils, the most prevalent white blood cells, after recirculation through scaled devices. These readings indicate the neutrophil’s level of activation and their likelihood of causing organ damage in septic patients. Multiparametric analysis allows for identification of two surface receptors CXCR-1 and CXCR-2 in addition to surface marker CD-15, to identify neutrophils from other white blood cells. By collecting these values over time, device performance can be quantified. These readings have helped optimize the NeRD design and are accelerating the technology’s transition from bench to bedside.
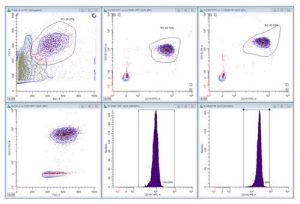
Figure 1: Whole blood is sampled from the device reservoir after recirculation. Red blood cells are lysed and remaining white blood cells are fixed with formaldehyde. The neutrophils population is selected for analysis by size, granularity, and CD-15+ signal. These cells are then analyzed for CXCR-1 (APC) and CXCR-2 (PE) surface expression.
The McGowan Institute for Regenerative Medicine Flow Cytometry Laboratory is ready to help YOU with your research efforts. Do you need to: Purify samples? Characterize complex samples? Define a rare subpopulation? Examine cellular function? Study proliferation or apoptosis? Design a flow experiment and need help? State-of-the-art Flow Cytometry is available to ALL researchers across the University of Pittsburgh campus. Instrumentation and experienced staff are available to assist users in the understanding, development, and usage of multi-parameter flow cytometry in order to further their research. Contact Lynda Guzik, the Flow Cytometry Lab Manager, for more information: 412-648-8660 or guzilj@upmc.edu.
UPCOMING EVENTS
Save the Date: Regenerative Rehabilitation Advanced Training Course
 The University of Pittsburgh and Magee-Womens Research Institute is hosting the NIH-sponsored Rehabilitative and Regenerative Medicine for Minority Health and Health Disparities Advanced Training Course from June 6-10, 2016. The purpose of this NICHD Continuing Education Training Program is to provide comprehensive and sophisticated training and state-of-the-art methods on bioengineering, cellular, molecular and genetic approaches for advancing the fields of Rehabilitative and Regenerative Medicine. Program organizers include Drs. Gerald Schatten, Fabrisia Ambrosio, and Michael Boninger. Researchers and clinicians from around the country have been selected to present during this week-long course, including Pioneer Award Lecturer, Dr. Donald Ingber from the Wyss Institute for Biologically Inspired Engineering at Harvard University.
The University of Pittsburgh and Magee-Womens Research Institute is hosting the NIH-sponsored Rehabilitative and Regenerative Medicine for Minority Health and Health Disparities Advanced Training Course from June 6-10, 2016. The purpose of this NICHD Continuing Education Training Program is to provide comprehensive and sophisticated training and state-of-the-art methods on bioengineering, cellular, molecular and genetic approaches for advancing the fields of Rehabilitative and Regenerative Medicine. Program organizers include Drs. Gerald Schatten, Fabrisia Ambrosio, and Michael Boninger. Researchers and clinicians from around the country have been selected to present during this week-long course, including Pioneer Award Lecturer, Dr. Donald Ingber from the Wyss Institute for Biologically Inspired Engineering at Harvard University.
This dynamic training course provides a series of daily lecturers on emerging concepts, followed by extended discussion, laboratory research, technologically intense workshops and informal seminars over a week-long period. The primary aim is to educate and update rehabilitation and regeneration researchers on the implications of stem cells and tissue engineering for mechanistic discoveries and on designing improved strategies for rehabilitation discoveries, especially in the CNS and skeletal-muscular systems. This course provides an innovative educational opportunity to motivate biomedical trainees to pursue scientific careers in rehabilitation and regenerative research research.
The target audience is young and promising faculty, as well as advanced postdoctoral fellows eager to learn about the newest findings and controversies. We intend to give participants knowledge of laboratory techniques, career mentoring and the ethical-legal-societal impact of rehabilitative and regenerative research to greatly enhance their successful entry into this field or to strengthen their knowledge in this field.
For more information, contact pdc@mwri.magee.edu or call 412-641-2400.
Link http://www.AR3T.pitt.edu/atc
SCIENTIFIC ADVANCES
Research Grant Received: Use of Cells from Patient’s Own Fat as Vascular Grafts in Arterial Bypass Surgery

The National Institutes of Health has awarded McGowan Institute for Regenerative Medicine faculty member David Vorp, PhD, the William Kepler Whiteford Professor of Bioengineering and Associate Dean for Research of the Swanson School of Engineering at Pitt, with a 2-year, $417,838 R21 grant for research into the use of cells from a patient’s own adipose (fat) as vascular grafts in arterial bypass surgery. This new method, which has been successful in rat subjects, would allow surgeons to perform bypass surgeries without harvesting arteries or veins from the patient or requiring the time to isolate and grow a specific cell type, such as a stem cell.
Coronary bypass procedures often use other arteries or veins as a source for grafts. Arteries that can be used safely as a bypass graft in a different location are in short supply. The great saphenous vein in the leg is one of the most common sources for arterial grafts, but re-purposing veins as arterial bypass grafts can cause complications.
“The vein graft, even though it is the most widely used graft material for the coronary or other small diameter artery applications, is not ideal,” said Dr. Vorp. “The problem is that veins are not arteries; they are built differently because they have different purposes in the body. Arteries pulse, and they are under higher pressure than veins. When you take a vein segment and put it under arterial conditions, it responds by thickening, which can cause the same blockage you were trying to treat.”
Dr. Vorp’s study, “An Autologous, Culture-free Adipose Cell-based Tissue Engineered Vascular Graft,” will explore ways to facilitate the translation of technology that has been under development by Dr. Vorp and his associates for a number of years. The current methodology requires stem cells to be carefully extracted from the fat and then spend time in culture before constructing a graft, which itself spends time in additional culture. This new research will explore the potential for skipping the culture steps entirely, first by using all of the fat cells from the patient (instead of isolating and expanding the stem cells alone) and then by implanting the graft immediately instead of culturing it first. The Pitt researchers will also design a way to “scale-up” the process that creates their engineered graft. The method used for small-scale grafts in rats won’t work when the construct is enlarged to a human-scale.
“The key focus of the study is the translational aspects,” said Dr. Vorp. “We have shown that we can regenerate a small-diameter aorta in a rat that functions for up to a year. We now need to determine how to overcome some logistical issues so that we can use this technology to help human patients, which is why we started the research in the first place. This R21 grant is really facilitating the start of that process.”
R21 research grants are designated for exploratory/developmental research, generally still in the conceptual stage. They are often awarded to high risk and high reward studies that have the potential to become much larger in scope. McGowan Institute faculty members J. Peter Rubin, MD, University of Pittsburgh Medical Center Endowed Professor, Chair of Plastic Surgery and Professor of Bioengineering, as well as William Wagner, PhD, director of McGowan Institute and Professor of Surgery, Bioengineering, and Chemical Engineering, will collaborate with Dr. Vorp on the study.
“This grant is another manifestation of the great environment here at Pitt where you have no walls between engineering, science, and clinical medicine. Research is really well integrated, and partnerships are encouraged,” said Dr. Vorp.
Voice of America Features Trilogy of McGowan Institute Research Programs
George Putic, Science and Technology Reporter, Voice of America, recently highlighted the research programs of three McGowan Institute for Regenerative Medicine affiliated faculty members. Featured were the on-going efforts in the laboratories of Stephen Badylak, DVM, PhD, MD; Ronald Poropatich, MD; and Paulo Fontes, MD.
Scientists Build Human Tissue on Animal Matrix
 The question has always been, if a gecko can grow back its tail, why can’t we regenerate our lost body parts? Well, maybe we can, someday. Scientists are moving towards the ability to rebuild fully functioning organs, and have made significant progress replacing muscles and other tissue.
The question has always been, if a gecko can grow back its tail, why can’t we regenerate our lost body parts? Well, maybe we can, someday. Scientists are moving towards the ability to rebuild fully functioning organs, and have made significant progress replacing muscles and other tissue.
The use of mammalian extracellular matrix (ECM) or its derivatives as an inductive template for constructive remodeling of tissue is a common theme of most research activities in the laboratory of McGowan Institute for Regenerative Medicine deputy director Stephen Badylak, DVM, PhD, MD, professor in the Department of Surgery and director of the Center for Pre-Clinical Tissue Engineering within the McGowan Institute. The focus of Dr. Badylak’s pioneering work has revolved around the structure and composition of naturally occurring ECM, and the signaling provided by this matrix to host cells toward functional tissue reconstruction. Dr. Badylak places high emphasis upon clinical translation of all activities in his laboratory, and work conducted there spans the full spectrum from basic science at the sub-cellular level to patient care at the bed side.
Watch the interview here.
Liver Transplants May Become Much Safer
 Although human liver transplants have been performed since 1963, it remains a complicated and risky surgery. About 28 percent of recipients die within 5 years. Now, a new procedure being developed at the University of Pittsburgh Medical Center is showing great promise by better preserving organs during the transplant process.
Although human liver transplants have been performed since 1963, it remains a complicated and risky surgery. About 28 percent of recipients die within 5 years. Now, a new procedure being developed at the University of Pittsburgh Medical Center is showing great promise by better preserving organs during the transplant process.
The pioneering research led by Paulo Fontes, MD, FACS, UPMC transplant surgeon, associate professor, Starzl Transplantation Institute, Department of Surgery, University of Pittsburgh School of Medicine, and a deputy director of the McGowan Institute for Regenerative Medicine, is in the development of a new hemoglobin-based oxygen carrier (HBOC) for organ and tissue preservation in combination with machine perfusion (MP).
A new HBOC molecule is being developed by VirTech Bio (VTB) LLC, Beverly, Massachusetts, under the leadership of W. Richard Light, PhD, in collaboration with faculty members from Ohio State University, Columbus, Ohio. Dr. Light, a leading HBOC scientist, has been collaborating with Dr. Fontes for several years in the development of this new approach to promote effective ex vivo oxygenation over an extended period of time. They initially developed and patented this technology in 2012, after a series of proof-of-concept pre-clinical experiments in liver transplantation at the McGowan Institute. They have further expanded this application for a discarded human liver and subsequently for vascularized composite allografts (VCA). The MP/HBOC technology is intended to enhance organ and tissue preservation prior to transplantation and it is expected to have a positive impact in clinical transplantation in the near future. Organ preservation is currently based on cold storage (CS), where the organs remain hypothermic and anoxic for a limited period of time. CS has a direct negative impact on organ quality and on the time and the distances imposed by the current system of organ allocation.
Watch the interview here.
Future of Trauma Care May Be in a Backpack
 In battlefield situations, taking even basic medical measurements may be complicated. Proper instruments, the knowledge of how to use them, and good decision-making skills are required. It would be great if soldiers had sophisticated, miniaturized medical devices that could easily be used to help an injured person.
In battlefield situations, taking even basic medical measurements may be complicated. Proper instruments, the knowledge of how to use them, and good decision-making skills are required. It would be great if soldiers had sophisticated, miniaturized medical devices that could easily be used to help an injured person.
If that were the case, said retired Army surgeon Ronald Poropatich, MD, “you can then assess the individual’s heart rate, blood pressure, and have a soft robotics collar with imaging devices in the neck that would tell me where the trachea is located, and have soft robotics with embedded sensors that tell me whether the lung is collapsed or not,” plus other vital information.
Dr. Poropatich, executive director of the Center for Military Medicine Research, Health Sciences at the University of Pittsburgh, professor of medicine in the Division of Pulmonary, Allergy, and Critical Care Medicine at the University of Pittsburgh, and senior advisor for Telemedicine, University of Pittsburgh Medical Center, is leading a team of researchers and engineers working on an “intelligent rucksack,” equipped with a computer, a range of sensors and robotic instruments, even syringes preloaded with a variety of medicines.
Read the interview here.
Voice of America (VOA) is the premier global broadcaster based in the United States. VOA and its entities average 203 million on-line visitors, TV viewers, and radio listeners per week. It produces 1,700 hours of news and feature programming weekly broadcast in 45 languages, including English.
Dr. Joon Sup Lee: Advancements in Cholesterol Control Help Move Cardiology Forward

Medical science and research has helped move many parts of cardiology from potentially fatal conditions to more chronic issues for patients and doctors to address. Cholesterol control is moving in that direction thanks to new advancements in medication and surgical options.
Joon Sup Lee, MD, co-director of the UPMC Heart and Vascular Institute, chief, Division of Cardiology at the University of Pittsburgh School of Medicine (UPMC), associate professor in the Department of Medicine at the University of Pittsburgh, and an affiliated faculty member of the McGowan Institute for Regenerative Medicine, discussed some of the highlights from this year’s meeting of the American College of Cardiology which was held in Chicago.
Located between the Cleveland Clinic and Philadelphia, Dr. Lee said the community-based approach UPMC takes to patient care is a small sampling of what life in the country is like as a whole and can help improve patient care elsewhere. Watch Dr. Lee’s video here as he answers these important questions:
- What are the biggest challenges in your field?
- What are your highlights from American College of Cardiology 2016?
- What can attendees take back to practice from this conference?
- What is the role of general cardiology?
- Are there certain areas that have seen more progress than others?
- What do you look to learn from the Hope3 Trial?
- How does your location affect your role in the field overall?
- What can patients do to improve their cardiovascular health?
- What is the future of this field?
Illustration: University of Pittsburgh Medical Center.
Pre-Surgical Exposure to Blue Light Reduces Organ Damage in Mice

According to new research from the University of Pittsburgh School of Medicine, a 24-hour exposure to bright blue light before surgery reduces inflammation and organ damage at the cellular level in a mouse model. The finding, reported recently in the Proceedings of the National Academy of Sciences, suggests a potential pre-treatment light therapy that could improve outcomes in patients undergoing procedures characterized by a period of blood restriction, such as liver resection or organ transplantation. McGowan Institute for Regenerative Medicine affiliated faculty member Derek Angus, MD, MPH, Distinguished Professor and the Mitchell P. Fink Endowed Chair of Critical Care Medicine at the University of Pittsburgh and the Director of the CRISMA (Clinical Research, Investigation, and Systems Modeling of Acute Illnesses) Center at the University of Pittsburgh, is a co-author on the study.
“We were incredibly surprised by our results,” said senior author Matthew R. Rosengart, MD, MPH, associate professor in the Pitt School of Medicine’s departments of Surgery and Critical Care Medicine. “There’s long been evidence suggesting that light and circadian rhythms profoundly influence our biology, and specifically the physiological response to stress. So while we were expecting to find some correlation with light spectrum and the immune response, we were not expecting results quite so striking.”
Light is complex and consists of intensity, duration of exposure, and wavelength. This study is one of the first that accounts for this complexity and derives results that could guide future clinical trials in humans.
Dr. Rosengart and his team compared what happened when mice were exposed to red light, ambient white fluorescent light similar to that in hospitals, and high-intensity blue light 24 hours before kidney or liver surgery involving periods of blood restriction and restoration.
The high-intensity blue light outperformed the red and white light, attenuating cellular and organ injury through at least two cellular mechanisms. The blue light brought about a reduction in the influx of neutrophils, a type of white blood cell involved in inflammation, which can lead to organ damage and other problems. Additionally, blue light inhibited dying cells from releasing a protein called HMGB1 that triggers organ-damaging inflammation.
The team then tested whether the blue light was acting through the optic pathway or some other mechanism, like the skin. Blind mice had the same healing response regardless of whether they were exposed to blue or red light, indicating that the protective impact of blue light does, indeed, act through the optic pathway.
The team then looked at whether one color of light might disrupt the circadian rhythm, which is linked to immunity, more than another. Blood from mice exposed to red, white, and blue light had similar concentrations of melatonin and corticosteroid hormones. Furthermore, the mice under each of the lights also had similar activity levels. These data indicate that the effects of blue light were not mediated by a disruption of sleep, activity, or circadian rhythms.
Finally, Dr. Rosengart stresses that mice are nocturnal animals with visual, circadian, and immune biology that is distinct from humans. Thus, the results of his study should not be broadly extended to patients or hospital settings until robust clinical trials have been performed to show whether or not pretreatment with intensive blue light is safe.
AWARDS AND RECOGNITION
Dr. Michel Modo Elected President, American Society for Neural Therapy and Repair

McGowan Institute for Regenerative Medicine affiliated faculty member Michel Modo, PhD, Associate Professor in the Departments of Radiology, Bioengineering, and the Center for Neural Basis of Cognition, University of Pittsburgh, has been elected president of the American Society for Neural Therapy and Repair and the chair of its 25th Anniversary Meeting in 2018. This will be a joint International Meeting with its sister societies Network of European CNS Transplantation and Restoration and the Pan Pacific Symposium on Stem Cells and Cancer Research.
Brain damage and neurodegenerative disease are disabling conditions that affect many millions of patients and their families. Yet, there is very little currently that can be done to reverse the effects of brain damage. As the Director of the Regenerative Imaging Laboratory at Pitt, Dr. Modo and his team are investigating whether some of the cells that have been lost can be replaced with transplanted neural stem cells (i.e. the cells that typical produce the brain during development) and can provide some recovery. Eventually they hope to have formulated methods that will allow the regeneration of lost brain tissue. The challenges for this are, nevertheless, considerable and Dr. Modo and his colleagues are but taking the very first steps in a long marathon to achieve this!
One key feature of tissue engineering in the brain is to inject cells and biomaterials into the right location where the damage is found. The skull, however, prevents easily seeing these locations and therefore there’s the need to use non-invasive imaging techniques that can peek deep into tissues. Magnetic Resonance Imaging (MRI) and Positron Emission Tomography (PET) are two of these techniques that can be used in pre-clinical feasibility studies, but the same techniques may one day be used on patients. Apart from locating where materials need injected, there is also the need to devise novel imaging methods that will allow identification of the materials injected and to monitor its development from a cells and biomaterials mixture into an integrated and functional brain tissue.
The American Society for Neural Therapy and Repair is dedicated to the development of pioneering biologic and reparative therapies to treat neurologic disorders. This is achieved through the utilization of novel cellular, gene, molecular, proteomic, and other biologic therapies. Members include visionaries and developing scientists who work in a collegial, collaborative manner. These relationships facilitate the rapid translation of research from the laboratory to the clinic, as well as the career advancement of young investigators in the field. These exchanges occur at annual scientific meetings, as well as at biannual social events.
Dr. Anthony Delitto Named SHRS Dean

Anthony Delitto, PhD, PT, FAPTA, has been appointed dean of the School of Health and Rehabilitation Sciences (SHRS). The former chair of the Department of Physical Therapy and SHRS associate dean for research had served as interim dean of the school since July 2015.
Dr. Delitto also is vice president for education and research at the UPMC Centers for Rehab Services (CRS). He is a member of the Board of Directors of the Pitt/UPMC Aging Institute and the steering committees of the Clinical Translational Science Institute and the Health Services Research Data Center of the Health Policy Institute. In addition, Dr. Delitto is a McGowan Institute for Regenerative Medicine affiliated faculty member.
Dr. Delitto began his career as a practicing physical therapist at the Irene Walter Johnson Institute of Rehabilitation and as a physical therapy faculty member at Washington University School of Medicine. He joined SHRS in 1992 as an assistant professor in the Department of Physical Therapy.
Over the past 25 years, Dr. Delitto’s research has focused on non-pharmacological management of painful musculoskeletal conditions, particularly low back pain. He and his colleagues pioneered research on a treatment-based classification system that combines physical and psychological principles in low back pain management.
His current research involves integrating the results of classification and treatment effectiveness studies into quality improvement initiatives, and he recently was awarded a multi-site $13 million Patient Centered Outcomes Research Institute (PCORI) grant to conduct a large pragmatic trial in which the primary aim is to reduce the transition from acute to chronic low back pain.
He received his BS in physical therapy from SUNY-Buffalo and his Master of Health Science degree in physical therapy and his PhD in social psychology, both from Washington University.
Congratulations, Dr. Delitto!
Students from McGowan Institute Laboratories Receive Whitaker International Program Awards

University of Pittsburgh Department of Bioengineering students Alex Josowitz, Saundria Moed, and Timothy Keane, PhD—students under the leadership of McGowan Institute for Regenerative Medicine affiliated faculty members David Vorp, PhD/Justin Weinbaum, PhD, Ian Sigal, PhD, and Stephen Badylak, DVM, PhD, MD, respectively—received awards from the Whitaker International Program. Mr. Josowitz and Ms. Moed were appointed as 2016 Whitaker Fellows, and Dr. Keane was appointed as the 2016 Whitaker Scholar.
Whitaker Fellows and Scholars spend 1 to 2 years abroad (outside of the U.S. and Canada) to conduct research in a university or laboratory, pursue coursework at an academic institution, or intern at a policy institute or in an industrial or non-profit setting.
The Whitaker award provides round-trip international airfare, a monthly living stipend, and other benefits to facilitate the students’ time abroad. Recipients of this national award must demonstrate outstanding scholarship, a strong background in research, and a well-thought out research plan for the duration of the fellowship to be selected.
“Whitaker Fellows and Scholars represent world-class student researchers and the future leaders of global collaboration in bioengineering,” said McGowan Institute for Regenerative Medicine faculty member Sanjeev Shroff, PhD, Distinguished Professor and Gerald McGinnis Chair of Bioengineering at Pitt. “These three students have an excellent opportunity not only to gain international experience but also to serve as ambassadors for Pitt research outside of the U.S.”
Mr. Josowitz will travel to the United Kingdom and work in the laboratory of Dr. Robert Krams at the Imperial College London. His research project will focus on optimizing the RNA isolation protocol for a microfluidic cell-capture system to provide new insight into the progression of cardiovascular diseases. Mr. Josowitz will also be enrolled in the Master of Research program in biomedical engineering at the Imperial College London. As an undergraduate student at Pitt, Mr. Josowitz has conducted research under the mentorship of Dr. Vorp, Associate Dean for Research and William Kepler Whiteford Professor of Bioengineering, and Dr. Weinbaum, research assistant professor. He has been working to develop and evaluate a novel technique of uniformly seeding 3D tubular constructs with cells or cell-like structures, such as microspheres, in tissue-engineered blood vessels.
Ms. Moed will travel to Israel and work in the laboratory of Prof. Rosa Azhari and Dr. Idit Golani at the Ort Braude College of Engineering in Karmiel, Israel. Her research project will focus on enhancing the delivery of active materials to the brain using nanoparticles. Ms. Moed participated in two independent research activities in Israel last summer at Ort Braude College of Engineering in Karmiel and at Technion Israel Institute of Technology in Haifa. As an undergraduate student at Pitt, Ms. Moed conducted research under the mentorship of Dr. Sigal, assistant professor of ophthalmology at the School of Medicine’s Ocular Biomechanics Laboratory. Her research involves analyzing optical coherence tomography images for quantifying ocular tissue microstructure, especially collagen fibers.
Dr. Keane will travel to the United Kingdom and work in the laboratory of Dr. Molly Stevens as a postdoctoral fellow at the Imperial College London. His work will focus on developing a biomaterial that can mitigate inflammation in the heart following myocardial infarction. As a PhD candidate at Pitt, Dr. Keane conducted his research work under the mentorship of Dr. Badylak, professor in the Department of Surgery and deputy director of the McGowan Institute. He is focusing on developing minimally invasive therapies for treating inflammatory diseases of the gastrointestinal tract.
Congratulations to all!
Dr. Bernhard Kühn Inducted into ASCI

McGowan Institute for Regenerative Medicine affiliated faculty member Bernhard Kühn, MD, has been inducted into the American Society for Clinical Investigation (ASCI), which was founded in 1908 and is “dedicated to the advancement of research that extends our understanding and improves the treatment of human diseases.”
Dr. Kühn is a board-certified pediatric cardiologist at Children’s Hospital of Pittsburgh of UPMC, a scholar at the Richard King Mellon Institute for Pediatric Research, and director of research, Division of Pediatric Cardiology. Dr. Kühn’s work is focused on the cells of the heart muscle and discovering ways to make them replicate to enable the heart to heal itself in cases of heart failure or congenital defects.
Election to ASCI reflects early career accomplishment as new members must be 50 or younger. ASCI represents physician-scientists who are “at the bedside, at the research bench, and at the blackboard.” Up to 80 new members are elected annually from hundreds of nominations.
Congratulations, Dr. Kühn!
Career Updates on Former McGowan Institute Students

Dr. Medberry:
Christopher Medberry, PhD, Senior Regulatory Affairs Specialist, DePuy Synthes Companies of Johnson and Johnson, is a presenter at the upcoming 2016 National Institute of Biomedical Imaging and Bioengineering (NIBIB) Training Grantees Meeting on July 11-12, 2016, Bethesda, Maryland. NIBIB is a part of the National Institutes of Health.
Dr. Medberry completed his PhD in Bioengineering at the McGowan Institute for Regenerative Medicine at the University of Pittsburgh in the lab of McGowan Institute Deputy Director Stephen Badylak, DVM, PhD, MD, Professor in the Department of Surgery and Director of the Center for Pre-Clinical Tissue Engineering within the Institute. Dr. Medberry’s research focused on the development and application of novel biomaterials, including hydrogels and scaffolds derived from nervous tissue extracellular matrix, and multiple preclinical models of traumatic central nervous tissue injury to investigate the mechanisms of biomaterial mediated tissue repair. His postdoctoral research explored therapeutic approaches that modulate the default healing response to prevent scarring and preserve or restore vision following injury and disease. Dr. Medberry is a graduate of the McGowan Institute’s Cellular Approaches to Tissue Engineering and Regeneration (CATER) Training Program.
Dr. Medberry worked as a Postdoctoral Fellow with the Coulter Translational Research Partners II Program at the University of Pittsburgh where he provided technical expertise and aided in the development of regulatory documents and in industry where he worked on medical device research, design, and development. He most recently completed the American Institute for Medical and Biological Engineering (AIMBE) Scholars Program, which is a Policy Fellowship Program at the U.S. Food and Drug Administration. As an AIMBE Scholar, Dr. Medberry utilized his technical background to influence decisions and help create policy within the Office of the Center Director, Center for Devices and Radiological Health. His long-term goal is to utilize his regulatory and policy background to foster innovative strategies for medical device development that improve patient care and clinical outcomes.
Dr. Hong:
A former post-doctoral associate and research assistant professor at the McGowan Institute for Regenerative Medicine is now a researcher at The University of Texas (UTA) at Arlington. Yi Hong, PhD, a UTA assistant professor of bioengineering, is developing a polymer that will allow engineers to develop a scaffold that is flexible, conductive, and biodegradable for biomedical applications such as tissue repair. Dr. Hong worked in the laboratory of McGowan Institute Director William Wagner, PhD, Professor of Surgery, Bioengineering, and Chemical Engineering at the University of Pittsburgh. Dr. Hong recently received a prestigious National Science Foundation Early CAREER grant to create conductive, single-component, and biodegradable elastomers. His technology is an advancement over conventional conductive polymers that are very stiff, hard to be processed, and non-degradable. The research holds great promise in biomedical fields such as tissue repair and drug delivery, but it also has potential to expand to biodegradable electronics and stretchable, wearable electronics.
“There is a gap between conductive polymers and biomedical technology, and many researchers have shown that conductivity can help in tissue remodeling,” Dr. Hong said. “My research will bridge the gap to design a new more conductive, biodegradable material made from a single polymer chain.”
Including his CAREER Award, Dr. Hong has been the primary investigator on research grants totaling more than $850,000 since beginning his career, including an American Heart Association grant to develop a bioactive heart patch that will help restore heart function after a heart attack. He also has been involved with other projects as a co-principal investigator in that time. His research interests include biomaterials, tissue engineering, medical devices, and drug delivery.
Dr. Hong holds 8 patents and has authored or co-authored more than 50 journal articles. He is a member of the Tissue Engineering and Regenerative Medicine International Society, the American Heart Association, the Biomedical Engineering Society, and the Society for Biomaterials. Dr. Hong earned his doctoral degree at Zhejiang University in 2005. While at Pitt, Dr. Hong received PITT Innovator Awards in 2009 and 2010. He joined the UTA Department of Bioengineering in 2012.
Regenerative Medicine Podcast Update
The Regenerative Medicine Podcasts remain a popular web destination. Informative and entertaining, these are the most recent interviews:
#159 –– Rebecca Davies, PhD is an Associate Professor in the Department of Veterinary Population Medicine – College of Veterinary Medicine at the University of Minnesota. Dr. Davies discusses her work in establishing quality assurance in an academic environment.
Visit www.regenerativemedicinetoday.com to keep abreast of the new interviews.
PICTURE OF THE MONTH
The Picture of the Month is a compliment to the longstanding features Grant of the Month and Publication of the Month. Each of these features highlights the achievements of McGowan affiliated faculty and their trainees. As we have always welcomed suggestions for grants and publications, please also consider submitting images that can highlight your pioneering work.
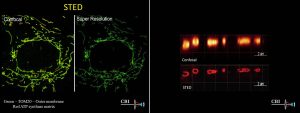
The CBI has just installed a new Leica STED3x super resolution microscope, increasing the image resolution up to 30 nm.
Here, in mitochondrial images made by Claudette St Croix, you can see the increase in resolution using STED compared to conventional confocal microscopy.
PUBLICATION OF THE MONTH
Author: Satish L, Palmer B, Liu F, Papatheodorou L, Rigatti L, Baratz ME, Kathju S
Title: Developing an animal model of Dupuytren’s disease by orthotopic transplantation of human fibroblasts into athymic rat
Summary:
BACKGROUND: Dupuytren’s disease (DD) is a slow, progressive fibroproliferative disorder affecting the palms of the hands. The disease is characterized by the formation of collagen rich- cords which gradually shorten by the action of myofibroblasts resulting in finger contractures. It is a disease that is confined to humans, and a major limiting factor in investigating this disorder has been the lack of a faithful animal model that can recapitulate its distinct biology. The aim of this study was to develop such a model by determining if Dupuytren’s disease (DD)- and control carpal tunnel (CT)-derived fibroblasts could survive in the forepaw of the nude rats and continue to exhibit the distinct characteristics they display in in vitro cultures.
METHODS: 1×10(7) fluorescently labeled DD- and CT-derived fibroblasts were transplanted into the left and right forepaws of nude rats respectively. Cells were tracked at regular intervals for a period of two months by quantifying emitted fluorescent signal using an IVIS imaging system. After a period of 62 days rat forepaw connective tissues were harvested for histology and total RNA was isolated. Human-specific probes were used to perform real time RT-PCR assays to examine the expression patterns of gene products associated with fibrosis in DD. Rat forepaw skin was also harvested to serve as an internal control.
RESULTS: Both CT- and DD-derived fibroblasts survived for a period of 62 days, but DD-derived cells showed a significantly greater level of persistent fluorescent signal at the end of this time than did CT-derived cells. mRNA expression levels of α-smooth muscle actin (α-SMA), type I- and type III- collagens were all significantly elevated in the forepaw receiving DD cord-derived fibroblasts in comparison to CT-derived fibroblasts. Masson’s trichrome stain confirmed increased collagen deposition in the forepaw that was injected with DD cord-derived fibroblasts.
CONCLUSIONS: For the first time we describe an animal model for Dupuytren’s disease at the orthotopic anatomical location. We further show that gene expression differences between control (CT) and diseased (DD) derived fibroblasts persist when these cells are transplanted to the forepaw of the nude rat. These preliminary findings indicate that, with further refinements, this animal model holds promise as a baseline for investigating novel therapeutic regimens to determine an effective strategy in treating DD.
Source: BMC Musculoskelet Disord. 2015 Jun 7;16:138.
GRANT OF THE MONTH
PI: Fabrisia Ambrosio, Bennett Van Houten
Co-I: Aaron Barchowsky, Mauricio Rojas, Donna Stolz
Title: The Anti-Aging Effect of Klotho on Skeletal Muscle Regeneration
Description:
An age-related impairment of the regenerative capacity of aged muscle is a major contributor to declines in functional mobility and is associated with an increased morbidity in an elderly population. Following an acute injury, young skeletal muscle initiates a highly effective regenerative response, which largely restores the original architecture of the damaged fibers. Conversely, with increasing age, the regenerative response to injury results in a considerable scar tissue deposition at the expense of functional contractile tissue. Much of this healing defect has been attributed to an age-related decrease in muscle stem, or satellite, cell (MuSC) functionality. In response to skeletal muscle injury, MuSCs become activated from a quiescent state to repair damaged myofibers. However, it has been suggested that the increased fibrosis deposition following injury is a result of a myogenic-to-fibrogenic conversion of MuSCs. Fortunately, these age-related changes are reversible.
Elegant studies employing heterochronic parabiosis, in which the circulatory systems of young and aged animals are conjoined, have revealed that rejuvenation of the systemic microenvironment significantly restores both whole tissue and MuSC regenerative capacity in aged muscle. These findings implicate that circulating factors, such as the longevity protein Klotho, play a critical role in dictating skeletal muscle regenerative potential over time. Elucidation of the origin and nature of circulating factors contributing to the aged muscle phenotype is critical for the development of strategies to prevent, delay or reverse age-related declines. These studies will evaluate the role of circulating Klotho as a key anti-geronic protein contributing to the benefit of heterochronic parabiosis on aged muscle regeneration.
In Specific Aim 1, we will test the hypothesis that aged parabionts paired with young mice heterozygously deficient for Klotho will display significantly impaired skeletal muscle regeneration when compared to aged parabionts paired with young wild type mice. In Specific Aim 2, we will test the hypothesis that restoration of circulating Klotho to youthful levels reverse age-related declines in skeletal muscle regenerative capacity. We anticipate that success in these aims will have a long and lasting impact on the field as they will establish Klotho as an important anti-geronic factor that regulates MuSC activity essential for functional muscle regeneration after injury.
Source: National Institute on Aging
Term: 5/01/16-4/30/2017
Source: $177,000
